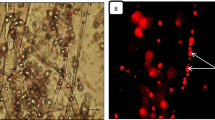
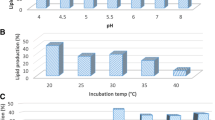
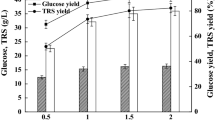
Abstract
In the present study, we evaluated extract of Jerusalem artichoke tubers (JAT) as a substrate for the production of lipid containing high levels of docosahexaenoic acid (DHA) by cultivated Aurantiochytrium sp. KRS101, an oleaginous protist. The optimal conditions for cultivation determined using response surface methods were as follows: pH, 5.9; enzyme loading, 282.6; and temperature, 27.7°C. The maximal levels of lipid (16.4 g/L) and productivity (3.6 g/L/d) were obtained during fed-batch fermentation, which achieved a DHA yield of 7.9 g/L (>48% of total fatty acids by weight). Moreover, JAT was more economical, reducing costs of expensive yeast extract as a nitrogen source by about 40%. These results suggest that JAT may be further explored as a low-cost feedstock for lipid production using microalgae strains.
Similar content being viewed by others
References
Simopoulos, A. P. (1999) Essential fatty acids in health and chronic disease. Am. J. Clin. Nut. 70: 560S–509S.
Innis, S. M. (2007) Fatty acids and early human development. Early Hum. Dev. 83: 761–766.
Helwani, Z., J.M. Othman, N. Aziz, M. J. N. Fernando, and J. Kim (2009) Technologies for production of biodiesel focusing on green catalytic techniques: a review. Furl. Proc. Technol. 90: 1193–1206.
Kim, K., J. E. Kim, B. G. Ryu, S. Park, Y. E. Choi, and J. W. Yang (2013) A novel fed–batch process based on the biology of Aurantiochytrium sp. KRS101 for the production of biodiesel and docosahexaenoic acid. Bioresour. Technol. 135: 269–274.
Barclay, W. R., K. M. Meager, and J. R. Abril (1994) Heterotrophic production of long–chain omega–3–fatty–acids utilizing algae and algae–like microorganisms. J. Appl. Phycol. 6: 123–129.
Amiri–Jami, M., G. Lapointe, and M. W. Griffiths (2014) Engineering of EPA/DHA omega–3 fatty acid production by Lactococcus lactis subsp. Cremoris MG1363. Appl. Microbiol. Biotechnol. 98: 3071–3080.
Ruiz–Lopez, N., O. Sayanova, J. A. Napier, and R. P. Haslam (2012) Metabolic engineering of the omega–3 long chin polyunsaturated fatty acid biosynthetic pathway into transgenic plants. J. Exp. Bot. 63: 2397–2410.
Hong, W. K., A. N. Yu, S. Y. Heo, B. R. Oh, C. H. Kim, J. H. Shon, J. W. Yang, A. Kondo, and J. W. Seo (2013) Production of lipids containing high levels of docosahexaenoic acid from empty palm fruit bunches by Aurantiochytrium sp. KRS101. Bioprocess Biosys. Eng. 36: 959–963.
Ratledge, C. (2004) Fatty acid biosynthesis in microorganisms being used for Single Cell Oil production. Biochimie 86: 807–815.
Chi, Z., B. Hu, Y. Liu, C. Frear, Z. Wen, and S. Chen (2007) Production of omega–3 polyunsaturated fatty acids from cull potato using an algae culture process. Appl. Biochem. Biotechnol. 137–140: 805–815.
Kim, S. H., J. M. Park, and C. H. Kim (2013) Ethanol production using whole plant biomass of Jerusalem artichoke by Kluyveromyces marxianus CBS1555. Appl. Biochem. Biotechnol. 169: 1531–1545.
Liang, Y., N. Sarkany, Y. Cui, J. Yesuf, J. Trushenski, and J. W. Blackburn (2010) Use of sweet sorghum juice for lipid production by Schizochytrium limacimum SR21. Bioresour. Technol. 101: 3623–3627.
Ryu, B. G., K. Kim, J. Kim, J. I. Han, and J. W. Yang (2013) Use of organic waste from the brewery industry for high–density cultivation of the docosahexaenoic acid–rich microalga, Aurantiochytrium sp. KRS101. Bioresour. Technol. 129: 351–359.
Chang, G., N. Gao, G. Tian, Q. Wu, M. Chang, and X. Wang (2013) Improvement of docosahexaenoic acid production on glycerol by Schizochytrium sp. S31 with constantly high oxygen transfer coefficient. Bioresour. Technol. 142: 400–406.
Scott, S. D., R. R. Armenta, K. T. Berryman, and A. W. Norman (2011) Use of raw glycerol to produce oil rich in polyunsaturated fatty acids by a thraustochytrid. Enz. Microb. Technol. 48: 267–272.
