Abstract
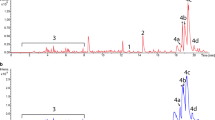
Heterologous expression can enhance production of diverse secondary metabolites by redirecting precursor pools towards compound of interest. In this study, Streptomyces venezuelae YJ028 was utilized as the heterologous host for the expression of four structural clavulanic acid biosynthesis genes, which encode carboxyethylarginine synthase (ceas2), β-lactam synthetase (bls2), clavaminate synthase (cas2), and proclavaminate amidinohydrolase (pah2). These genes were cloned into pIBR25 expression vector containing ermE* promoter to generate pBS4. The cas2 gene was also cloned into pSET152 to generate pCas2. It was then integrated into the genome of S. venezuelae YJ028. Upon metabolite profiling of recombinant strains by ultra-pressure liquid chromatography-photodiode array (UPLC-PDA) and high resolution liquid chromatography quadruple time-offlight electrospray ionization mass spectrometry (HR-LC-QTOF-ESI/MS), the production of following clavulanic acid intermediates in S. venezuelae recombinant were confirmed: deoxygaunidinoproclavaminic acid, guanidinoproclavaminic acid, and dihydroclavaminic acid. This work demonstrates the production of β-lactam intermediates of the clavulanic acid pathway by heterologous expression in S. venezuelae YJ028.
Similar content being viewed by others
References
Brown, A. G., D. Butterworth, M. Cole, G. Hanscomb, J. D. Hood, C. Reading, and G. N. Rolinson (1976) Naturally-occurring beta-lactamase inhibitors with antibacterial activity. J. Antibiot. 29: 668–669.
Townsend, C. A. (2002) New reactions in clavulanic acid biosynthesis. Curr. Opin. Chem. Biol. 6: 583–589
Bachmann, B. O., R. Li, and C. A. Townsend (1998) β-Lactam synthetase: A new biosynthetic enzyme. Proc. Natl. Acad. Sci. 95: 9082–9086.
Elander, R. P. (2003) Industrial production of β-lactam antibiotics. Appl. Microbiol. Biotechnol. 61: 385–392.
Li, R., N. Khaleeli, and C. A. Townsend (2000) Expansion of the clavulanic acid gene cluster: Identification and in vivo functional analysis of three new genes required for biosynthesis of clavulanic acid by Streptomyces clavuligerus. J. Bacteriol. 182: 4087–4095.
Mellado, E., L. M. Lorenzana, M. Rodrı́guez-Sáiz, B. Dı́ez, P. Liras, and J. L. Barredo (2002) The clavulanic acid biosynthetic cluster of Streptomyces clavuligerus: Genetic organization of the region upstream of the car gene. Microbiol. 148: 1427–1438.
Jensen, S. E. and A. S. Paradkar (1995) Biosynthesis and molecular genetics of clavulanic acid. Antonie Van Leeuwenhoek 75: 125–133.
Khaleeli, N., R. Li, and C. A. Townsend (1999) Origin of the β-lactam carbons in clavulanic acid from a unusual thiamine pyrophosphate-mediated reaction. J. Am. Chem. Soc. 121: 9223–9224.
Pérez-Redondo, R., A. Rodríguez-García, J. F. Martín, and P. Liras (1999) Deletion of the pyc gene blocks clavulanic acid biosynthesis except in glycerol-containing medium: Evidence for two different genes in formation of the C3 unit. J. Bacteriol. 181: 6922–6928.
Bachmann, B. O., R. Li, and C. A. Townsend (1998) β-Lactam synthetase: A new biosynthetic enzyme. Proc. Natl. Acad. Sci. 95: 9082–9086.
Baldwin, J. E., R. M. Adlington, J. S. Bryans, A. O. Bringhen, J. B. Coates, N. P. Crouch, M. D. Lloyd, C. J. Schofield, S. W. Elson, K. H. Baggaley, and R. Cassels (1991) Isolation of dihydroclavaminic acid, an intermediate in the biosynthesis of clavulanic acid. Tetrahedron 47: 4089–4100.
Marsh, E. N., M. D. Chang, and C. A. Townsend (1992) Two isozymes of clavaminate synthase central to clavulanic acid formation: cloning and sequencing of both genes from Streptomyces clavuligerus. Biochem. 31: 12648–12657.
Elson, S. W., K. H. Baggaley, M. Davison, M. Fulston, N. H. Nicholson, G. D. Risbridger, and J. W. Tyler (1993) The identification of three new biosynthetic intermediates and one further biosynthetic enzyme in the clavulanic acid pathway. J. Chem. Soc. Chem. Commun. 15: 1212–1214.
Aidoo, K. A., A. Wong, D. C. Alexander, R. A. Rittammer, and S. E. Jensen (1994) Cloning, sequencing and disruption of a gene from Streptomyces clavuligerus involved in clavulanic acid biosynthesis. Gene 147: 41–46.
