Abstract
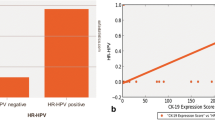
The aim of the current study was to investigate the presence of human papillomavirus (HPV) and evaluate its association with Ki-67 and cyclooxygenase-2 (COX-2) expressions in keratocystic odontogenic tumor (KCOT). Nineteen cases were included in the present study. Conventional PCR method and immunohistochemical analysis were performed for the detection of HPV-DNA and HPV-L1 capsid protein. Moreover, the expressions of Ki-67 and COX-2 proteins were analyzed immunohistochemically. HPV-DNA was detected in 36.8 % (7/19) of tumor samples, whilst HPV-L1 protein was identified in 68.4 % (13/19) of them. The Kappa coefficient statistical test showed a moderate agreement (κ 0.424) between PCR and IHC assays for HPV detection. Expression of HPV-DNA was positively correlated with Ki-67 and COX-2 expressions (p < 0.05), whereas HPV-L1 positive staining was positively correlated with COX-2 (p < 0.05) and highly associated with those of Ki-67 (p < 0.01). There was no significant correlation between the presence of HPV and the recurrence of the studied lesions. The results of the current study showed that active HPV infection was present in the odontogenic epithelium of KCOT, and it was associated with increased proliferation rate and COX-2 expression. These findings suggest that HPV may have a role in the pathogenesis and aggressiveness of KCOT. Based on these conclusions, we recommend further investigations of HPV vaccine or antiviral therapy and COX-2 inhibitors as nonsurgical options in the prevention and management of KCOT.

Similar content being viewed by others
References
Barnes L, Eveson JW, Reichart P, Sidransky D (2005) World Health Organization classification of tumours. Pathology and genetics of head and neck tumours. IARC, Lyon
Stoelinga PJ, Bronkhorst FB (1988) The incidence, multiple presentation and recurrence of aggressive cysts of the jaws. J Craniomaxillofac Surg 16:184–195
Lund VJ (1985) Odontogenic keratocyst of the maxilla: a case report. Br J Oral Maxillofac Surg 23:210–215
Madras J, Lapointe H (2008) Keratocystic odontogenic tumour: reclassification of the odontogenic keratocyst from cyst to tumour. J Can Dent Assoc 74:165–165h
Shear M, Speight PM (2007) Odontogenic keratocyst. In: Shear M, Speight PM (eds) Cysts of the Oral and Maxillofacial Regions, 4th edn. Blackwell Munksgaard, Oxford, pp 6–58
Scholzen T, Gerdes J (2000) The Ki-67 protein: from the known and the unknown. J Cell Physiol 182:311–322
Gurgel CA, Ramos EA, Azevedo RA, Sarmento VA, da Silva Carvalho AM, dos Santos JN (2008) Expression of Ki-67, p53 and p63 proteins in keratocyst odontogenic tumours: an immunohistochemistry study. J Mol Histol 39:311–316
Gadbail AR, Chaudhary M, Patil S, Gawande M (2009) Actual Proliferating Index and p53 protein expression as prognostic marker in odontogenic cysts. Oral Dis 15:490–498
de Oliveira MG, Lauxen Ida S, Chaves AC, Rados PV, Sant'Ana Filho M (2011) Odontogenic epithelium: immunolabeling of Ki-67, EGFR and survivin in pericoronal follicles, dentigerous cysts and keratocystic odontogenic tumors. Head Neck Pathol 5:1–7
Smith WL, DeWitt DL, Garavito RM (2000) Cyclooxygenases: structural, cellular, and molecular biology. Annu Rev Biochem 69:145–182
Marnett LJ, DuBois RN (2002) COX-2: a target for colon cancer prevention. Annu Rev Pharmacol Toxicol 42:55–80
Kurihara Y, Hatori M, Ando Y, Ito D, Toyoshima T, Tanaka M, Shintani S (2009) Inhibition of cyclooxygenase-2 suppresses the invasiveness of oral squamous cell carcinoma cell lines via down-regulation of matrix metalloproteinase-2 production and activation. Clin Exp Metastasis 26:425–432
Chang F, Syrjänen S, Kellokoski J, Syrjänen K (1991) Human papillomavirus (HPV) infections and their associations with oral disease. J Oral Pathol Med 20:305–317
Psyrri A, Boutati E, Karageorgopoulou S (2011) Human papillomavirus in head and neck cancers: biology, prognosis, hope of treatment, and vaccines. Anticancer Drugs 22:586–590
Kahn MA (1989) Ameloblastoma in young persons: a clinicopathologic analysis and etiologic investigation. Oral Surg Oral Med Oral Pathol 67:706–715
Kahn MA (1992) Demonstration of human papillomavirus DNA in a peripheral ameloblastoma by in situ hybridization. Hum Pathol 23:188–191
