Abstract
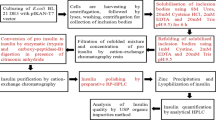
Vascular endothelial growth factors (VEGFs) are a family of proteins that promote angiogenesis and participate in a variety of physiological and pathological processes. In this work, the gene encoding the human VEGF isoform 165 (hVEGF165) was cloned into the expression vector pET32a (+) to construct a fusion expression plasmid that induced the thioredoxin (Trx) gene and transformed into Escherichia coli. The recombinant fusion protein TrxhVEGF165 was expressed optimally as inclusion bodies in the case of being cultivated for 4 h at 30°C and 1 mM IPTG concentration. The Trx-hVEGF165 was refolded and purified effectively from urea-solubilized inclusion bodies by the immobilized metal affinity chromatography. Released from the fusion protein by enterokinase cleavage and purified to homogeneity, the recombinant hVEGF165 (rhVEGF165) was biologically active as assessed by the human umbilicalvein endothelial cells (HUVECs) proliferation and the chicken chorioallantoic membrane (CAM) assay. The expression and in vitro refolding of rhVEGF165 resulted in production of an active molecule in a yield of 4.04 mg/L flask cultivation.
Similar content being viewed by others
References
Carmeliet, P. (2000) Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 6: 389–395.
Biselli-Chicote, P. M., A. R. C. P. Oliveira, E. C. Pavarino, and E. M. Goloni-Bertollo (2012) VEGF gene alternative splicing: Proand anti-angiogenic isoforms in cancer. J. Cancer Res. Clin. Oncol. 138: 363–370.
Houck, K. A., N. Ferrara, J. Winer, G. Cachianes, B. Li, and D. W. Leung (1991) The vascular endothelial growth factor family: Identification of a fourth molecular species and characterization of alternative splicing of RNA. Mol. Endocrinol. 5: 1806–1814.
Nowak, D. G., J. Woolard, E. M. Amin, O. Konopatskaya, M. A. Saleem, A. J. Churchill, M. R. Ladomery, S. J. Harper, and D. O. Bates (2008) Expression of pro- and anti-angiogenic isoforms of VEGF is differentially regulated by splicing and growth factors. J. Cell Sci. 121: 3487–3495.
Soker, S., H. Fidder, G. Neufeld, and M. Klagsbrun (1996) Characterization of novel vascular endothelial growth factor (VEGF) receptors on tumor cells that bind VEGF165 via its exon 7-encoded domain. J. Biol. Chem. 271: 5761–5767.
Ongay, S., A. Puerta, J.C. Díez-Masa, J. Bergquist, and M. de Frutos (2009) Development of CE methods to analyze potential components of the angiogenic glycoprotein vascular endothelial growth factor 165. Electrophoresis 30: 315–324.
Perrin, R. M., O. Konopatskaya, Y. Qiu, S. Harper, D. O. Bates, and A. J. Churchill (2005) Diabetic retinopathy is associated with a switch in splicing from anti- to pro-angiogenic isoforms of vascular endothelial growth factor. Diabetologia 48: 2422–2427.
Ferrara, N., K. A. Houck, L. B. Jakeman, J. Winer, and D. W. Leung (1991) The vascular endothelial growth factor family of polypeptides. J. Cell Biochem. 47: 211–218.
Carmeliet, P. (2003) Angiogenesis in health and disease. Nat. Med. 9: 653–660.
Belotti, D., P. Paganoni, L. Manenti, A. Garofalo, S. Marchini, G. Taraboletti, and R. Giavazzi (2003) Matrix metalloproteinases (MMP9 and MMP2) induce the release of vascular endothelial growth factor (VEGF) by ovarian carcinoma cells: Implications for ascites formation. Cancer Res. 63: 5224–5229.
Ferrara, N., J. Winer, and T. Burton (1991) Aortic smooth muscle cells express and secrete vascular endothelial growth factor. Growth Fact. 5: 141–148.
Tessler, S., P. Rockwell, D. Hicklin, T. Cohen, B. Levi, L. Witte, I. R. Lemischka, and G. Neufeld (1994) Heparin modulates the interaction of VEGF165 with soluble and cell associated flk-1 receptors. J. Biol. Chem. 269: 12456–12461.
Gitay-Goren, H., T. Cohen, S. Tessler, S. Soker, S. Gengrinovich, P. Rockwell, M. Klagsbrun, B. Z. Levi, and G. Neufeld (1996) Selective binding of VEGF to one of the three vascular endothelial growth factor receptors of vascular endothelial cells. J. Biol. Chem. 271: 5519–5523.
Soker, S., S. Takashima, H. Q. Miao, G. Neufeld, and M. Klagsbrun (1998) Neuropilin-1 is expressed by endothelial and tumer cells as an isoform-specific receptor for vascular endothelial growth factor. Cell 92: 735–745.
Siemeister, G., B. Schnurr, K. Mohrs, C. Schächtele, D. Marmé, and G. Martiny-Baron (1996) Expression of biologically active isoforms of the tumor angiogenesis factor VEGF in Escherichia coli. Biochem. Biophys. Res. Commun. 222: 249–255.
