Abstract
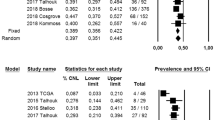
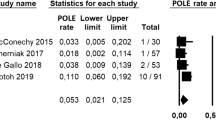
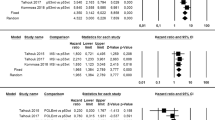
We aimed to classify undifferentiated/dedifferentiated carcinoma (UDC/DDC) according to the four TCGA molecular subgroups of endometrial cancer: microsatellite-instable/hypermutated (MSI), POLE-mutant/ultramutated (POLE), copy-number-low/p53-wild-type (p53wt), and copy-number-high/p53-abnormal (p53abn), through a systematic review and meta-analysis. Electronic databases were searched from January 2013 to July 2019 for studies assessing the TCGA classification in endometrial UDC/DDC series. Pooled prevalence of each TCGA subgroup on the total UDC/DDCs was calculated. Three studies with 73 patients were included. Pooled prevalence of the TCGA subgroups were: 12.4% for the POLE subgroup, 44% for the MSI subgroup, 18.6% for the p53abn subgroup, 25% for the p53wt group. All TCGA groups are represented in UDC/DDC, with a predominance of the MSI group, indicating a biological heterogeneity. Hypermutated/ultramutated cancers constitute the majority of UDC/DDC, suggesting a crucial difference with other high-risk histologies of endometrial carcinoma.



Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65(1):5–29
Travaglino A, Raffone A, Saccone G et al (2019) Immunohistochemical nuclear expression of β-catenin as a surrogate of CTNNB1 exon 3 mutation in endometrial Cancer. Am J Clin Pathol 151(5):529–538
Raffone A, Travaglino A, Mascolo M et al (2019) TCGA molecular groups of endometrial cancer: Pooled data about prognosis. Gynecol Oncol. https://doi.org/10.1016/j.ygyno.2019.08.019
Raffone A, Travaglino A, Santoro A et al (2019) Accuracy of one-step nucleic acid amplification in detecting lymph node metastases in endometrial Cancer. Pathol Oncol Res. https://doi.org/10.1007/s12253-019-00727-9
Travaglino A, Raffone A, Saccone G et al (2019) Immunohistochemical predictive markers of response to conservative treatment of endometrial hyperplasia and early endometrial cancer: a systematic review. Acta Obstet Gynecol Scand. https://doi.org/10.1111/aogs.13587
Cancer Genome Atlas Research Network et al (2013) Integrated genomic characterization of endometrial carcinoma. Nature 497(7447):67–73
Stelloo E, Nout RA, Osse EM et al (2016) Improved risk assessment by integrating molecular and clinicopathological factors in early-stage endometrial cancer-combined analysis of the PORTEC cohorts. Clin Cancer Res 22(16):4215–4224
Bosse T, Nout RA, McAlpine JN et al (2018) Molecular classification of grade 3 endometrioid endometrial cancers identifies distinct prognostic subgroups. Am J Surg Pathol 42(5):561–568
Cosgrove CM, Tritchler DL, Cohn DE et al (2018 Jan) An NRG oncology/GOG study of molecular classification for risk prediction in endometrioid endometrial cancer. GynecolOncol. 148(1):174–180
Talhouk A, McConechy MK, Leung S et al (2015) A clinically applicable molecular-based classification for endometrial cancers. Br J Cancer 113(2):299–310
Talhouk A, McConechy MK, Leung S et al (2017) Confirmation of ProMisE: a simple, genomics-based clinical classifier for endometrial cancer. Cancer. 123(5):802–813
Kommoss S, McConechy MK, Kommoss F et al (2018 May 1) Final validation of the ProMisE molecular classifier for endometrial carcinoma in a large population-based case series. AnnOncol. 29(5):1180–1188
Hoang LN, Kinloch MA, Leo JM et al (2017 Feb) Interobserver agreement in endometrial carcinoma Histotype diagnosis varies depending on the Cancer genome atlas (TCGA)-based molecular subgroup. Am J Surg Pathol 41(2):245–252
Raffone A, Travaglino A, Saccone G et al (2019) Management of women with atypical polypoid adenomyoma of the uterus: a quantitative systematic review. Acta Obstet Gynecol Scand. https://doi.org/10.1111/aogs.13553
