Abstract
Malignant phyllodes tumors of the breast are fairly rare and fast-growing tumors. They are not sensitive to chemotherapy or hormonal therapy. Therefore, the primary treatment for malignant phyllodes tumors is wide surgical excision. Herein, we report a case study which featured a 26-year-old woman presented with a giant malignant phyllodes tumor measuring 20 × 17 × 13 cm. In order to reduce the chance of local recurrence, treatment for these types of tumors usually involves extensive excision with at least 1 cm of surgical margins. The patient underwent mastectomy with negative surgical margins, which left a large skin defect of 25 × 15 cm. Repair of such a large skin defect is a challenge to breast surgeons. This is the first reported case in which a “kiss” flap was successfully used to repair the skin defect created after resection of a giant malignant phyllodes tumor. The kiss flap could be considered as an effective and simple method to repair large chest wall defects after resection of giant phyllodes tumors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phyllodes tumors are fairly uncommon fibroepithelial neoplasms accounting for 0.3–0.9% of all breast tumors [1]. They are fast-growing tumors with benign, borderline, or malignant behavior, depending on histologic features including stromal cellularity, infiltration at the tumor’s edge, and mitotic activity. Their size ranges from 1 to 45 cm, and they may occupy the entire breast. Giant phyllodes tumors are typically defined as those > 10 cm in diameter [2]. Reports in the literature have been focused on surgical approaches to tumor resection. An adequate surgical margin is preferred with extended lumpectomy or mastectomy to prevent recurrence and metastasis. However, repair of the skin defect created after wide excision with at least 1 cm of surgical margins is a great challenge to breast surgeons. Repair with a pedicled latissimus dorsi kiss flap has not been reported in the literature (the kiss technique is first to harvest two or more small skin paddles from the donor site, which have the same size and shape, then rearrange them side-by-side at the recipient site, letting them “kiss” together to get a single, wide, unified flap, so named “kiss flap”). We report a case of giant malignant phyllodes tumor treated with mastectomy followed by successful defect repair with a kiss flap.
Patients and Techniques
Kiss flap is a surgical technique for patients who underwent enormous tumor resection and with huge skin defect. The surgery included a 26-year-old Chinese woman presented with a giant mass on her right breast, which had enlarged in the past year. This patient was included in September 18, 2016. We obtained written informed consent from the patient presented in this case report in accordance with the Helsinki Declaration of 1975. The patient consented to the treatment and the use of her data, including photos, for research and publication. This study was approved by the medical ethics committee.
Results
Her familial and personal history was non-contributory. Except for the breast tumor, laboratory tests showed no significant findings. Physical examination revealed a protruding and hardened palpable mass occupying almost the entire right breast (Fig. 1a). The mass measured approximately 20 × 17 × 13 cm. The skin had been stretched thin, with areas of ulceration and engorged superficial veins. Bilateral axillary lymph nodes were palpable, which are relatively soft and have good activities. A mammogram was not feasible because of the tumor size. Ultrasound of the breast mass indicated a heterogeneous echo and internal structures containing small cystic components, calcification, and hyperechoic separations, and was classified as BI-RADS 4 (Fig. 1b). Multiple enlarged hypoechoic bilateral axillary lymph nodes were detected, and some of them had lost their normal internal architecture and fatty hila. Magnetic resonance imaging (MRI) revealed a giant, lobulated mass with heterogeneous signal intensity, as well as septa and well-circumscribed margins, indicating typical features of phyllodes tumor (Fig. 1c). Image-guided core needle biopsy was performed twice, but a definite diagnosis was not obtained. Subsequently, a right breast excisional biopsy demonstrated a fibroepithelial lesion suspicious for a malignant phyllodes tumor. The results of immunohistochemical (IHC) staining on tumor biomarkers showed estrogen receptor negative (ER −), cytokeratin negative (CK −), CD34(+ / −), P63( −), and smooth muscle antibody negative (SMA −).
