Abstract
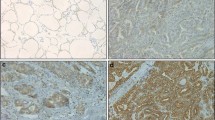
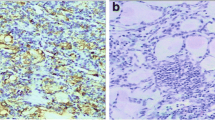
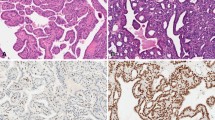
Thyroid cancer comprises a heterogeneous group of lesions with great diversity of biological behaviour. Markers which could help clinicians to identify high-risk patients for tailored optimization of clinical management are of crucial importance. HBME-1 protein level was analysed immunohistochemically using routinely prepared archival tissue sections of a broad range of papillary thyroid carcinoma (PTC) variants and in corresponding lymph node metastases (LNM). The results were evaluated in comparison with clinicopathological features of PTC. Positive immunoreaction was noticed in most classical (83/92; 90.2 %), follicular (60/71; 84.5 %) and trabecular (4/5; 80.0 %) variants of PTC. All cases of macrofollicular, Warthin-like and diffuse sclerosing PTC variants were HBME-1 positive (4/4, 3/3, 2/2; 100 % respectively). Tall cell and solid PTC variants showed diversity of staining (2/3; 66.67 % and 13/23; 56.52 % respectively), while PTCs with mixed histological pattern containing insular areas were mainly weakly positive (2/5; 40.0 %). A single case of clear cell PTC variant showed no reaction. Moreover, all matched metastatic PTC into lymph nodes (LNM) were HBME-1 positive (17/17; 100 %) and expressed HBME-1 in a similar pattern to the matched primary tumour. We also found a statistically significant association between high HBME-1 expression and the presence of lymph node metastasis, advanced pT status and pTNM stage (P < 0.05), but only a tendency for association with extrathyroidal invasion of the tumour (P = 0.058). Therefore, we recommend using immunoexpression of HBME-1 as useful mean to increase the likelihood of detecting most PTC variants and to predict some unfavourable clinical parameters in these patients.



Similar content being viewed by others
References
Siegel R, DeSantis C, Virgo K, Stein K, Mariotto A, Smith T, Cooper D, Gansler T, Lerro C, Fedewa S, Lin C, Leach C, Cannady RS, Cho H, Scoppa S, Hachey M, Kirch R, Jemal A, Ward E (2012) Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin 62:220–241. doi:10.3322/caac.21149
Treseler PA, Clark OH (1997) Prognostic factors in thyroid carcinoma. Surg Oncol Clin N Am 6:555–598
Gilliland FD, Hunt WC, Morris DM, Key CR (1997) Prognostic factors for thyroid carcinoma. A population-based study of 15,698 cases from surveillance, epidemiology and END results (SEER) program 1973–1991. Cancer 79:564–573
Sipos JA, Mazzaferri EL (2010) Thyroid cancer epidemiology and prognostic variables. Clin Oncol (R Coll Radiol) 22:395–404. doi:10.1016/j.clon.2010.05.004
Podnos YD, Smith D, Wagman LD, Ellenhorn JD (2005) The implication of lymph node metastasis on survival in patients with well-differentiated thyroid cancer. Am Surg 71:731–734
Fischer S, Asa SL (2008) Application of immunohistochemistry to thyroid neoplasms. Arch Pathol Lab Med 132:359–372. doi:10.1043/1543-2165(2008)132[359:AOITTN]2.0.CO;2
Barut F, Onak Kandemir N, Bektas S, Bahadir B, Keser S, Ozdamar S (2010) Universal markers of thyroid malignancies: galectin-3, HBME-1, and cytokeratin-19. Endocr Pathol 21:80–89. doi:10.1007/s12022-010-9114-y
Sethi K, Sarkar S, Das S, Mohanty B, Mandal M (2010) Biomarkers for the diagnosis of thyroid cancer. J Exp Ther Oncol 8:341–352
Cheng S, Serra S, Mercado M, Ezzat S, Asa S (2011) A high-throughput proteomic approach provides distinct signatures for thyroid cancer behavior. Clin Cancer Res 17:2385–2394. doi:10.1158/1078-0432.CCR-10-2837