Lionetti R, De Luca M, Travaglino A et al (2019 Jul) Treatments and overall survival in patients with Krukenberg tumor. Arch Gynecol Obstet 300(1):15–23
Travaglino A, Raffone A, Saccone G et al (2019) Nuclear expression of β-catenin in endometrial hyperplasia as marker of premalignancy. APMIS. https://doi.org/10.1111/apm.12988
Raffone A, Travaglino A, Saccone G et al (2019) Should progesterone and estrogens receptors be assessed for predicting the response to conservative treatment of endometrial hyperplasia and cancer? A systematic review and meta-analysis. Acta Obstet Gynecol Scand. https://doi.org/10.1111/aogs.13586
Travaglino A, Raffone A, Saccone A et al (2019) Immunophenotype of atypical Polypoid Adenomyoma of the uterus: diagnostic value and insight on pathogenesis. Appl Immunohistochem Mol Morphol. https://doi.org/10.1097/PAI.0000000000000780
Moher D, Shamseer L, Clarke M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic Reviews 4:1
Whiting PF, Rutjes AW, Westwood ME et al (2011) QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med 155(8):529–536
Travaglino A, Raffone A, Saccone G et al (2019) Congruence between 1994 WHO classification of endometrial hyperplasia and endometrial intraepithelial neoplasia system. Am J Clin Pathol. https://doi.org/10.1093/ajcp/aqz132
Raffone A, Travaglino A, Saccone G et al (2019) PTEN expression in endometrial hyperplasia and risk of cancer: a systematic review and meta-analysis. Arch Gynecol Obstet 299(6):1511–1524
Travaglino A, Raffone A, Mascolo M et al (2019) Clear cell endometrial carcinoma and the TCGA classification. Histopathology. https://doi.org/10.1111/his.13976
Travaglino A, Raffone A, Saccone G et al (2019) PTEN immunohistochemistry in endometrial hyperplasia: which are the optimal criteria for the diagnosis of precancer? APMIS 127(4):161–169
Raffone A, Travaglino A, Saccone G et al (2019) Endometrial hyperplasia and progression to cancer: which classification system stratifies the risk better? A systematic review and meta-analysis. Arch Gynecol Obstet 299(5):1233–1242
Travaglino A, Raffone A, Saccone G et al (2019) Significant risk of occult cancer in complex non-atypical endometrial hyperplasia. Arch Gynecol Obstet. https://doi.org/10.1007/s00404-019-05299-2
Raffone A, Travaglino A, Saccone G et al (201) Diabetes mellitus is associated with occult cancer in endometrial hyperplasia. Pathol Oncol Res. https://doi.org/10.1007/s12253-019-00684-3
Raffone A, Travaglino A, Saccone G et al (2019) Diagnostic and prognostic value of ARID1A in endometrial hyperplasia: a novel marker of occult cancer. APMIS. https://doi.org/10.1111/apm.12977
Raffone A, Travaglino A, Saccone G et al (2019) Diabetes mellitus and responsiveness of endometrial hyperplasia and early endometrial cancer to conservative treatment. Gynecol Endocrinol 5:1–6. https://doi.org/10.1080/09513590.2019.1624716
Meng B, Hoang LN, McIntyre JB et al (2014) POLE exonuclease domain mutation predicts long progression-free survival in grade 3 endometrioid carcinoma of the endometrium. Gynecol Oncol 134(1):15–19
Stewart CJ, Crook ML (2015) SWI/SNF complex deficiency and mismatch repair protein expression in undifferentiated and dedifferentiated endometrial carcinoma. Pathology. 47(5):439–445
Coatham M, Li X, Karnezis AN et al (2016) Concurrent ARID1A and ARID1B inactivation in endometrial and ovarian dedifferentiated carcinomas. Mod Pathol 29(12):1586–1593
Ramalingam P, Croce S, McCluggage WG (2017) Loss of expression of SMARCA4 (BRG1), SMARCA2 (BRM) and SMARCB1 (INI1) in undifferentiated carcinoma of the endometrium is not uncommon and is not always associated with rhabdoid morphology. Histopathology. 70(3):359–366
Rosa-Rosa JM, Leskelä S, Cristóbal-Lana E et al (2016) Molecular genetic heterogeneity in undifferentiated endometrial carcinomas. Mod Pathol 29(11):1390–1398
