Abstract
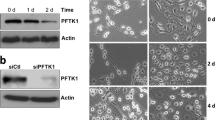
Oligodendrocytes (OLs) are glial cells that form myelin sheaths around axons in the central nervous system (CNS). Loss of the myelin sheath in demyelinating and neurodegenerative diseases can lead to severe impairment of movement. Understanding the extracellular signals and intracellular factors that regulate OL differentiation and myelination during development can help to develop novel strategies for enhancing myelin repair in neurological disorders. Here, we report that TAPP1 was selectively expressed in differentiating OL precursor cells (OPCs). TAPP1 knockdown promoted OL differentiation and myelin gene expression in culture. Conversely, over-expression of TAPP1 in immature OPCs suppressed their differentiation. Moreover, TAPP1 inhibition in OPCs altered the expression of Erk1/2 but not AKT. Taken together, our results identify TAPP1 as an important negative regulator of OPC differentiation through the Mek/Erk signaling pathway.
Similar content being viewed by others
References
Miron VE, Kuhlmann T, Antel JP. Cells of the oligodendroglial lineage, myelination, and remyelination. Biochim Biophys Acta 2010,1812: 184–193.
Nave KA. Myelination and support of axonal integrity by glia. Nature 2011, 468: 244–252.
Emery B. Regulation of oligodendrocyte differentiation and myelination. Science 2010, 330: 779–782.
Huang H, Zhao XF, Zheng K, Qiu M. Regulation of the timing of oligodendrocyte differentiation: mechanisms and perspectives. Neurosci Bull 2013, 29: 155–164.
Bruce CC, Zhao C, Franklin RJ. Remyelination - An effective means of neuroprotection. Horm Behav 2009, 57: 56–62.
Steinman L. Multiple sclerosis: a coordinated immunological attack against myelin in the central nervous system. Cell 1998, 85: 299–302.
Feng Y. Convergence and divergence in the etiology of myelin impairment in psychiatric disorders and drug addiction. Neurochem Res 2008, 33: 1940–1949.
Bartzokis G. Age-related myelin breakdown: a developmental model of cognitive decline and Alzheimer's disease. Neurobiol Aging 2004, 25: 5–18.
Sokolov BP. Oligodendroglial abnormalities in schizophrenia, mood disorders and substance abuse. Comorbidity,shared traits, or molecular phenocopies? Int J Neuropsychopharmacol 2007, 10: 547–555.
Zhu Q, Zhao XF, Zheng K, Li H, Huang H, Zhang Z, et al. Genetic evidence that Nkx2.2 and Pdgfra are major determinants of the timing of oligodendrocyte differentiation in the developing CNS. Development 2014, 141: 548–555.
Dowler S, Currie RA, Campbell DG, Deak M, Kular G, Downes CP, et al. Identification of pleckstrin-homologydomain- containing proteins with novel phosphoinositidebinding specificities. Biochem J 2000, 351: 19–31.
Hogan A, Yakubchyk Y, Chabot J, Obagi C, Daher E, Maekawa K, et al. The Phosphoinositol 3,4-Bisphosphatebinding protein TAPP1 interacts with syntrophins and regulates actin cytoskeletal organization. J Biol Chem 2004, 279: 53717–53724.
Schaeren-Wiemers N, Gerfin-Moser A. A single protocol to detect transcripts of various types and expression levels in neural tissue and cultured cells: in situ hybridization using digoxigenin-labeled cRNA probes. Histochemistry 1993, 100: 431–440.
Zhao X, Chen Y, Zhu Q, Huang H, Teng P Zheng K, et al. Control of astrocyte progenitor specification, migration and maturation by Nkx6.1 homeodomain transcription factor. PLoS ONE 2014, 9e109171.
Lu QR, Sun T, Zhu Z, Ma N, Garcia M, Stiles CD, et al. Common developmental requirement for Olig function indicates a motor neuron/oligodendrocyte connection. Cell 2002, 109: 75–86.
Bansal R, Magge S, Winkler S. Specific inhibitor of FGF receptor signaling: FGF-2-mediated effects on proliferation, differentiation, and MAPK activation are inhibited by PD173074 in oligodendrocyte lineage cells. J Neurosci Res 2003, 74: 486–493.
Baron W, Metz B, Bansal R, Hoekstra D, de Vries H. PDGF and FGF-2 signaling in oligodendrocyte progenitor cells: Regulation of proliferation and differentiation by multiple intracellular signaling pathways. Mol Cell Neurosci 2000, 15: 314–329.
Cui QL, Almazan G. IGF-I-induced oligodendrocyte progenitor proliferation requires PI3K/Akt, MEK/ERK, and Src-like tyrosine kinases. J Neurochem 2007, 100: 1480–1493.
Ebner S, Dunbar M, McKinnon RD. Distinct roles for PI3K in proliferation and survival of oligodendrocyte progenitor cells. J Neurosci Res 2000, 62: 336–345.
Flores AI, Narayanan SP, Morse EN, Shick HE, Yin X, et al. Constitutively active Akt induces enhanced myelination in the CNS. J Neurosci 2008, 28: 7174–7183.
Frost EE, Zhou Z, Krasnesky K, Armstrong RC. Initiation of oligodendrocyte progenitor cell migration by a PDGF-A activated extracellular regulated kinase (ERK) signaling pathway. Neurochem Res 2009, 34: 169–181.
Van´t Veer A, Du Y, Fischer TZ, Boetig DR, Wood MR, Dreyfus CF. Brain-derived neurotrophic factor effects on oligodendrocyte progenitors of the basal forebrain are mediated through trkB and the MAP kinase pathway. J Neurosci Res 2009, 87: 69–78.
Guardiola-Diaz HM, Ishii A, Bansal R. Erk1/2 MAPK and mTOR signaling sequentially regulates progression through distinct stages of oligodendrocyte differentiation. Glia 2012, 60: 476–486.
Chen Y, Wu H, Wang S, Koito H, Li J, Ye F, et al. The oligodendrocyte-specific G protein–coupled receptor GPR17 is a cell-intrinsic timer of myelination. Nat Neurosci 2009, 12: 1398–1406.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, Y., Mei, R., Teng, P. et al. TAPP1 inhibits the differentiation of oligodendrocyte precursor cells via suppressing the Mek/Erk pathway. Neurosci. Bull. 31, 517–526 (2015). https://doi.org/10.1007/s12264-015-1537-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-015-1537-5