Imaging data of the patient. a Preoperative view. b Ultrasound of the right breast mass shows small cystic components, calcification, and hyperechoic separations. c Magnetic resonance imaging of the breast reveals a giant, lobulated mass with heterogeneous signal intensity, as well as septa and well-circumscribed margins, typical of a phyllodes tumor
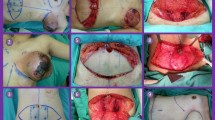
The patient underwent mastectomy and axillary lymph node biopsy. Before surgery, a “kiss” flap was carefully designed to cover the skin defect. First, we detected and localized the thoracodorsal artery by color Doppler ultrasound; then, a paper template of the huge skin defect was created. The template was then split into two small pieces of the same size and shape. These paper templates were strategically oriented onto the right back donor sites, allowing direct primary donor-site closure, and an exact match of the assembled flaps to the large defect. The skin defect was 25 × 15 cm. The template was used to mark the flaps on the patient’s right back. The location of the two-lobed flap depended on the vascular pedicle to ensure paddles were nourished by independent myocutaneous perforators from the thoracodorsal artery. An incision was made along the outlined semicircular skin island until the latissimus dorsi muscle was visualized, making sure both flaps were connected with the muscle. Before transposing the flaps to the chest wall through a subcutaneous tunnel, the two narrow skin paddles were arranged side by side and carefully sutured together to create a “kiss” flap. Finally, the donor site was sutured in layers, and the kiss flap was placed according to the design to cover the chest defect and sutured without tension (Fig. 2a–e). The wound recovered well (Fig. 2f, g). The resected primary tumor measured 18 × 14 × 14 cm (Fig. 2h). The cut surface of the tumor was white to gray with hemorrhagic foci (Fig. 3a). There was no involvement of the cutting edges, skin, tissue beneath the nipple, or superficial fascia. The sentinel lymph nodes were negative (0/4) with IHC biomarkers ER( −), CK( −), CD34(+ / −), P63( −), and SMA(-). Genetic testing showed no TP53 mutation. Postoperative pathology revealed a high-grade malignant phyllodes tumor with multifocal necrosis (Fig. 3b, c). After surgery, the patient was given six cycles of chemotherapy with docetaxel (160 mg), epirubicin (100 mg), and cyclophosphamide (800 mg). The patient also received twenty-five fractions of adjuvant chest irradiation treatment (total of 50 Gy). The patient was followed up for more than 4 years postoperatively, and there was no local recurrence or distant metastasis. Since she recovered well, the patient now is planning to have a delayed implant breast reconstruction plus fat grafting.
Postoperative imaging data: a The skin defect measuring 25 × 15 cm. b A double-paddle latissimus dorsi kiss flap is marked on the patient’s back, with each flap measuring 15 × 6 cm. The two flaps have an angle of about 90°. c Harvesting of parts A and B of the two-lobed flap. d Flaps A and B are sutured together before being transposed to the chest wound. e Immediate postoperative view. f Postoperative chest view at 3 months. g The back view. h The tumor measured 18 × 14 × 14 cm with massive hemorrhagic necrosis
Macroscopic findings and pathology images of the right breast tumor. a The cut surface of the tumor. b Tumor cells are mainly spindle cells, which are bidirectionally differentiated. c Tumor cells proliferate actively, mitotic figures are easy to see, and the cells are moderately and severely atypia. CHematoxylin and eosin, 200 × magnification Note. DHematoxylin and eosin, 400 × magnification
Discussion
Phyllodes tumor is a disease of the epithelial and stroma tissue in the breast. It is classified as benign, borderline, and malignant. Malignant tumors have high stroma cellularity and tend to be permeative, whereas benign tumors have low stroma cellularity and are circumscribed [3]. Malignant phyllodes tumors are distinguished only pathologically by identification of marked stromal cellularity, more than 5 mitoses per 10 high-powered fields, invasive margins, and marked stromal overgrowth [4]. Giant phyllodes tumors are rare fibroepithelial breast neoplasms typically > 10 cm in diameter by definition [2]. In general, it is difficult to differentiate phyllodes tumors from benign fibroadenoma by clinical presentation, radiology, or even core needle biopsy. The most accurate diagnosis of breast phyllodes tumor is postoperative pathology. Unlike breast cancer which can be downsized by neoadjuvant therapy, phyllodes tumor is not sensitive to chemotherapy, radiotherapy, or endocrine therapy [5, 6]. Surgery is regarded as the primary treatment method of phyllodes tumors. Negative margins rather than surgery type, such as extended lumpectomy or total mastectomy, determine the recurrence rate [7]. The National Comprehensive Cancer Network guidelines [8] advocate a wide excision with surgical margins of 1 cm or more. A negative margin is an independent prognostic factor for disease-free survival and local recurrence [9, 10]. Patients with a positive margin and malignant histology should undergo further surgery to obtain clear margins.
