Abstract
Background
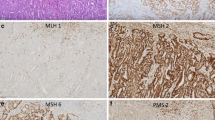
The Cancer Genome Atlas (TCGA) identified four prognostic subgroups of endometrial carcinoma: copy-number-low/p53-wild-type (p53wt), POLE-mutated/ultramutated (POLEmt), microsatellite-instability/hypermutated (MSI), and copy-number-high/p53-mutated (p53mt). However, it is still unclear if they may be integrated with the current histopathological prognostic factors, such as histotype.
Objective
To assess the impact of histotype on the prognostic value of the TCGA molecular subgroups of endometrial carcinoma.
Methods
A systematic review and meta-analysis was performed by searching 7 electronic databases from their inception to April 2019 for studies assessing prognosis in all TCGA subgroups of endometrial carcinoma. Pooled hazard ratio (HR) for overall survival (OS) was calculated in two different groups (“all-histotypes” and “endometrioid”), using p53wt subgroup as reference standard; HR for non-endometrioid histotypes was calculated indirectly. Disease-specific survival and progression-free survival were assessed as additional analyses.
Results
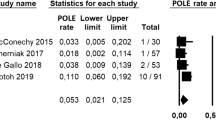
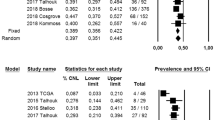
Six studies with 2818 patients were included. In the p53mt subgroup, pooled HRs for OS were 4.322 (all-histotypes), 2.505 (endometrioid), and 4.937 (non-endometrioid). In the MSI subgroup, pooled HRs were 1.965 (all-histotypes), 1.287 (endometrioid), and 6.361 (non-endometrioid). In the POLEmt subgroup, pooled HRs were 0.763 (all-histotypes), 0.481 (endometrioid), and 2.634 (non-endometrioid). Results of additional analyses were consistent for all subgroups except for non-endometrioid POLEmt carcinomas.
Conclusion
Histotype of endometrial carcinoma shows a crucial prognostic value independently of the TCGA molecular subgroup, with non-endometrioid carcinomas having a worse prognosis in each TCGA subgroup. Histotype should be integrated with molecular characterization for the risk stratification of patients in the future.



Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65(1):5–29
Travaglino A, Raffone A, Mascolo M et al (2019) TCGA Molecular subgroups in endometrial undifferentiated/dedifferentiated carcinoma. Pathol Oncol Res. https://doi.org/10.1007/s12253-019-00784-0(Epub ahead of print)
Gilks CB, Oliva E, Soslow RA (2013) Poor interobserver reproducibility in the diagnosis of high-grade endometrial carcinoma. Am J Surg Pathol 37:874–881
Hoang LN, McConechy MK, Kobel M et al (2013) Histotype-genotype correlation in 36 high-grade endometrial carcinomas. Am J Surg Pathol 37:1421–1432
Cancer Genome Atlas Research Network et al (2013) Integrated genomic characterization of endometrial carcinoma. Nature 497(7447):67–73
Talhouk A, McConechy MK, Leung S et al (2015) A clinically applicable molecular-based classification for endometrial cancers. Br J Cancer 113(2):299–310
Stelloo E, Nout RA, Osse EM et al (2016) Improved risk assessment by integrating molecular and clinicopathological factors in early-stage endometrial cancer-combined analysis of the PORTEC cohorts. Clin Cancer Res 22(16):4215–4224
Talhouk A, McConechy MK, Leung S et al (2017) Confirmation of ProMisE: a simple, genomics-based clinical classifier for endometrial cancer. Cancer 123(5):802–813
Bosse T, Nout RA, McAlpine JN et al (2018) Molecular classification of grade 3 endometrioid endometrial cancers identifies distinct prognostic subgroups. Am J Surg Pathol 42(5):561–568
Cosgrove CM, Tritchler DL, Cohn DE et al (2018) An NRG Oncology/GOG study of molecular classification for risk prediction in endometrioid endometrial cancer. Gynecol Oncol 148(1):174–180
Kommoss S, McConechy MK, Kommoss F et al (2018) Final validation of the ProMisE molecular classifier for endometrial carcinoma in a large population-based case series. Ann Oncol 29(5):1180–1188