Pizarro, S. A., J. Gunson, M. J. Field, R. Dinges, S. Khoo, M. Dalal, M. Lee, K. A. Kaleas, K. Moiseff, S. Garnick, D. E. Reilly, M. W. Laird, and C. H. Schmelzer (2010) High-yield expression of human vascular endothelial growth factor VEGF165 in Escherichia coli and purification for therapeutic applications. Protein Expr. Purif. 72: 184–193.
Lee, I. L., P. S. Li, W. L. Yu, and H. H. Shen (2011) Prokaryotic expression, refolding, and purification of functional human vascular endothelial growth factor isoform 165: Purification procedures and refolding conditions revisited. Protein Expr. Purif. 76: 54–58.
Scrofani, S. D. B., L. J. Fabri, P. Xu, P. Maccarone, and A. D. Nash (2000) Purification and refolding of vascular endothelial growth factor-B. Protein Sci. 9: 2018–2025.
Backer, M. V. and J. M. Backer (2001) Functionally active VEGF fusion proteins. Protein Expr. Purif. 23: 1–7.
Cothran, A., R. J. St John, C. H. Schmelzer, and S. A. Pizarro (2011) High-pressure refolding of human vascular endothelial growth factor (VEGF) recombinantly expressed in bacterial inclusion bodies: Refolding optimization, and feasibility assessment. Biotechnol. Prog. 27: 1273–1281.
Sharapova, O. A., M. S. Yurkova, D. K. Laurinavichyute, S. M. Andronova, A. N. Fedorov, S. E. Severin, and E. S. Severin (2011) Efficient refolding of a hydrophobic protein with multiple S-S bonds by on-resin immobilized metal affinity chromatography. J. Chromatogr. A 1218: 5115–5119.
Lee, G. Y., W. W. Jung, C. S. Kang, and I. S. Bang (2006) Expression and characterization of human vascular endothelial growth factor (VEGF165) in insect cells, Protein Expr. Purif. 46: 503–509.
Rogl, H., K. Kosemund, and I. Collinson (1998) Refolding of E. coli produced membrane protein inclusion bodies immobilized by nickel chelating chromatography. FEBS Lett. 432: 21–26.
Bradford, M. M. (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 72: 248–254.
Laemmli, U. K. (1970) Cleavage of structural protein during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Towbin, H., T. Staehelin, and J. Gordon (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 76: 4350–4354.
Jeong, M. S., C. S. Kang, Y. S. Han, and I. S. Bang (2010) Expression, purification, and characterization of recombinant fibulin-5 in a prokaryote expression system. J. Microbiol. 48: 695–700.
Schoner, R. G., L. F. Ellis, and B. E. Schoner (1992) Isolation and purification of protein granules from Escherichia coli cells overproducing bovine growth hormone. Biotechnol. 24: 349–352.
Werner, M. H., G. M. Clore, A. M. Gronenborn, A. Kondoh, and R. J. Fisher (1994) Refolding proteins by gel filtration chromatography. FEBS Lett. 345: 125–130.
Rudolph, R. and H. Lilie (1996) In vitro folding of inclusion body proteins. FASEB J. 10: 49–56.
Cao, P., J. J. Mei, Z. Y. Diao, and S. Q. Zhang (2005) Expression, refolding, and characterization of human soluble BAFF synthesized in Escherichia coli. Protein Expr. Purif. 41: 199–206.
Stempfer, G., B. Holl-Neugebauer, and R. Rudolph (1996) Improved refolding of an immobilized fusion protein. Nat. Biotechnol. 14: 329–334.
Kweon, D. H., D. H. Lee, N. S. Han, and J. H. Seo (2004) Solidphase refolding of cyclodextrin glycosyltransferase adsorbed on cation-exchange resin. Biotechnol. Prog. 20: 277–283.
Ferrara, N., J. Winer, and T. Burton (1991) Aortic smooth muscle cells express and secrete vascular endothelial growth factor. Growth factors 5: 141–148.
Ferrara, N. and W. J. Henzel (1989) Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem. Biophys. Res. Commun. 161: 851–858.
Houck, K. A., N. Ferrara, J. Winer, G. Cachianes, B. Li, and D. W. Leung (1991) The vascular endothelial growth factor family: Identification of a fourth molecular species and characterization of alternative splicing of RNA. Mol. Endo. 5: 1806–1814.
Yeo, T. K., D. R. Senger, H. F. Dvorak, L. Freter, and K. T. Yeo (1991) Glycosylation is essential for efficient secretion but not for permeability-enhancing activity of vascular permeability factor (vascular endothelial growth factor). Biochem. Biophys. Res. Commun. 179: 1568–1575.
Lee, S. B., J. S. Park., S. H. Lee, J. H. Park, S. R. Yu, H. C. Kim, D. J. Kim, T. H. Byun, K. H. Baek, Y. J. Ahn, and J. S. Yoon (2008) Overproduction of recombinant human VEGF (vascular endothelial growth factor) in chinese hamster ovary cells. J. Microbiol. Biotechnol. 18: 183–187.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bang, S.K., Kim, Y.S., Chang, B.S. et al. Production and on-column re-folding of human vascular endothelial growth factor 165 in Escherichia coli . Biotechnol Bioproc E 18, 835–842 (2013). https://doi.org/10.1007/s12257-012-0829-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-012-0829-0