Abstract
Background
Amniotic membrane is considered a promising procedure as a graft in the field of ophthalmology and skin reconstruction. It has been shown to decrease inflammation, fibrosis, elicit no host immune reaction and also has antibiotic actions.
Aim
Recently, the amnion has been shown to contain cells capable of plasticity. Therefore, we felt its application could be extended to include surgical reconstruction.
Methods
We used ureteric reconstruction to study the feasibility of the amnion as a graft in these situations. We induced different degrees of injury in the ureter of dogs and reconstructed the defect with amniotic membrane. In addition, we also studied the effects of supplying the graft with a source of blood supply.
Results
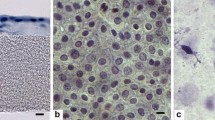
The subjects were sacrificed after 6 weeks and histopathological examination showed impressive repair of the defect.
Conclusion
The post reconstruction complications seen with present grafts were not seen with amniotic membrane. Therefore, amniotic membrane has produced noteworthy results and its potential should be further evaluated in order to be used as a cheap, readily available source of graft for the various surgical reconstruction procedures.
Similar content being viewed by others
References
Toda A, Okabe M, Yoshida T, Nikaido T (2007) The Potential of Amniotic Membrane/Amnion-derived cells for Regeneration of Various Tissues. J Pharmacol Sci 105(3): 215–228
Davis JW (1910) Skin transplantation with a review of 550 cases at the Johns Hopkins hospital. Johns Hopkins Med J 15:307–396
Trelford JD, Trelford-Sauder M (1979) The amnion in surgery, past and present. Am J Obstet Gynecol 134:833–846
Colocho G, Graham WP 3rd, Greene AE, Matheson DW, Lynch D (1974) Human amniotic membrane as a physiologic wound dressing. Arch Surg 109: 370–373
Faulk WP, Matthews R, Stevens PJ, Bennett JP, Burgos H, Hsi BL (1980) Human amnion as an adjunct in wound healing. Lancet 1:1156–1158
Subrahmanyam M (1995) Amniotic membrane as a cover for microskin grafts. Br J Plast Surg 48:477–478
Gruss JS, Jirsch DW (1978) Human amniotic membrane: a versatile wound dressing. Can Med Assoc J 118: 1237–1246
Ward DJ, Bennett JP (1984) The long-term Results of the use of human amnion in the treatment of leg ulcers. Br J Plast Surg 37:191–193
Ward DJ, Bennett JP, Burgos H, Fabre J (1989) The healing of chronic venous leg ulcers with prepared human amnion. Br J Plast Surg 42:463–467
Miyamoto K, Hayashi K, Suzuki T, Ichihara S, Yamada T, Kano Y, Yamabe T, Ito Y (2004) Human Placenta Feeder Layers Support Undifferentiated Growth of Primate Embryonic Stem Cells. Stem Cells 22:433–440. Cells 2007
Miki T, Lehmann T, Cai H et al. (2005) Stem Cell Characteristics of Amniotic Epithelial Cells. Stem Cells 23:1549–1559
Gekas C, Dieterlen-Lievre F, Orkin SH et al. (2005) The placenta is a Niche for Hematopoietic Stem Cells. Dev Cell 8:365–375
Tachibna M, Nagamatsu GR, Addonizio JC (1985) Ureteral replacement using collagen sponge tue grafts. J Urol 133:866
Fishman IJ, Flores FN, Brantly Scott F, Spjut HJ, Morrow B (1987) Use of fresh placental membranes for bladder reconstruction. J Urol 138:1291
Lord JW Jr, Eckel JH (1942) The use of vitallium tubes in the urinary tract of the dog. J Urol 48:412
Lubash (1947) Experience with tantalum tubes in the re implantation of the ureters into the sigmoid in dogs and humans. J Urol 57:1010