Extended lumpectomy or mastectomy with adequate surgical margins is the best choice for large malignant phyllodes tumors. Although a “fish-tail” incision has the ability to close even a larger skin defect as large as 15 cm × 15 cm without resorting to an additional flap [11], the resulting large skin defect always requires a skin graft or transplanted flap. To the best of our knowledge, this case is the first documented use of a kiss flap to repair the large skin defect resulting from removal of a giant malignant phyllodes tumor of the breast. There are some other options to repair the defect such as a transverse rectus abdominis myocutaneous (TRAM) flap or a deep inferior epigastric artery perforator (DIEP) flap. TRAM and DIEP can immediately reconstruct a new breast after mastectomy. Although DIEP and TRAM have little influence on pregnancy, the young girl has not been married or pregnant; she worried about a long scar on the abdominal wall would affect beauty and future pregnancy. Moreover, DIEP and TRAM need a long operative time and are highly traumatic, and DIEP requires microsurgical techniques. The young girl also worried about a quick recurrence soon after surgery, so she refused complicated immediate breast reconstruction such as DIEP and TRAM, and wanted to choose a simple procedure. The latissimus dorsi flap is close to the postoperative chest wound and usually be used to repair the chest wall defect after breast dissection. However, the skin of the back lacks elasticity; the donor area cannot be directly sutured and generally requires a skin graft if the width of the flap exceeds 8 cm. This prolongs postoperative recovery and limits the wide application of the latissimus dorsi flap. The kiss flap involves the excision of double skin paddles, which has an independent blood supply from the donor stem. These paddles are spliced in the recipient area; then, they “kiss” each other side-by-side to create a much larger flap, accurately matching the size of the defect. This technique allows flexible design of the flap shape, while increasing the surface area of skin flap coverage and minimizing incision dehiscence and non-healing complications. A careful presurgical flap design is necessary to make maximum use of the limited human tissue available and ensure minimum damage while performing the autologous tissue transfer. The functional and aesthetic outcome of the donor site should also be considered. In this case, the postoperative flap had a good appearance with no hyperplastic scar and the activities of shoulder joint were not affected. The outcome of this case suggests that the kiss flap is a simple and feasible technique for repair of large skin defect following giant phyllodes tumor resection. If there is no recurrence or metastasis more than one year after surgery, the patient may plan a delayed breast reconstruction. After communication with the patient, she planned to have a delayed implant breast reconstruction plus fat grafting. For some patients with small breasts, wing-shaped skin component of latissimus dorsi myocutaneous flap design may be a good choice since it not only facilitates the primary closure of the donor site defect in a VY or M-plasty manner but also creates a mild breast mound and may avoid the use of implants and its associated complications [12].
Local recurrence of phyllodes tumors has been associated with positive margins, younger age, larger tumor size, and malignant pathologic diagnosis [1, 13, 14]. The tumors rarely spread via the lymphatic system, and axillary lymph node metastasis rate is < 5%. Therefore, axillary lymph node dissection is unnecessary, yet the removal of suspicious axillary lymph nodes is recommended [15, 16]. In this case, since the tumor is too large, a sarcoma mixed with breast cancer components is not ruled out, and the axillary lymph nodes were palpable, so, we have an axillary sentinel lymph node biopsy. However, postoperative pathology proved that all the lymph nodes were negative. Studies have shown that adjuvant radiotherapy can lower the rate of local reoccurrence, particularly for patients with positive margins for borderline and malignant tumors [17,18,19]. Margin-negative resection combined with adjuvant radiotherapy is very effective for local control and prevention of recurrence. According to NCCN guidelines [8], adjuvant chemotherapy is not recommended since there is no evidence that adjuvant chemotherapy provides benefit in reduction of recurrence or metastasis. Adjuvant chemotherapy is not the standard care since it is of controversial value for malignant phyllodes tumors, yet some institutions support doxorubicin-based adjuvant chemotherapy for first-line treatment of breast sarcomas, especially with > 5.0 cm large high-risk tumors [20, 21]. During postoperative follow-up, no local recurrence or distant metastasis were found.
Conclusion
The kiss flap could be considered as an effective and simple method to repair large chest wall defects after resection of giant phyllodes tumors.
