Summary
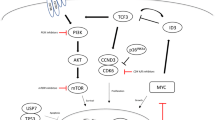
At the American Society of Hematology Meeting (ASH) 2018 promising data were presented reflecting the impressing advances of clinical research in aggressive lymphoma. Besides promising and partially practice-changing therapies in first-line treatment, there was a focus on Chimeric Antigen Receptor T Cell (CART) therapy for patients with relapsed/refractory (r/r) diffuse large B cell lymphoma (DLBCL): longer follow-up, real world data and new developments were reported. A bit overshadowed by CART therapy hype promising data for adding venetoclax to chemoimmuntherapy, bispecific antibodies, antibody drug conjugates and new monoclonal antibodies were presented. Important data to detect patients at high risk for R/CHOP (Rituximab/Cyclophospamide, Doxorubicin, Vincristine, Prednisone) failure and to detect possible targets for this high-risk subgroup were shown. The focus of phase III studies in the future should be on this high-risk population as the next phase III trial—the PHOENIX study adding ibrutinib to R/CHOP in non GCB DLBCL—failed to improve standard first-line therapy with R/CHOP.

Similar content being viewed by others
References
Rosenwald A, et al. Prognostic Significance of MYC Single, Double, Triple Hit and MYC-Translocation Partner Status in Diffuse Large B‑Cell Lymphoma—a Study By the Lunenburg Lymphoma Biomarker Consortium (LLBC). Blood. 2018;132:344.
Staiger AM, et al. A new Stromal signature applicable to formalin-fixed paraffin-embedded tissues identifies patients at risk in prospective clinical trials of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2018;132:343.
Poeschel V, et al. Excellent outcome of young patients (18–60 years) with favourable-prognosis diffuse large B‑cell Lymphoma (DLBCL) treated with 4 cycles CHOP plus 6 applications of Rituximab: results of the 592 patients of the flyer trial of the Dshnhl/GLA. Blood. 2018;132:781.
Younes A, et al. A global, randomized, placebo-controlled, phase 3 study of Ibrutinib plus rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (RCHOP) in patients with previously untreated non-germinal center B‑cell-like (GCB) diffuse large B‑cell Lymphoma (DLBCL). Blood. 2018;132:784.
Davies A, et al. Gene-expression profiling of bortezomib added to standard chemoimmunotherapy for diffuse large B‑cell lymphoma (REMoDL-B): an open-label, randomised, phase 3 trial. Lancet Oncol. 2019;20(5):P649–P62.
Leonard JP, et al. Randomized phase II study of R‑CHOP with or without Bortezomib in previously untreated patients with non-germinal center B‑cell-like diffuse large B‑cell Lymphoma. J Clin Oncol. 2017;35(31):3538–46.
Vitolo U, et al. Obinutuzumab or rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone in previously untreated diffuse large B‑cell lymphoma. J Clin Oncol. 2017;35(31):3529–37.
Witzig TE, et al. Adjuvant everolimus in high-risk diffuse large B‑cell lymphoma: final results from the PILLAR-2 randomized phase III trial. Ann Oncol. 2018;29(3):707–14.
Crump M, et al. Randomized, double-blind, phase III trial of enzastaurin versus placebo in patients achieving remission after first-line therapy for high-risk diffuse large B‑cell Lymphoma. J Clin Oncol. 2016;34(21):2484–92.
Franck Morschhauser, et al. Venetoclax plus rituximab, cyclophosphamide, doxorubicin, vincristine and prednisolone (R-CHOP) improves outcomes in BCL2-positive first-line diffuse large B‑cell Lymphoma (DLBCL): first safety, efficacy and Biomarker analyses from the phase II CAVALLI study. Blood. 2018;132:782.
Chamuleau MED, et al. Successful treatment of MYC rearrangement positive large B cell lymphoma patients with R‑CHOP21 plus lenalidomide: results of a multicenter phase II HOVON trial. Blood. 2018;132:786.
Locke FL, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B‑cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1–2 trial. Lancet Oncol. 2019;20(1):31–42.
Nastoupil LJ, et al. Axicabtagene ciloleucel (Axi-cel) CD19 chimeric antigen receptor (CAR) T‑cell therapy for relapsed/refractory large B‑cell lymphoma: real world experience. Blood. 2018;132:91.
Neelapu SS, et al. 2‑year follow-up and high-risk subset analysis of zuma-1, the pivotal study of axicabtagene ciloleucel (Axi-Cel) in patients with refractory large B cell lymphoma. Blood. 2018;132:2967.
Lung-Ji Chang, et al. Phase I/II trial of multi-target chimeric antigen receptor modified T cells (4SCAR2.0) against relapsed or refractory lymphomas. Blood. 2018;132:225.
Park JH, et al. A phase I first-in-human clinical trial of CD19-targeted 19-28z/4-1BBL “armored” CAR T cells in patients with relapsed or refractory NHL and CLL including richter’s transformation. Blood. 2018;132:224.
Henia Dar, et al. Preclinical development of CTX120, an allogeneic CAR-T cell targeting Bcma. Blood. 2018;132:1921.
Chong EA, et al. Sequential anti-CD19 directed chimeric antigen receptor modified T‑cell therapy (CART19) and PD-1 blockade with pembrolizumab in patients with relapsed or refractory B‑cell non-Hodgkin lymphomas. Blood. 2018;132:4198.
Hirayama AV, et al. Efficacy and toxicity of JCAR014 in combination with durvalumab for the treatment of patients with relapsed/refractory aggressive B‑cell non-hodgkin Lymphoma. Blood. 2018;132:1680.
Salles GA, et al. Single-arm phase II study of MOR208 combined with Lenalidomide in patients with relapsed or refractory diffuse large B‑cell lymphoma: L‑mind. Blood. 2018;132:227.
Caimi P, et al. Safety and efficacy of Venetoclax combined with rituximab, Ifosfamide, carboplatin and etoposide chemoimmunotherapy (VICER) for treatment of relapsed diffuse large B cell Lymphoma: results from the phase 1 study. Blood. 2018;132:397.
Sehn LH, et al. Polatuzumab Vedotin (Pola) plus Bendamustine (B) with Rituximab (R) or Obinutuzumab (G) in relapsed/refractory (R/R) diffuse large B‑cell Lymphoma (DLBCL): updated results of a phase (Ph) Ib/II study. Blood. 2018;132:1683.
Budde LE, et al. Mosunetuzumab, a full-length bispecific CD20/CD3 antibody, displays clinical activity in relapsed/refractory B‑cell non-Hodgkin lymphoma (NHL): interim safety and efficacy results from a phase 1 study. Blood. 2018;132:399.
Hutchings M, et al. CD20-Tcb (RG6026), a novel “2:1” format T‑cell-engaging Bispecific antibody, induces complete remissions in relapsed/refractory B‑cell non-hodgkin’s lymphoma: preliminary results from a phase I first in human trial. Blood. 2018;132:226.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Panny declares that he has no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Panny, M. ASH 2018—aggressive lymphoma. memo 12, 207–211 (2019). https://doi.org/10.1007/s12254-019-0510-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12254-019-0510-9