Espinosa I, Lee CH, D’Angelo E, Palacios J, Prat J (2017) Undifferentiated and dedifferentiated endometrial carcinomas with POLE exonuclease domain mutations have a favorable prognosis. Am J Surg Pathol 41(8):1121–1128
Köbel M, Hoang LN, Tessier-Cloutier B et al (2018) Undifferentiated endometrial carcinomas show frequent loss of core switch/sucrose nonfermentable complex proteins. Am J Surg Pathol 42(1):76–83
Murali R, Davidson B, Fadare O et al (2019) High-grade endometrial carcinomas: morphologic and Immunohistochemical features, diagnostic challenges and recommendations. Int J Gynecol Pathol 38 Suppl 1:S40–S63
Silverberg SG, Nogales F, Tavassoli FA, Devilee P (eds) (2003) Tumours of the uterine corpus. Pathology and genetics: Tumours of the breast and female genital organs. IARC Press. World Health Organization Classification of Tumours, Lyon, pp 217–257
Zaino R, Carinelli SG, Eng C, Kurman RJ, Carcangiu ML, Herrington CS, Young RH (eds) (2014) Tumours of the uterine corpus. WHO classification of tumours of female reproductive organs. IARC Press. World Health Organization Classification of Tumours, Lyon, pp 121–154
Karnezis AN, Hoang LN, Coatham M et al (2016) Loss of switch/sucrose non-fermenting complex protein expression is associated with dedifferentiation in endometrial carcinomas. Mod Pathol 29:302–314
Abu-Rustum NR, Yashar CM, Bean S, et al (2019) NCCN clinical practice guidelines in oncology (NCCN Guidelines®) – Uterine Neoplasms. Version 3.Feb 11 2019
Stelloo E, Bosse T, Nout RA et al (2015) Refining prognosis and identifying targetable pathways for high-risk endometrial cancer; a TransPORTEC initiative. Mod Pathol 28(6):836–844
Cherniack AD, Shen H, Walter V et al (2017) Integrated molecular characterization of uterine Carcinosarcoma. Cancer Cell 31(3):411–423
Hacking S, Jin C, Komforti M, Liang S, Nasim M (2019) MMR deficient undifferentiated/dedifferentiated endometrial carcinomas showing significant programmed death ligand-1 expression (sp 142) with potential therapeutic implications. Pathol Res Pract 22:152552. https://doi.org/10.1016/j.prp.2019.152552
Goodman AM, Kato S, Bazhenova L et al (2017) Tumor mutational burden as an independent predictor of response to immunotherapy in diverse cancers. Mol Cancer Ther 16(11):2598–2608
Author information
Authors and Affiliations
Contributions
AT, AR and MG independently assessed electronic search, eligibility of the studies, inclusion criteria, risk of bias, data extraction and data analysis. Disagreements were resolved by discussion with MM, LI, GFZ and FZ. MG, MM and LI contributed to the elaboration of methods for risk of bias assessment, data extraction and analysis. AT, AR and FZ conceived the study; MM, MG, LI and FZ worked on the design of the study; AT, AR, MG, MM, LI, GFZ and FZ worked on the manuscript preparation; GFZ and FZ supervised the whole study.
Corresponding author
Ethics declarations
Conflict of Interest
Authors report no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
Molecular/immunohistochemical features and TCGA subgroup assigned for each patient in the included studies. (DOCX 21 kb)
ESM 2
Flow diagram of studies identified in the systematic review (Prisma template [Preferred Reporting Item for Systematic Reviews and Meta-analyses]). (PNG 68 kb)
ESM 3
Summary of risk of bias for each study; Plus sign: low risk of bias; minus sign: high risk of bias; question mark: unclear risk of bias. (PNG 31 kb)
Rights and permissions
About this article
Cite this article
Travaglino, A., Raffone, A., Mascolo, M. et al. TCGA Molecular Subgroups in Endometrial Undifferentiated/Dedifferentiated Carcinoma. Pathol. Oncol. Res. 26, 1411–1416 (2020). https://doi.org/10.1007/s12253-019-00784-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-019-00784-0