Abstract
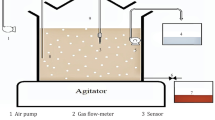
The batch bioreactor with a maximum capacity of 0.28 m3 was designed and manufactured according to oilfield conditions Acinetobacter johnsonii ioslated from waste oil in an aerobic environment was employed to cleanse waste oil and wash-down water using the bioreactor in a pilot scale. The aerating process was optimized from 2 to 4 m3/h corresponding to the different cell growth phases, ensuring sufficient dissolved oxygen and avoiding cell damage from the shearing forces due to strong aeration. The bio-treatment of waste oil underwent two stages:one was the wettability reversal of inorganic admixtures from oil phase into waster phase through enlarged wetting angles, and the other was the bio-flocculation in waster phase by intermediate metabolites excreted by cells. Finally, inorganic admixtures were effectively removed from waste oil and wash-down water at the optimal conditions, with the content less than 0.5% in oil phase and turbidity and sulfide less than 100 NTU and 1 mg/L in the water phase, respectively. Such bio-treatment made the oil-water interface clear against oil and water emulsification and in favor of smooth reclamations for waste oil and wash-down water.
Similar content being viewed by others
References
John, L. P. (2001) SPE65030 Iron sulfide scale deposit formation and prevention under anaerobic conditions typically found in the oil field. SPE International Symposium on Oilfield Chemistry. February 13. Houston, Texas.
Benka-Coker, M. O., W. Metseagharun, and J. A. Ekundayo (1995) Abundance of sulphate-reducing bacteria in Niger Delta oilfield waters. Bioresour. Technol. 54: 151–154.
Fakhru’l-Razi, A., A. Pendashteh, Z. Z. Abidin, L. C. Abdullah, D. R. A. Biak, and S. S. Madaeni (2010) Application of membrane-coupled sequencing batch reactor for oilfield produced water recycle and beneficial re-use. Bioresour. Technol. 101: 6942–6949.
Joseph, P. J. and A. Joseph (2009) Microbial enhanced separation of oil from a petroleum refinery sludge. J. Hazard. Mater. 161: 522–525.
Kiss, A. A. (2009) Novel process for biodiesel by reactive absorption. Sep. Purif. Technol. 69: 280–287.
Cuetos, M. J., C. Fernandez, X. Gomez, and A. Moran (2011) Anaerobic co-digestion of swine manure with energy crop residues. Biotechnol. Bioproc. Eng. 5: 1044–1052.
Haddar, A., N. Hmidet, O. Ghorbel-Bellaaj, N. Fakhfakh-Zouari, A. Sellami-Kamoun, and M. Nasri (2011) Alkaline proteases produced by Bacillus licheniformis RP1 grown on shrimp wastes: Application in chitin extraction, chicken featherdegradation and as a dehairing agent. Biotechnol. Bioproc. Eng. 4: 669–678.
Jiang, Y., X. Cai, D. Wu, and N. Q. Ren (2011) A method of inorganic admixture removal from waste oil by microorganism. CHN Patent ZL 2009 1 0223569.3.
Labille, J., F. Thomas, M. Milas, and C. Vanhaverbeke (2005) Flocculation of colloidal clay by bacterial polysaccharides: Effect of macromolecule charge and structur. J. Colloid Inter. Sci. 284: 149–156.
Zhang, W., R. C. Xiong, and G. Wei (2009) Biological flocculation treatment on distillery wastewater and recirculation of wastewater. J. Hazard. Mater. 172: 1252–1257.
Buthelezi, S. P., A. O. Olaniran, and B. Pillay (2010) Production and characterization of bioflocculants from bacteria isolated from wastewater treatment plant in South Africa. Biotechnol. Bioproc. Eng. 15: 874–881.
Liu, W. J., K. Wang, B. Z. Li, H. L. Yuan, and J. S. Yang (2010) Production and characterization of an intracellular bioflocculant by Chryseobacterium daeguense W6 culturd in low nutrition medium. Bioresour. Technol. 101: 1044–1048.
Xing, W., H. H. Ngo, W. S. Guo, Z. Q. Wu, T. T. Nguyen, P. Cullum, A. Listowski, and N. Yang (2010) Enhancement of the performance of anaerobic fluidized bed bioreactors (AFBBRs) by a new starch based flocculant. Sep. Purif. Technol. 72: 140–146.
Patra, P. and K. A. Natarajan (2008) Role of mineral specific bacterial proteins in selective flocculation and flotation. Int. J. Miner. Proc. 88: 53–58.
Seki, H., H. Maruyama, and Y. Shoji (2010) Flocculation of diatomite by a soy protein-based bioflocculant. Biochem. Eng. J. 51: 14–18.
Caposio, P., E. Pessione, G. Giuffrida, A. Conti, S. Landolfo, C. Giunta, and G. Gribaudo (2002) Cloning and characterization of two catechol 1,2-dioxygenase genes from Acinetobacter radioresistens S13. Res. Microbiol. 153: 69–74.
Vanbroekhoven, K., A. Ryngaert, P. Wattiau, De R. Mot, and D. Springael (2004) Acinetobacter diversity in environmental samples assessed by 16S rRNA gene PCR-DGGE fingerprinting. FEMS Microbiol. Ecol. 50: 37–50.
Jiang, Y., J. P. Wen, X. Q. Jia, Q. G. L. Caiyin, and Z. D. Hu (2007) Mutation of Candida tropicalis by irradiation with a He-Ne laser to increase its ability to degrade phenol. Appl. Environ. Microbiol. 73: 226–231.
Rena, C. Q., D. X. Liu, Z. Q. Bai, and T. H. Li (2005) Corrosion behavior of oil tube steel in stimulant solution with hydrogen sulfide and carbon dioxide. Mater. Chem. Phys. 93: 305–309.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, Y., Zhang, X., Chen, G. et al. The pilot study for waste oil removal from oilfields by Acinetobacter johnsonii using a specialized batch bioreactor. Biotechnol Bioproc E 17, 1300–1305 (2012). https://doi.org/10.1007/s12257-012-0232-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-012-0232-x