Paradkar, A. (2013) Clavulanic acid production by Streptomyces clavuligerus: biogenesis, regulation and strain improvement. J. Antibiot. 66: 411–420.
Jensen S. E (2012) Biosynthesis of clavam metabolites. Ind. Microbiol. Biotechnol. 39: 1407–1419.
Álvarez-Álvarez, R., Y. Martínez-Burgo, R. Pérez-Redondo, A. F. Braña, J. F. Martín, and P. Liras (2013) Expression of the endogenous and heterologous clavulanic acid cluster in Streptomyces flavogriseus: Why a silent cluster is sleeping. Appl. Microbiol. Biotechnol. 97: 9451–9463.
Arulanantham, H., N. J. Kershaw, K. S. Hewitson, C. E. Hughes, J. E. Thirkettle, and C. J. Schofield (2006) ORF17 from the clavulanic acid biosynthesis gene cluster catalyzes the ATPdependent formation of N-glycyl-clavaminic acid. J. Biol. Chem. 281: 279–287.
MacNeil, D. J., J. L. Occi, K. M. Gewain, T. MacNeil, P. H. Gibbons, C. L. Ruby, and S. J. Danis (1992) Complex organization of the Streptomyces avermitilis genes encoding the avermectin polyketide synthase. Gene 115: 11925.
Sthapit, B., T. J. Oh, R. Lamichhane, K. Liou, H. C. Lee, C. G. Kim, and J. K. Sohng (2004) Neocarzinostatin naphthoate synthase: An unique iterative type I PKS from neocarzinostatin producer Streptomyces carzinostaticus. FEBS Lett. 566: 201–206.
Bierman, M., R. Logan, K. O’Brien, E. T. Seno, R.N. Rao, and B. E. Schoner (1992) Plasmid cloning vectors for the conjugal transfer of DNA from Escherichia coli to Streptomyces spp. Gene 116: 43–49.
Sambrook, J. and D. W. Russell (2001) Molecular cloning: A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, USA.
Keiser, T., M. J. Bibb, M. J. Buttner, K. F. Chater, and D. A. Hopwood (2000) Practical streptomyces genetics. The John Innes Foundation, Norwich.
Salowe, S. P., E. N. Marsh, and C. A. Townsend (1990) Purification and characterization of clavaminate synthase from Streptomyces clavuligerus: An unusual oxidative enzyme in natural product biosynthesis. Biochem. 29: 6499–6508.
Jung, W., A. Han, J. S. Hong, S. R. Park, C. Y. Choi, J. W. Park, and Y. J. Yoon (2007) Bioconversion of 12-, 14-,and 16-membered ring aglycones to glycosylated macrolidesin an engineered strain of Streptomyces venezuelae. Appl. Microbiol. Biotechnol. 76: 1373–1381.
Xue, Y. and D. H. Sherman (2001) Biosynthesis and combinatorial biosynthesis of pikromycin-related macrolides in Streptomyces venezuelae. Metab. Eng. 3: 15–26.
Maharjan, S., J. W. Park, Y. J. Yoon, H. C. Lee, and J. K. Sohng (2010) Metabolic engineering of Streptomyces venezuelae for malonyl-CoA biosynthesis to enhance heterologous production of polyketides. Biotechnol. Lett. 32: 277–282.
Pokhrel, A. R., D. Dhakal, A. K. Jha, and J. K. Sohng (2015) Herboxidiene biosynthesis, production, and structural modifications: prospect for hybrids with related polyketide. Appl. Microbiol. Biotechnol. 99: 8351–8362.
Jha, A. K., D. Dhakal, P. T. T. Van, A. R. Pokhrel, T. Yamaguchi, H. J. Jung, Y. J. Yoon, and J. K. Sohng (2015) Structural modification of herboxidiene by substrate-flexible cytochrome P450 and glycosyltransferase. Appl. Microbiol. Biotechnol. 99: 3421–3431.
Chaudhary, A. K., D. Dhakal, and J. K. Sohng (2013) An insight into the “-omics” based engineering of streptomycetes for secondary metabolite overproduction. BioMed Res. Int. 2013: 15.
Egan, L. A., R. W. Busby, D. Iwata-Reuyl, and C. A. Townsend (1997) Probable role of clavaminic acid as the terminal intermediate in the common pathway to clavulanic acid and the antipodal clavam metabolites. J. Am. Chem. Soc. 119: 2348–2355.
Saudagar, P. S., S. A. Survase, and R. S. Singhal (2008) Clavulanic acid: A review. Biotechnol. Adv. 26: 335–351.
Sohng, J. K. and D. Dhakal (2015) Commentary: Toward a new focus in antibiotic and drug discovery from the Streptomyces arsenal. Front. Microbiol. 6: 727.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shrestha, B., Dhakal, D., Darsandhari, S. et al. Heterologous production of clavulanic acid intermediates in Streptomyces venezuelae . Biotechnol Bioproc E 22, 359–365 (2017). https://doi.org/10.1007/s12257-017-0187-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0187-z