Paunovic I, Isic T, Havelka M, Tatic S, Cvejic D, Savin S (2012) Combined immunohistochemistry for thyroid peroxidase, galectin-3, CK19 and HBME-1 in differential diagnosis of thyroid tumors. APMIS 120:368–379. doi:10.1111/j.1600-0463.2011.02842.x
Raphael SJ (2002) The meanings of markers: ancillary techniques in diagnosis of thyroid neoplasia. Endocr Pathol 13:301–311
de Matos PS, Ferreira AP, de Oliveira F, Assumpcao LVM, Matze K, Ward LS (2005) Usefulness of HBME-1, cytokeratin 19 and galectin-3 immunostaining in the diagnosis of thyroid malignancy. Histopathol 47:391–401
Saleh HA, Jin B, Barnwell J, Alzohaili O (2010) Utility of immunohistochemical markers in differentiating benign from malignant follicular-derived thyroid nodules. Diagn Pathol 5:9. doi:10.1186/1746-1596-5-9
Rossi ED, Raffaelli M, Mule A, Miraglia A, Lombardi CP, Vecchio FM, Fadda G (2006) Simultaneous immunohistochemical expression of HBME-1 and galectin-3 differentiates papillary carcinomas from hyperfunctioning lesions of the thyroid. Histopathology 48:795–800. doi:10.1111/j.1365-2559.2006.02428.x
Cheung CC, Ezzat S, Freeman JL, Rosen IB, Asa SL (2001) Immunohistochemical diagnosis of papillary thyroid carcinoma. Mod Pathol 14:338–342
Prasad M, Pellegata N, Huang Y, Nagaraja H, de la Chapelle A, Kloos R (2005) Galectin-3, fibronectin-1, CITED-1, HBME-1 and cytokeratin-19 immunohistochemistry is useful for the differential diagnosis of thyroid tumors. Mod Pathol 18:48–57
Scognamiglio T, Hyjek E, Kao J, Chen YT (2006) Diagnostic usefulness of HBME1, galectin-3, CK19, and CITED1 and evaluation of their expression in encapsulated lesions with questionable features of papillary thyroid carcinoma. Am J Clin Pathol 126:700–708
Nga ME, Lim GS, Soh CH, Kumarasinghe MP (2008) HBME-1 and CK19 are highly discriminatory in the cytological diagnosis of papillary thyroid carcinoma. Diagn Cytopathol 36:550–556. doi:10.1002/dc.20841
Choi YL, Kim MK, Suh JW, Han J, Kim JH, Yang JH, Nam SJ (2005) Immunoexpression of HBME-1, high molecular weight cytokeratin, cytokeratin 19, thyroid transcription factor-1, and E-cadherin in thyroid carcinomas. J Korean Med Sci 20:853–859
Rossi ED, Straccia P, Palumbo M, Stigliano E, Revelli L, Lombardi CP, Santeusanio G, Pontecorvi A, Fadda G (2013) Diagnostic and prognostic role of HBME-1, galectin-3, and β-catenin in poorly differentiated and anaplastic thyroid carcinomas. Appl Immunohistochem Mol Morphol 21:237–241. doi:10.1097/PAI.0b013e3182688d0f
Torregrossa L, Faviana P, Camacci T, Materazzi G, Berti P, Minuto M, Elisei R, Vitti P, Miccoli P, Basolo F (2007) Galectin-3 is highly expressed in nonencapsulated papillary thyroid carcinoma but weakly expressed in encapsulated type; comparison with Hector Battifora mesothelial cell 1 immunoreactivity. Hum Pathol 38:1482–1488
Hirokawa M, Horiguchi H, Wakatsuki S, Miki H, Sonoo H, Manabe T, Sano T (2001) Intranodal benign thyroid tissue: significance of HBME-1 in differentiation from metastatic papillary thyroid carcinoma. APMIS 109:875–880
Liang H, Zhong Y, Luo Z, Huang Y, Lin H, Zhan S, Xie K, Li QQ (2011) Diagnostic value of 16 cellular tumor markers for metastatic thyroid cancer: an immunohistochemical study. Anticancer Res 31:3433–3440
Cui W, Sang W, Zheng S, Ma Y, Liu X, Zhang W (2012) Usefulness of cytokeratin-19, galectin-3, and Hector Battifora mesothelial-1 in the diagnosis of benign and malignant thyroid nodules. Clin Lab 58:673–680