Travaglino A, Raffone A, Saccone G et al (2019) Immunohistochemical nuclear expression of β-catenin as a surrogate of CTNNB1 exon 3 mutation in endometrial cancer. Am J Clin Pathol 151(5):529–538
Kim G, Kurnit KC, Djordjevic B et al (2018) Nuclear β-catenin localization and mutation of the CTNNB1 gene: a context-dependent association. Mod Pathol 31:1553–1559
Raffone A, Travaglino A, Saccone G et al (2019) Should progesterone and estrogens receptors be assessed for predicting the response to conservative treatment of endometrial hyperplasia and cancer? A systematic review and meta-analysis. Acta Obstet Gynecol Scand. https://doi.org/10.1111/aogs.13586(Epub ahead of print)
Raffone A, Travaglino A, Mascolo M, Insabato L, Zullo F (2020) Predictive accuracy of hormone receptors in conservatively treated endometrial hyperplasia and early endometrioid carcinoma. Acta Obstet Gynecol Scand 99(1):140
Travaglino A, Raffone A, Saccone G et al (2018) PTEN as a predictive marker of response to conservative treatment in endometrial hyperplasia and early endometrial cancer. A systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol 231:104–110
Travaglino A, Raffone A, Saccone G et al (2019) Immunohistochemical predictive markers of response to conservative treatment of endometrial hyperplasia and early endometrial cancer: a systematic review. Acta Obstet Gynecol Scand. https://doi.org/10.1111/aogs.13587(Epub ahead of print)
Raffone A, Travaglino A, Saccone G et al (2019) PTEN expression in endometrial hyperplasia and risk of cancer: a systematic review and meta-analysis. Arch Gynecol Obstet 299(6):1511–1524
Raffone A, Travaglino A, Saccone G et al (2019) PAX2 in endometrial carcinogenesis and in differential diagnosis of endometrial hyperplasia. A systematic review and meta-analysis of diagnostic accuracy. Acta Obstet Gynecol Scand 98(3):287–299
Travaglino A, Raffone A, Saccone G et al (2019) PTEN immunohistochemistry in endometrial hyperplasia: which are the optimal criteria for the diagnosis of precancer? APMIS 127(4):161–169
Raffone A, Travaglino A, Saccone G et al (2019) Loss of PTEN expression as diagnostic marker of endometrial precancer: a systematic review and meta-analysis. Acta Obstet Gynecol Scand 98(3):275–286
Raffone A, Travaglino A, D’Antonio A et al (2020) BAG3 expression correlates with the grade of dysplasia in squamous intraepithelial lesions of the uterine cervix. Acta Obstet Gynecol Scand 99(1):99–104
McAlpine J, Leon-Castillo A, Bosse T (2018) The rise of a novel classification system for endometrial carcinoma; integration of molecular subclasses. J Pathol 244(5):538–549
Köbel M, Nelson GS (2018) Letter in response to: McAlpine J, Leon-Castillo A, Bosse T. The rise of a novel classification system for endometrial carcinoma; integration of molecular subclasses. J Pathol 2018; 244: 538–549. J Pathol 245:249–250
Moher D, Shamseer L, Clarke M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4:1
Slim K, Nini E, Forestier D et al (2003) Methodological index for non-randomized studies (minors): development and validation of a newinstrument. ANZ J Surg 73(9):712–716
Raffone A, Travaglino A, Saccone G et al (2019) Endometrial hyperplasia and progression to cancer: which classification system stratifies the risk better? A systematic review and meta-analysis. Arch Gynecol Obstet 299(5):1233–1242
Raffone A, Travaglino A, Saccone G et al (2019) Management of women with atypical polypoid adenomyoma of the uterus: a quantitative systematic review. Acta Obstet Gynecol Scand. https://doi.org/10.1111/aogs.13553(Epub ahead of print)
Raffone A, Travaglino A, Saccone G et al (2019) Diabetes mellitus is associated with occult cancer in endometrial hyperplasia. Pathol Oncol Res. https://doi.org/10.1007/s12253-019-00684-3(Epub ahead of print)
Travaglino A, Raffone A, Saccone G et al (2019) Complexity of glandular architecture should be reconsidered in the classification and management of endometrial hyperplasia. APMIS 127(6):427–434
