Abstract
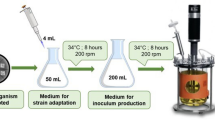
Ginsenoside compound K is an essential ingredient in nutritional supplements, cosmetics, and traditional medicines. However, cultivation for the production of enzymes involved in ginsenoside biotransformation has not been attempted in a fermenter. The host strain Escherichia coli ER2566 and the constitutive pHCE vector were selected for the efficient production of β-D-glycosidase, and expression medium composition to produce Sulfolobus solfataricus β-glycosidase expressed in E. coli was optimized in flask and batch cultures. The total activity of β-Dglycosidase in fed-batch culture using a fermenter increased 14-fold before optimization. S. solfataricus β-D-glycosidase and Thermotoga petrophila α-L-arabinofuranosidase were produced in a fed-batch culture. These two enzymes completely converted protopanaxadiol-type ginsenosides in ginseng leaf extract obtained from discarded ginseng leaves as a renewable substrate to compound K. The effective bioprocess for compound K production developed here will contribute to the industrial biological production of compound K.
Similar content being viewed by others
References
Lu, J. M., Q. Yao, and C. Chen (2009) Ginseng compounds: An update on their molecular mechanisms and medical applications. Curr. Vasc. Pharmacol. 7: 293–302.
Bae, E. A., M. K. Choo, E. K. Park, S. Y. Park, H. Y. Shin, and D. H. Kim (2002) Metabolism of ginsenoside Rc by human intestinal bacteria and its related antiallergic activity. Biol. Pharm. Bull. 25: 743–747.
Lee, S. Y., G. T. Kim, S. H. Roh, J. S. Song, H. J. Kim, S. S. Hong, S. W. Kwon, and J. H. Park (2009) Proteome changes related to the anti-cancer activity of HT29 cells by the treatment of ginsenoside Rd. Pharmazie 64: 242–247.
Yoshikawa, M., T. Morikawa, Y. Kashima, K. Ninomiya, and H. Matsuda (2003) Structures of new dammarane-type Triterpene Saponins from the flower buds of Panax notoginseng and hepatoprotective effects of principal Ginseng Saponins. J. Nat. Prod. 66: 922–927.
Wang, L., Y. Zhang, J. Chen, S. Li, Y. Wang, L. Hu, L. Wang, and Y. Wu (2012) Immunosuppressive effects of ginsenoside-Rd on skin allograft rejection in rats. J. Surg. Res. 176: 267–274.
Cho, W. C., W. S. Chung, S. K. Lee, A. W. Leung, C. H. Cheng, and K. K. Yue (2006) Ginsenoside Re of Panax ginseng possesses significant antioxidant and antihyperlipidemic efficacies in streptozotocin-induced diabetic rats. Eur. J. Pharmacol. 550: 173–179.
Kang, T. H., H. M. Park, Y. B. Kim, H. Kim, N. Kim, J. H. Do, C. Kang, Y. Cho, and S. Y. Kim (2009) Effects of red ginseng extract on UVB irradiation-induced skin aging in hairless mice. J. Ethnopharmacol. 123: 446–451.
Park, C. S., M. H. Yoo, K. H. Noh, and D. K. Oh (2010) Biotransformation of ginsenosides by hydrolyzing the sugar moieties of ginsenosides using microbial glycosidases. Appl. Microbiol. Biotechnol. 87: 9–19.
Son, J. W., H. J. Kim, and D. K. Oh (2008) Ginsenoside Rd production from the major ginsenoside Rb1 by β-glucosidase from Thermus caldophilus. Biotechnol. Lett. 30: 713–716.
Kim, M. K., J. W. Lee, K. Y. Lee, and D. C. Yang (2005) Microbial conversion of major ginsenoside Rb1 to pharmaceutically active minor ginsenoside Rd. J. Microbiol. 43: 456–462.
Shin, K. C. and D. K. Oh (2016) Classification of glycosidases that hydrolyze the specific positions and types of sugar moieties in ginsenosides. Crit. Rev. Biotechnol. 36: 1036–1049.
Ye, L., C. Q. Zhou, W. Zhou, P. Zhou, D. F. Chen, X. H. Liu, X. L. Shi, and M. Q. Feng (2010) Biotransformation of ginsenoside Rb1 to ginsenoside Rd by highly substrate-tolerant Paecilomyces bainier 229-7. Bioresour. Technol. 101: 7872–7876.
Wakabayashi, C., K. Murakami, H. Hasegawa, J. Murata, and I. Saiki (1998) An intestinal bacterial metabolite of ginseng protopanaxadiol saponins has the ability to induce apoptosis in tumor cells. Biochem. Biophy. Res. Commun. 246: 725–730.
Lee, H. U., E. A. Bae, M. J. Han, N. J. Kim, and D. H. Kim (2005) Hepatoprotective effect of ginsenoside Rb1 and compound K on tert-butyl hydroperoxide-induced liver injury. Liver Int. 25: 1069–1073.