References
Ditsatham C, Chongruksut W (2019) Phyllodes tumor of the breast: diagnosis, management and outcome during a 10-year experience. Cancer Manag Res 11:7805–7811. https://doi.org/10.2147/CMAR.S215039
Kolia AK, Kalles V, Zografos CG et al (2020) Giant phyllodes tumor occupying the whole breast in a young lady: feasibility of mastectomy and immediate reconstruction. J Surg Case Rep 2020(4):rjz410. https://doi.org/10.1093/jscr/rjz410
Crenshaw SA, Roller MD, Chapman JK (2011) Immediate breast reconstruction with a saline implant and AlloDerm, following removal of a phyllodes tumor. World J Surg Oncol 9:34. https://doi.org/10.1186/1477-7819-9-34
Gordon PB, Gagnon FA, Lanzkowsky L (2003) Solid breast masses diagnosed as fibroadenoma at fine-needle aspiration biopsy: acceptable rates of growth at long-term follow-up. Radiology 229(1):233–238. https://doi.org/10.1148/radiol.2291010282
Ruiz-Flores L, Ebuoma LO, Benveniste MF et al (2018) Case report: metastatic phyllodes tumor. Semin Ultrasound CT MR 39(1):122–126. https://doi.org/10.1053/j.sult.2017.05.011
Yung RL, Hassett MJ, Chen K et al (2012) Initiation of adjuvant hormone therapy by Medicaid insured women with nonmetastatic breast cancer. J Natl Cancer Inst 104(14):1102–1105. https://doi.org/10.1093/jnci/djs273
Zhou ZR, Wang CC, Sun XJ et al (2018) Prognostic factors in breast phyllodes tumors: a nomogram based on a retrospective cohort study of 404 patients. Cancer Med 7(4):1030–1042. https://doi.org/10.1002/cam4.1327
Gradishar WJ, Moran MS, Abraham J et al (2022) Breast cancer, version 3.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 691–722. https://doi.org/10.6004/jnccn.2022.0030
Wurdinger S, Herzog AB, Fischer DR et al (2005) Differentiation of phyllodes breast tumors from fibroadenomas on MRI. AJR Am J Roentgenol 185(5):1317–1321. https://doi.org/10.2214/AJR.04.1620
Barth RJ Jr (2019) Borderline and malignant phyllodes tumors: how often do they locally recur and is there anything we can do about it? Ann Surg Oncol 26(7):1973–1975. https://doi.org/10.1245/s10434-019-07278-y
Piyush R, Rajendra M, Gopal P et al (2021) FISH mastectomy incision: an innovative AIIMS technique. Indian J Surg. https://doi.org/10.1007/s12262-020-02705-x
Shivangi S, Piyush R, Anita D et al (2021) Wing-shaped skin component of latissimus dorsi myocutaneous flap design for breast reconstruction—useful innovation. J Surg 83(6):1426–1432
Neron M, Sajous C, Thezenas S et al (2020) Surgical margins and adjuvant therapies in malignant phyllodes tumors of the breast: a multicenter retrospective study. Ann Surg Oncol 27(6):1818–1827. https://doi.org/10.1245/s10434-020-08217-y
Di Liso E, Bottosso M, Lo Mele M et al (2020) Prognostic factors in phyllodes tumours of the breast: retrospective study on 166 consecutive cases. ESMO Open 5(5):e000843. https://doi.org/10.1136/esmoopen-2020-000843
Ibreaheem MH, Naguib S, Gamal M, Boutrus R, Gomaa MMM, Talaat O (2020) Phyllodes tumors of the breast (the Egyptian experience). Indian J Surg Oncol 11(3):423–432. https://doi.org/10.1007/s13193-020-01107-5
Liew KW, Siti Zubaidah S, Doreen L (2018) Malignant phyllodes tumors of the breast: a single institution experience. Med J Malaysia 73(5):297–300
Wong RX, Koh YS, Wong FY et al (2020) The impact of radiotherapy and histological risk factors on outcomes in malignant phyllodes tumors. Clin Breast Cancer 20(6):e695–e700. https://doi.org/10.1016/j.clbc.2020.05.004
Choi N, Kim K, Shin KH et al (2018) Malignant and borderline phyllodes tumors of the breast: a multicenter study of 362 patients (KROG 16–08). Breast Cancer Res Treat 171(2):335–344. https://doi.org/10.1007/s10549-018-4838-3
Chao X, Chen K, Zeng J et al (2019) Adjuvant radiotherapy and chemotherapy for patients with breast phyllodes tumors: a systematic review and meta-analysis. BMC Cancer 19(1):372. https://doi.org/10.1186/s12885-019-5585-5
Abe H, Teramoto A, Takei Y, Tanaka Y, Yoneda G (2020) Malignant phyllodes tumor of the breast with rapid progression: a case report. Surg Case Rep 6(1):308. https://doi.org/10.1186/s40792-020-00986-8
Yılmaz S, Aykota MR, Arman Karakaya Y, Özgen U, Erdem E (2020) Phyllodes tumors of the breast: a single-center experience. Eur J Breast Health 17(1):36–41. https://doi.org/10.4274/ejbh.2020.5961
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Consent for Publication
Yes.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luo, Y., Zou, C., Hu, J. et al. The Defect Repair After a Giant Malignant Phyllodes Tumor Resection of Breast Using a Kiss Flap. Indian J Surg 85, 929–933 (2023). https://doi.org/10.1007/s12262-022-03560-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-022-03560-8