Travaglino A, Raffone A, Saccone G et al (2019) Congruence between 1994 who classification of endometrial hyperplasia and endometrial intraepithelial neoplasia system. Am J Clin Pathol. https://doi.org/10.1093/ajcp/aqz132(Epub ahead of print)
Travaglino A, Raffone A, Saccone G et al (2019) Endometrial hyperplasia and risk of coexistent cancer: WHO vs EIN criteria. Histopathology 74(5):676–687
Raffone A, Travaglino A, Saccone G et al (2019) Diabetes mellitus and responsiveness of endometrial hyperplasia and early endometrial cancer to conservative treatment. Gynecol Endocrinol. https://doi.org/10.1080/09513590.2019.1624716(Epub ahead of print)
Lionetti R, De Luca M, Travaglino A et al (2019) Treatments and overall survival in patients with Krukenberg tumor. Arch Gynecol Obstet 300(1):15–23
Raffone A, Travaglino A, Saccone G et al (2019) Diagnostic and prognostic value of ARID1A in endometrial hyperplasia: a novel marker of occult cancer. APMIS 127:597–606
Travaglino A, Raffone A, Mascolo M et al (2019) Clear cell endometrial carcinoma and the TCGA classification. Histopathology. https://doi.org/10.1111/his.13976(Epub ahead of print)
Raffone A, Travaglino A, Santoro A et al (2019) Accuracy of one-step nucleic acid amplification in detecting lymph node metastases in endometrial cancer. Pathol Oncol Res. https://doi.org/10.1007/s12253-019-00727-9(Epub ahead of print)
Travaglino A, Raffone A, Saccone G et al (2019) Nuclear expression of β-catenin in endometrial hyperplasia as marker of premalignancy. APMIS. https://doi.org/10.1111/apm.12988(Epub ahead of print)
Travaglino A, Raffone A, Saccone G et al (2019) Significant risk of occult cancer in complex non-atypical endometrial hyperplasia. Arch Gynecol Obstet. https://doi.org/10.1007/s00404-019-05299-2(Epub ahead of print)
Raffone A, Travaglino A, Mascolo M et al (2019) TCGA molecular groups of endometrial cancer: pooled data about prognosis. Gynecol Oncol 155(2):374–383
Hu S, Hinson JL, Matnani R, Cibull ML, Karabakhtsian RG (2018) Are the uterine serous carcinomas underdiagnosed? Histomorphologic and immunohistochemical correlates and clinical follow-up in high-grade endometrial carcinomas initially diagnosed as high-grade endometrioid carcinoma. Mod Pathol 31(2):358–364
Fadare O, Roma AA, Parkash V, Zheng W, Walavalkar V (2018) Does a p53 “wild-type” immunophenotype exclude a diagnosis of endometrial serous carcinoma? Adv Anat Pathol 25(1):61–70
Ritterhouse LL, Howitt BE (2016) Molecular pathology: predictive, prognostic, and diagnostic markers in uterine tumors. Surg Pathol Clin 9(3):405–426
Taskin OÇ, Onder S, Topuz S et al (2017) A selected immunohistochemical panel aids in differential diagnosis and prognostic stratification of subtypes of high-grade endometrial carcinoma: a clinicopathologic and immunohistochemical study at a single institution. Appl Immunohistochem Mol Morphol 25(10):696–702
Stewart CJ, Crook ML (2015) SWI/SNF complex deficiency and mismatch repair protein expression in undifferentiated and dedifferentiated endometrial carcinoma. Pathology 47(5):439–445
Rosa-Rosa JM, Leskelä S, Cristóbal-Lana E et al (2016) Molecular genetic heterogeneity in undifferentiated endometrial carcinomas. Mod Pathol 29(11):1390–1398
Espinosa I, Lee CH, D’Angelo E, Palacios J, Prat J (2017) Undifferentiated and dedifferentiated endometrial carcinomas with POLE exonuclease domain mutations have a favorable prognosis. Am J Surg Pathol 41(8):1121–1128
Köbel M, Hoang LN, Tessier-Cloutier B et al (2018) Undifferentiated endometrial carcinomas show frequent loss of core switch/sucrose nonfermentable complex proteins. Am J Surg Pathol 42(1):76–83
Wortman BG, Bosse T, Nout RA et al (2018) Molecular-integrated risk profile to determine adjuvant radiotherapy in endometrial cancer: evaluation of the pilot phase of the PORTEC-4a trial. Gynecol Oncol 151(1):69–75