Swanton, C. J., P. B. Cavers, D. R. Clements, and M. J. Moore (1992) The biology of Canadian weeds, 101. Helianthus tuberous L. Can. J. Plant Sci. 72: 1367–1382.
Szambelan, K., J. Nowak, and Z. Czarnecki (2004). Use of Zymomonas mobilis and Saccharomyces cerevisiae mixed with Kluyveromyces fragilis for improved ethanol production from Jerusalem artichoke tubers. Biotechnol. Let. 26: 845–848.
Bekers, M., M. Grube, D. Upite, E. Kaminska, R. Linde, R. Scherbaka, and A. Danilevics (2007) Carbohydrates in Jerusalem artichoke powder suspension. Nutri. Food Sci. 37: 42–49.
Zhao, C. H., W. Cui, X. Y. Liu, Z. M. Chi, and C. Madzak (2010) Expression of inulinase gene in the oleaginous yeast Yarrowia lipolytica and single cell oil production from inulin–containing materials. Metab. Eng. 12: 510–517.
Zhao, X., S. Wu, C. Hu, Q. Wang, Y. Hua, and Z. K. Zhao (2010) Lipid production from Jerusalem artichoke by Rhodosporidium toruloides Y4. J. Ind. Microbiol. Biotechnol. 37: 581–585.
Sung, M., Y. H. Seo, S. Han, and J. I. Han (2014) Biodiesel production from yeast Cryptococcus sp. using Jerusalem artichoke. Bioresour. Technol. 155: 77–83.
Li, D., J. Y. Dai, and Z. L. Xiu (2010) A novel strategy for integrated utilization of Jerusalem artichoke stalk and tuber for production of 2.3–butanediol by Klebsiella pneumoniae. Bioresour. Technol. 101: 8342–8347.
Sun, L. H., X. D. Wang, J. Y. Dai, and Z. L. Xiu (2009) Microbial production of 2,3–butanediol from Jerusalem artichoke tubers by Klebsiella pneumoniae. Appl. Microbiol. Biotechnol. 82: 847–852.
Kaldy, M. S., A. Johnston, and D. B. Wilson (1980). Nutritive value of Indian bread–root, squaw–root, and Jerusalem artichoke. Econ. Bot. 34: 352–357.
Kim, J. K., B. R. Oh, H. J. Shin, C. Y. Eom, and S. W. Kim (2008) Statistical optimization of enzymatic saccharification and ethanol fermentation using food waste. Process Biochem. 43: 1308–1312.
Oh, B. R., J. W. Seo, S. Y. Heo, W. K. Hong, L. H. Luo, J. H. Son, D. H. Park, and C. H. Kim (2012) Fermentation strategies for 1,3–propanediol production from glycerol using a genetically engineered Klebsiella pneumoniae strain to eliminate by–product formation. Bioprocess Biosys. Eng. 35: 159–165.
Burja, A. M., H. Radianingtyas, A. Windust, and C. J. Barrow (2006) Isolation and characterization of polyunsaturated fatty acid producing Thraustochytrium species: screening of strains and optimization of omega–3 production. Appl. Microbiol. Biotechnol. 72: 1161–1169.
Park, J. M., B. R. Oh, I. Y. Kang, S. Y. Heo, J. W. Seo, S. M. Park, W. K. Hong, and C. H. Kim (2017) Enhancement of 2,3–butanediol production from Jerusalem artichoke tuber extract by a recombinant Bacillus sp. strain BRC1 with increased inulinase activity. Microbiol. Biotechnol. 44: 1107–1113.
Paul, G. C., C. A. Kent, and C. R. Thomas (1992) Quantitative characterization of vacuolization in Penicillium chyrsogenum using automatic image analysis. Food Bioprod. Process. 70: 13–20.
Margaritis, A. and P. Bajpai (1982) Ethanol production from Jerusalem artichoke tubers (Helianthus tuberosus) using Kluyveromyces marxianus and Saccharomyces rosei. Biotechnol. Bioeng. 24: 941–953.
Oh, B. R., S. M. Lee, S. Y. Heo, J. W. Seo, and C. H. Kim (2018) Efficient production of 1,3–propanediol from crude glycerol by repeated fed–batch fermentation strategy of lactate and 2,3–butanediol deficient mutant of Klebsiella penimoniae. Microb. Cell Fact. 17: 92–100.
Wang, G. Y., Z. Chi, B. Song, Z. P. Wang, and Z. M. Chi (2012) High level lipid productin by a novel inulinase–producing yeast Pichia guilliermondii Pcla22. Bioresour. Technol. 124: 77–82.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ju, JH., Oh, BR., Ryu, SK. et al. Production of Lipid Containing High Levels of Docosahexaenoic Acid by Cultivation of Aurantiochytrium sp. KRS101 Using Jerusalem Artichoke Extract. Biotechnol Bioproc E 23, 726–732 (2018). https://doi.org/10.1007/s12257-018-0419-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-018-0419-x