Namin AK, Azad TM, Eslami B, Sarkarat F, Shahrokhi M, Kashanian F (2003) A study of the relationship between ameloblastoma and human papilloma virus. J Oral Maxillofac Surg 61:467–470
Cox M, Eveson J, Scully C (1991) Human papillomavirus type 16 DNA in an odontogenic keratocyst. J Oral Pathol Med 20:143–145
González-Moles MA, Mosqueda-Taylor A, Delgado-Rodríguez M et al (2006) Analysis of p53 protein by PAb240, Ki-67 expression and human papillomavirus DNA detection in different types of odontogenic keratocyst. Anticancer Res 26:175–181
Kleter B, van Doorn LJ, ter Schegget J et al (1998) Novel short-fragment PCR assay for highly sensitive broad-spectrum detection of anogenital human papillomaviruses. Am J Pathol 153:1731–1739
Syrjänen S, Lodi G, von Bültzingslöwen I et al (2011) Human papillomaviruses in oral carcinoma and oral potentially malignant disorders: a systematic review. Oral Dis 17(Suppl 1):58–72
Miller CS, Johnstone BM (2001) Human papillomavirus as a risk factor for oral squamous cell carcinoma: a meta-analysis, 1982–1997. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 91:622–635
Montaldo C, Mastinu A, Quartuccio M et al (2007) Detection and genotyping of human papillomavirus DNA in samples from healthy Sardinian patients: a preliminary study. J Oral Pathol Med 36:482–487
SahebJamee M, Boorghani M, Ghaffari SR, AtarbashiMoghadam F, Keyhani A (2009) Human papillomavirus in saliva of patients with oral squamous cell carcinoma. Med Oral Patol Oral Cir Bucal 14:e525–528
Adelstein DJ, Ridge JA, Gillison ML et al (2009) Head and neck squamous cell cancer and the human papillomavirus: summary of a National Cancer Institute State of the Science Meeting, November 9–10, 2008, 31. Head Neck, Washington DC, pp 1393–1422
Balan R, Simion N, Giuşcă SE et al (2011) Immunohistochemical assessment of p16, COX-2 and EGFR in HPV-positive cervical squamous intraepithelial lesions. Rom J Morphol Embryol 52:1187–1194
Ozbun MA, Meyers C (1997) Characterization of late gene transcripts expressed during vegetative replication of human papillomavirus type 31b. J Virol 71:5161–5172
Correnti M, Rossi M, Avila M, Perrone M, Rivera H (2010) Human papillomavirus in ameloblastoma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 110:e20–24
van Heerden WF, van Rensburg EJ, Raubenheimer EJ, Venter EH (1993) Detection of human papillomavirus DNA in an ameloblastoma using the in situ hybridization technique. J Oral Pathol Med 22:109–112
Sand L, Jalouli J, Larsson PA, Magnusson B, Hirsch JM (2000) Presence of human papilloma viruses in intraosseous ameloblastoma. J Oral Maxillofac Surg 58:1129–1134
Migaldi M, Pecorari M, Rossi G et al (2005) Does HPV play a role in the etiopathogenesis of ameloblastoma? An immunohistochemical, in situ hybridization and polymerase chain reaction study of 18 cases using laser capture microdissection. Mod Pathol 18:283–289
Dalgleish AG (1991) Viruses and cancer. Br Med Bull 47:21–46
Stubenrauch F, Laimins LA (1999) Human papillomavirus life cycle: active and latent phases. Semin Cancer Biol 9:379–386
Garner-Hamrick PA, Fostel JM, Chien WM et al (2004) Global effects of human papillomavirus type 18 E6/E7 in an organotypic keratinocyte culture system. J Virol 78:9041–9050
Samir R, Asplund A, Tot T, Pekar G, Hellberg D (2011) High-risk HPV infection and CIN grade correlates to the expression of c-myc, CD4+, FHIT, E-cadherin, Ki-67, and p16INK4a. J Low Genit Tract Dis 15:280–286
de Vicente JC, Torre-Iturraspe A, Gutiérrez AM, Lequerica-Fernández P (2010) Immunohistochemical comparative study of the odontogenic keratocysts and other odontogenic lesions. Med Oral Patol Oral Cir Bucal 15:e709–715
González-Alva P, Tanaka A, Oku Y, Yoshizawa D, Itoh S, Sakashita H, Ide F, Tajima Y, Kusama K (2008) Keratocystic odontogenic tumor: a retrospective study of 183 cases. J Oral Sci 50:205–212
Kim SH, Oh JM, No JH, Bang YJ, Juhnn YS, Song YS (2009) Involvement of NF-kappaB and AP-1 in COX-2 upregulation by human papillomavirus 16 E5 oncoprotein. Carcinogenesis 30:753–757
Wu R, Coniglio SJ, Chan A, Symons MH, Steinberg BM (2007) Up-regulation of Rac1 by epidermal growth factor mediates COX-2 expression in recurrent respiratory papillomas. Mol Med 13:143–150