Oh, S. H., H. Q. Yin, and B. H. Lee (2004) Role of the Fas/Fas ligand death receptor pathway in ginseng saponin metaboliteinduced apoptosis in HepG2 cells. Arch. Pharm. Res. 27: 402–406.
Lim, T. G., A. J. Jeon, J. H. Yoon, D. Song, J. E. Kim, J. Y. Kwon, J. R. Kim, N. J. Kang, J. S. Park, M. H. Yeom, D. K. Oh, Y. Lim, C. C. Lee, C. Y. Lee, and K. W. Lee (2015) 20-O-β-DGlucopyranosyl-20(S)-protopanaxadiol, a metabolite of ginsenoside Rb1, enhances the production of hyaluronic acid through the activation of ERK and Akt mediated by Src tyrosin kinase in human keratinocytes. Int. J. Mol. Med. 35: 1388–1394.
Shin, D. J., J. E. Kim, T. G. Lim, E. H. Jeong, G. Park, N. J. Kang, J. S. Park, M. H. Yeom, D. K. Oh, A. M. Bode, Z. Dong, H. J. Lee, and K. W. Lee (2014) 20-O-β-D-glucopyranosyl-20(S)-protopanaxadiol suppresses UV-Induced MMP-1 expression through AMPK-mediated mTOR inhibition as a downstream of the PKA-LKB1 pathway. J. Cell. Biochem. 115: 1702–1711.
Shin, K. C., H. Y. Choi, M. J. Seo, and D. K. Oh (2015) Compound K production from red ginseng extract by β-glycosidase from Sulfolobus solfataricus supplemented with α-Larabinofuranosidase from Caldicellulosiruptor saccharolyticus. PLoS One 10: e0145876.
Poo, H., J. J. Song, S. P. Hong, Y. H. Choi, S. W. Yun, J. H. Kim, S. C. Lee, S. G. Lee, and M. H. Sung (2002) Novel high-level constitutive expression system, pHCE vector, for a convenient and cost-effective soluble production of human tumor necrosis factor-alpha. Biotechnol. Lett. 24: 1185–1189.
Hsu, B. Y., T. J. Lu, C. H. Chen, S. J. Wang, and L. S. Hwang (2013) Biotransformation of ginsenoside Rd in the ginseng extraction residue by fermentation with lingzhi (Ganoderma lucidum). Food Chem. 141: 4186–4193.
Zhou, W., Q. Yan, J. Y. Li, X. C. Zhang, and P. Zhou (2008) Biotransformation of Panax notoginseng saponins into ginsenoside compound K production by Paecilomyces bainier sp. 229. J. Appl. Microbiol. 104: 699–706.
Choi, H. J., E. A. Kim, D. H. Kim, and K. S. Shin (2014) The bioconversion of red ginseng ethanol extract into compound K by Saccharomyces cerevisiae HJ-014. Mycobiol. 42: 256–261.
Noh, K. H. and D. K. Oh (2009) Production of the rare ginsenosides compound K, compound Y, and compound Mc by a thermostable β-glycosidase from Sulfolobus acidocaldarius. Biol. Pharm. Bull. 32: 1830–1835.
Noh, K. H., J. W. Son, H. J. Kim, and D. K. Oh (2009) Ginsenoside compound K production from ginseng root extract by a thermostable β-glycosidase from Sulfolobus solfataricus. Biosci. Biotechnol. Biochem. 73: 316–321.
Kim, B. H., S. Y. Lee, H. J. Cho, S. N. You, Y. J. Kim, Y. M. Park, J. K. Lee, M. Y. Baik, C. S. Park, and S. C. Ahn (2006) Biotransformation of Korean Panax ginseng by Pectinex. Biol. Pharm. Bull. 29: 2472–2478.
Kim, E. H., S. Lim, S. O. Kim, S. H. Ahn, and Y. J. Choi (2013) Optimization of enzymatic treatment for compound K production from white ginseng extract by response surface methodology. Biosci. Biotechnol. Biochem. 77: 1138–1140.
Shin, K. C., H. J. Oh, B. J. Kim, and D. K. Oh (2013) Complete conversion of major protopanaxadiol ginsenosides to compound K by the combined use of a-L-arabinofuranosidase and β-galactosidase from Caldicellulosiruptor saccharolyticus and β-glucosidase from Sulfolobus acidocaldarius. J. Biotechnol. 167: 33–40.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic Supplementary Material (ESM) The online version of this article (doi: 10.1007/s12257-018-0027-9) contains supplementary material, which is available to authorized users.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kim, TH., Yang, EJ., Shin, KC. et al. Enhanced Production of β-D-glycosidase and α-L-arabinofuranosidase in Recombinant Escherichia coli in Fed-batch Culture for the Biotransformation of Ginseng Leaf Extract to Ginsenoside Compound K. Biotechnol Bioproc E 23, 183–193 (2018). https://doi.org/10.1007/s12257-018-0027-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-018-0027-9