LiVolsi VA, Albores-Saavedra J, Asa SL et al (2004) Papillary carcinoma. In: DeLellis RA, Lloyd R, Heitz PU, Eng C (eds) World health organization classification of tumors. Pathology and genetics of tumors of endocrine organs. IARC Press, Lyon, pp 50–66
Volante M, Collini P, Nikiforov YE, Sakamoto A, Kakudo K, Katoh R, Lloyd RV, LiVolsi VA, Papotti M, Sobrinho-Simoes M, Bussolati G, Rosai J (2007) Poorly differentiated thyroid carcinoma: the Turin proposal for the use of uniform diagnostic criteria and an algorithmic diagnostic approach. Am J Surg Pathol 31:1256–1264
Edge SB, Byrd DR, Compton CC et al. (2010) (ed). AJCC cancer staging manual. In: Thyroid, 7thedn. Springer, New York, pp 87–96
Park YJ, Kwak SH, Kim DC, Kim H, Choe G, do Park J, Jang HC, Park SH, Cho BY, Park SY (2007) Diagnostic value of galectin-3, HBME-1, cytokeratin 19, high molecular weight cytokeratin, cyclin D1 and p27 (kip1) in the differential diagnosis of thyroid nodules. J Korean Med Sci 22:621–628. doi:10.3346/jkms.2007.22.4.621
Zhu X, Sun T, Lu H, Zhou X, Lu Y, Cai X (2010) Diagnostic significance of CK19, RET, galectin-3 and HBME-1 expression for papillary thyroid carcinoma. J Clin Pathol 63:786–789. doi:10.1136/jcp.2010.076901
de Matos LL, Del Giglio AB, Matsubayashi CO, de Lima FM, Del Giglio A, da Silva Pinhal MA (2012) Expression of CK-19, galectin-3 and HBME-1 in the differentiation of thyroid lesions: systematic review and diagnostic meta-analysis. Diagn Pathol 7:97. doi:10.1186/1746-1596-7-97, Review
Emad R, Maha A, Kfoury HK, Al-Sheikh AM, Zaidi SN (2011) Three cases of macrofollicular variant of papillary thyroid carcinoma. Ann Saudi Med 31:644–647. doi:10.4103/0256-4947.87104
Cardenas MG, Kini S, Wisgerhof M (2009) Two patients with highly aggressive macrofollicular variant of papillary thyroid carcinoma. Thyroid 19:413–416. doi:10.1089/thy.2008.0178
Ghossein R, Leboeuf R, Patel KN, Rivera M, Katabi N, Carlson DL, Tallini G, Shaha A, Singh B, Tuttle RM (2007) Tall cell variant of papillary thyroid carcinoma without extrathyroid extension: biologic behavior and clinical implications. Thyroid 17:655–661
Rigau V, Martel B, Evrard C, Rousselot P, Galateau-Salle F (2001) HBME-1 immunostaining in thyroid pathology. Ann Pathol 21:15–20
Ziadi S, Trimeche M, Zermani R, Elmay A, Baltagi-Ben Jilani S (2005) Diagnostic contribution of HBME-1 and anti-cytokeratin-19 antibodies in thyroid pathology: a retrospective study of 163 cases. Tunis Med 83:274–278
Acknowledgments
Supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia, project 173050: “Molecular characterization of thyroid gland tumours: biological and clinical aspects.”
The authors wish to express their gratitude to Dr Anna Nikolic, PhD for language correction of the manuscript.
We also wish to thank Prof M. Havelka and Prof S. Tatic from the Institute of Pathology, Medical Faculty, University of Belgrade, 11000 Belgrade, Serbia for help in revising the histological slides, and Dr D. Babic for his expert contribution to the statistical analyses.
Conflict of Interest
The authors declare that no conflict of interests exists.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Dencic, T.M.I., Savin, S.B., Selemetjev, S.A. et al. Strong Expression of HBME-1 Associates with High-Risk Clinicopathological Factors of Papillary Thyroid Carcinoma. Pathol. Oncol. Res. 21, 735–742 (2015). https://doi.org/10.1007/s12253-014-9883-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9883-6