Abstract
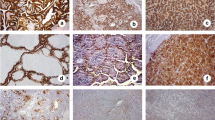
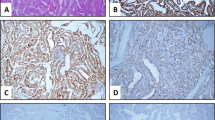
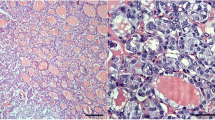
Cribriform morular variant of PTC (CMV-PTC) frequently shows activation of the CTNNB1/Wnt pathway with nuclear accumulation of beta catenin. The utility of LEF-1, also in the CTNNB1/WNT pathway, in the diagnosis of CMV-PTC has not been previously studied. LEF-1 immunohistochemistry was performed on seven CMV-PTC, 52 benign cases and 101 malignant thyroid neoplasms. LEF-1 was scored by stain intensity (0 = no nuclear stain, 1 = weak nuclear stain, less than lymphocyte and 2 = strong nuclear stain, intense as lymphocyte) and percentage of positive cells at each intensity, for a maximum total score of 200. Sensitivity and specificity of LEF-1 stain for all cases and to differentiate between regular PTC and CMV-PTC was also calculated. Six of the seven CMV-PTCs showed ≥ 30% strong (2+) nuclear LEF-1 staining and a total score over 100. Beta catenin also showed strong and diffuse nuclear staining in these cases. One CMV-PTC was negative for both LEF-1 and beta catenin and did not have a history of FAP. All control PTC cases uniformly lacked LEF-1 staining at 2+ intensity. LEF-1 had a sensitivity of 86% and specificity of 98% for the diagnosis of CMV-PTC. LEF-1 is highly sensitive and specific marker for CMV-PTC, especially when used in the setting of a PTC neoplasm. The pattern of staining is important with ≥ 30% of cells showing strong 2+ nuclear staining having the highest combined sensitivity and specificity.




Similar content being viewed by others
References
Pradhan D, Sharma A, Mohanty SK. Cribriform-morular variant of papillary thyroid carcinoma. Pathol Res Pract. 2015;211(10):712–6.
Harach HR, Williams GT, Williams ED. Familial adenomatous polyposis associated thyroid carcinoma: a distinct type of follicular cell neoplasm. Histopathology. 1994;25(6):549–61.
Cameselle-Teijeiro J, Chan JK. Cribriform-morular variant of papillary carcinoma: a distinctive variant representing the sporadic counterpart of familial adenomatous polyposis-associated thyroid carcinoma? Mod Pathol. 1999;12(4):400–11.
Boonyaarunnate T, Olson MT, Bishop JA, Yang GC, Ali SZ. Cribriform morular variant of papillary thyroid carcinoma: clinical and cytomorphological features on fine-needle aspiration. Acta Cytol. 2013;57(2):127–33.
Levy RA, Hui VW, Sood R, Fish S, Markowitz AJ, Wong RJ, et al. Cribriform-morular variant of papillary thyroid carcinoma: an indication to screen for occult FAP. Fam Cancer. 2014;13(4):547–51.
Jung CK, Choi YJ, Lee KY, Bae JS, Kim HJ, Yoon SK, et al. The cytological, clinical, and pathological features of the cribriform-morular variant of papillary thyroid carcinoma and mutation analysis of CTNNB1 and BRAF genes. Thyroid. 2009;19(8):905–13.
Kaya S, Gumus M, Gurbuz Y, Cabuk D, Acikgoz O, Temiz S, et al. The prognostic value of beta-catenin and LEF-1 expression in patients with operable gastric carcinoma. Am J Transl Res. 2016;8(2):1228–36.
Tandon B, Peterson L, Gao J, Nelson B, Ma S, Rosen S, et al. Nuclear overexpression of lymphoid-enhancer-binding factor 1 identifies chronic lymphocytic leukemia/small lymphocytic lymphoma in small B-cell lymphomas. Mod Pathol. 2011;24(11):1433–43.
Singhi AD, Lilo M, Hruban RH, Cressman KL, Fuhrer K, Seethala RR. Overexpression of lymphoid enhancer-binding factor 1 (LEF1) in solid-pseudopapillary neoplasms of the pancreas. Mod Pathol. 2014;27(10):1355–63.
Bilodeau EA, Acquafondata M, Barnes EL, Seethala RR. A comparative analysis of LEF-1 in odontogenic and salivary tumors. Hum Pathol. 2015;46(2):255–9.
Kermanshahi TR, Jayachandran P, Chang DT, Pai R. LEF-1 is frequently expressed in colorectal carcinoma and not in other gastrointestinal tract adenocarcinomas: an immunohistochemical survey of 602 gastrointestinal tract neoplasms. Appl Immunohistochem Mol Morphol. 2014;22(10):728–34.
Kurihara K, Shimizu S, Chong J, Hishima T, Funata N, Kashiwagi H, et al. Nuclear localization of immunoreactive beta-catenin is specific to familial adenomatous polyposis in papillary thyroid carcinoma. Jpn J Cancer Res. 2000;91(11):1100–2.
Hirokawa M, Maekawa M, Kuma S, Miyauchi A. Cribriform-morular variant of papillary thyroid carcinoma–cytological and immunocytochemical findings of 18 cases. Diagn Cytopathol. 2010;38(12):890–6.
Koo JS, Jung W, Hong SW. Cytologic characteristics and beta-catenin immunocytochemistry on smear slide of cribriform-morular variant of papillary thyroid carcinoma. Acta Cytol. 2011;55(1):13 – 8.
Kononen J, Bubendorf L, Kallioniemi A, Barlund M, Schraml P, Leighton S, et al. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med. 1998;4(7):844–7.
Garcia-Rostan G, Tallini G, Herrero A, D’Aquila TG, Carcangiu ML, Rimm DL. Frequent mutation and nuclear localization of beta-catenin in anaplastic thyroid carcinoma. Cancer Res. 1999;59(8):1811–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohindra, S., Sakr, H., Sturgis, C. et al. LEF-1 is a Sensitive Marker of Cribriform Morular Variant of Papillary Thyroid Carcinoma. Head and Neck Pathol 12, 455–462 (2018). https://doi.org/10.1007/s12105-017-0873-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-017-0873-3