Subbaramaiah K, Dannenberg AJ (2007) Cyclooxygenase-2 transcription is regulated by human papillomavirus 16 E6 and E7 oncoproteins: evidence of a corepressor/coactivator exchange. Cancer Res 67:3976–3985
Zhuang ZH, Tsao SW, Deng W et al (2008) Early upregulation of cyclooxygenase-2 in human papillomavirus type 16 and telomerase-induced immortalization of human esophageal epithelial cells. J Gastroenterol Hepatol 23:1613–1620
Prescott SM, Fitzpatrick FA (2000) Cyclooxygenase-2 and carcinogenesis. Biochim Biophys Acta 1470:M69–78
Mohan S, Epstein JB (2003) Carcinogenesis and cyclooxygenase: the potential role of COX-2 inhibition in upper aerodigestive tract cancer. Oral Oncol 39:537–546
Mestre JR, Subbaramaiah K, Sacks PG et al (1997) Retinoids suppress epidermal growth factor-induced transcription of cyclooxygenase-2 in human oral squamous carcinoma cells. Cancer Res 57:2890–2895
Mendes RA, Carvalho JF, Waal I (2009) An overview on the expression of cyclooxygenase-2 in tumors of the head and neck. Oral Oncol 45:e124–128
Zhang Y, McCluskey K, Fujii K, Wahl LM (1998) Differential regulation of monocyte matrix metalloproteinase and TIMP-1 production by TNF-α, granulocyte-macrophage CSF and IL-1β through prostaglandin-dependent and -independent mechanisms. J Immunol 161:3071–3076
Callejas NA, Casado M, Diaz-Guerra MJ, Bosca L, Martin-Sanz P (2001) Expression of cyclooxygenase-2 promotes the release of matrix metalloproteinase-2 and −9 in fetal rat hepatocytes. Hepatology 33:860–867
Ottino P, Bazan HE (2001) Corneal stimulation of MMP-1, −9 and uPA by platelet-activating factor is mediated by cyclooxygenase-2 metabolites. Curr Eye Res 23:77–85
Gomes CC, Duarte AP, Diniz MG, Gomez RS (2010) Review article: Current concepts of ameloblastoma pathogenesis. J Oral Pathol Med 39:585–591
Harris M (1978) Odontogenic cyst growth and prostaglandin-induced bone resorption. Ann R Coll Surg Engl 60:85–91
Fracon RN, Teófilo JM, Satin RB, Lamano T (2008) Prostaglandins and bone: potential risks and benefits related to the use of nonsteroidal anti-inflammatory drugs in clinical dentistry. J Oral Sci 50:247–252
Gao L, Li TJ (2005) In vitro study on bone resorption of odontogenic cysts and ameloblastomas. Zhonghua Kou Qiang Yi Xue Za Zhi 40:233–236
Mendes RA, Carvalho JF, van der Waal I (2011) Potential relevance of cyclooxygenase-2 expression in keratocystic odontogenic tumours - an immunohistochemical study. J Oral Pathol Med 40:497–503
Tsai CH, Huang FM, Yang LC, Chou MY, Chang YC (2002) Immunohistochemical localization of cyclooxygenase-2 in radicular cysts. Int Endod J 35:854–858
Johnson RL, Rothman AL, Xie J et al (1996) Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science 272:1668–1671
Ren C, Amm HM, DeVilliers P et al (2012) Targeting the sonic hedgehog pathway in keratocystic odontogenic tumor. J Biol Chem 287:27117–27125
Bhargava D, Deshpande A, Pogrel MA (2012) Keratocystic odontogenic tumour (KCOT)–a cyst to a tumour. Oral Maxillofac Surg 16:163–170
Xuan YH, Li GL, Jiang HY, Lin ZH (2009) Relationship between hedgehog signaling pathway molecules and HPV16 infection in uterine cervical cancers. Zhonghua Bing Li Xue Za Zhi 38:178–182
Samarzija I, Beard P (2012) Hedgehog pathway regulators influence cervical cancer cell proliferation, survival and migration. Biochem Biophys Res Commun 425:64–69
Yagyuu T, Kirita T, Sasahira T, Moriwaka Y, Yamamoto K, Kuniyasu H (2008) Recurrence of keratocystic odontogenic tumor: clinicopathological features and immunohistochemical study of the Hedgehog signaling pathway. Pathobiology 75:171–176
Acknowledgments
We would like to thank the Noble Prize winner, Prof. Harald zur Hausen for supplying us with the HPV positive PCR control. Also we thank Prof. Wei Min Chen, Prof. Xue Jin Tao, As. Prof. Le Nan Shao, As. Prof. Xin Yu Ji, and Dr. Guo Xiong Tang for their help in clinical samples collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alsaegh, M.A., Miyashita, H. & Zhu, S.R. Expression of Human Papillomavirus is Correlated with Ki-67 and COX-2 Expressions in Keratocystic Odontogenic Tumor. Pathol. Oncol. Res. 21, 65–71 (2015). https://doi.org/10.1007/s12253-014-9789-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9789-3