Solomon A, Wajngarten M, Alviano F, Anteby I, Elchalal U, Pe’er J, Levi-Schaffer F (2005) Suppression of Inflammatory and Fibrotic Responses in Allergic Inflammation by the Amniotic Membrane Stromal Matrix. Clin Exp Allergy 35(7):941–948
Lapides J, Caffery EL (1955) Observations on Healing of Ureteral Muscle: Relationship to Intubated Ureterotomy. J Urol 73:47
Trelford JD, Trelford-Saunder MV (1979) The amnion in surgery, past and present. AM J Obstet Gynecol 134:833
Weaver RG (1985) Ureteral regeneration: Experimental and clinical, Part III. J Urol 79:31
Lapides J, Caffery EL (1955) Observations on Healing of Ureteral Muscle: Relationship to Intubated Ureterectomy. J Urol 73:47
Hinman F Jr., Oppenheimer, R (1956) Smooth muscle regeneration in repair of experimental ureteral defects: the significance of double lumen. J Urol 75:428
His BL, Yeh CJ, Faulk WP (1982) Class antigens of the major histocompatibility complex on cytotrophoblast of human chorion leave. Immunology 53:621
Koziak A, Salagierski M, Marcheluk A, Szcześniewski R, Sosnowski M (2007) Early Experience in Reconstruction of Long Ureteral Strictures with Allogenic Amniotic Membrane. Int J Urol 14(7):607–611
His BL, Yeh CJ, Faulk WP (1982) Class antigens of the major histocompatibility complex on cytotrophoblast of human chorion leave. Immunology 53:621
Kubo M, Sonoda Y, Muramatsu R, Usui M (2001) Immunogenicity of Human Amniotic Membrane in Experimental Xenotransplantation. Invest Ophthalmol Vis Sci 42:1539–1546
Allan DS, Lepin EJ, Braud VM, O’Callaghan CA, McMichael AJ (2002) Tetrameric complexes of HLA-E, HLA-F, and HLA-G. J Immunol Methods 268:43–50
Colonna M, Samaridis J, Cella M, Angman L, Allen RL, O’Callaghan CA, Dunbar R, Ogg GS, Cerundolo V, Rolink A (1998) Human Myelomonocytic Cells Express anInhibitory Receptor for Classical and non Classical MHC class I molecules. J Immunol 160:3096–3100
Llano M, Lee N, Navarro F, Garcia P, Albar JP, Geraghty DE, Lopez-Botet M (1998) HLA-E-bound peptides influence recognition by inhibitory and triggering CD94/NKG2 receptors: Preferential Response to an HLA-G-derived Nonamer. Eur J Immunol 28:2854–2863
Fournel S, Aguerre-Girr M, Huc X, Lenfant F, Alam A, Toubert A, Bensussan A, Le Bouteiller P (2000) Cutting edge: Soluble HLA-G1 triggers CD95/CD95 Ligand Mediated Apoptosis in Activated CD8+ Cells by Interacting with CD8. J Immunol 164:6100–6104
Bainbridge DR, Ellis SA, Sargent IL (2000) HLA-G Suppresses Proliferation of CD4 (+) T-lymphocytes. J Reprod Immunol 48:17–26
Ueta M, Kweon MN, Sano Y, Sotozono C, Yamada J, Koizumi N, Kiyono H, Kinoshita S (2002) Immunosuppressive Properties of Human Amniotic Membrane for Mixed Lymphocyte Reaction. Clin Exp Immunol 129: 464–470
Alviano F, Fossati V, Marchionni C, Arpinati M, Bonsi L, Franchina M (2007) Term Amniotic Membrane is a high throughput source for Multipotent Mesenchymal Stem Cells with the ability to differentiate into Endothelial Cells in vitro. BMC Dev Biol 7:11
Loh YH, Wu Q, Chew JL et al. (2006) The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nat Genet 38:431–440
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ismail, A., Marcos, R.R., Sherif, A.A. et al. Amniotic stem cells repair ureteric defect: a study to evaluate the feasibility of amniotic membrane as a graft in surgical reconstruction. Indian J Surg 71, 121–127 (2009). https://doi.org/10.1007/s12262-009-0035-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-009-0035-2