Soumerai TE, Donoghue MTA, Bandlamudi C et al (2018) Clinical utility of prospective molecular characterization in advanced endometrial cancer. Clin Cancer Res 24(23):5939–5947
Stelloo E, Bosse T, Nout RA et al (2015) Refining prognosis and identifying targetable pathways for high-risk endometrial cancer; a TransPORTEC initiative. Mod Pathol 28(6):836–844
Murali R, Davidson B, Fadare O et al (2019) High-grade endometrial carcinomas: morphologic and immunohistochemical features, diagnostic challenges and recommendations. Int J Gynecol Pathol 38(Suppl 1):S40–S63
Singh N, Hirschowitz L, Zaino R et al (2019) Pathologic prognostic factors in endometrial carcinoma (other than tumor type and grade). Int J Gynecol Pathol 38(Suppl 1):S93–S113
Han KH, Kim HS, Lee M, Chung HH, Song YS (2017) Prognostic factors for tumor recurrence in endometrioid endometrial cancer stages IA and IB. Medicine (Baltimore) 96(21):e6976
Cuylan ZF, Oz M, Ozkan NT et al (2018) Prognostic factors and patterns of recurrence in lymphovascular space invasion positive women with stage IIIC endometriod endometrial cancer. J Obstet Gynaecol Res 44(6):1140–1149
Funding
No financial support was received for this study.
Author information
Authors and Affiliations
Contributions
AT and AR independently assessed electronic search, eligibility of the studies, inclusion criteria, risk of bias, data extraction, and data analysis. CS, RE, PM, CG, and GO contributed to the elaboration of methods for risk of bias assessment, data extraction, and analysis. AT, AR, LI, and FZ conceived the study; AT, AR, CS, LI, and FZ worked on the design of the study; AT, AR, RE, PM, and CG worked on the manuscript preparation; LI and FZ supervised the whole study.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
404_2020_5542_MOESM1_ESM.tif
Flow diagram of studies identified in the systematic review (Prisma template [Preferred Reporting Item for Systematic Reviews and Meta-analyses]). (TIFF 19 kb)
404_2020_5542_MOESM2_ESM.tif
Risk of bias within studies. Upper graph: Summary of risk of bias for each study; Plus sign: low risk of bias; minus sign: high risk of bias; question mark: unclear risk of bias. Lower graph: Risk of bias graph about each risk of bias item presented as percentages across all included studies (TIFF 22 kb)
404_2020_5542_MOESM3_ESM.tif
Forest plots of hazard ratio (HR) for disease-specific survival in the TCGA molecular subgroups of endometrial carcinoma (“all-histotypes” group) (TIFF 203 kb)
404_2020_5542_MOESM4_ESM.tif
Forest plots of hazard ratio (HR) for progression-free survival in the TCGA molecular subgroups of endometrial carcinoma (“all-histotypes” group) (TIFF 204 kb)
404_2020_5542_MOESM5_ESM.tif
Forest plots of hazard ratio (HR) for disease-specific survival in the TCGA molecular subgroups of endometrial carcinoma (“endometrioid” group) (TIFF 155 kb)
404_2020_5542_MOESM6_ESM.tif
Forest plots of hazard ratio (HR) for progression-free survival in the TCGA molecular subgroups of endometrial carcinoma (“endometrioid” group) (TIFF 182 kb)
404_2020_5542_MOESM7_ESM.tif
Scale of hazard ratio for disease-specific survival (DSS) in the TCGA subgroups of endometrial carcinoma stratified by histotype: endometrioid (pink squares), non-endometrioid (blue squares) or all histotypes (no squares) (TIFF 65 kb)
404_2020_5542_MOESM8_ESM.tif
Scale of hazard ratio for progression-free survival (PFS) in the TCGA subgroups of endometrial carcinoma stratified by histotype: endometrioid (pink squares), non-endometrioid (blue squares) or all histotypes (no squares) (TIFF 66 kb)
Rights and permissions
About this article
Cite this article
Travaglino, A., Raffone, A., Stradella, C. et al. Impact of endometrial carcinoma histotype on the prognostic value of the TCGA molecular subgroups. Arch Gynecol Obstet 301, 1355–1363 (2020). https://doi.org/10.1007/s00404-020-05542-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-020-05542-1