Summary
We test the hypothesis that the Thesium (Santalaceae) collection previously known as Thesium sp. D.A.Zhigila & A.M.Muasya 1006 is new to science. We formally describe and name it T. schmiedeliae. The new species is an erect to suberect shrublet, c. 20 cm tall, glaucous in overall appearance, with stems terete in transverse section, and leaves bracts, and bracteoles succulent. Molecular and morphological data support the placement of T. schmiedeliae in subgenus Frisea section Annulata. The new species is endemic to the Knersvlakte Nature Reserve and occurs in the edaphically unique quartz islands. Thesium schmiedeliae is preliminarily assessed for conservation purposes as Vulnerable as it is threatened by drought, severe fragmentation of its quartz-field habitat, small population size, and a small area of occupancy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Angiosperm Phylogeny Group IV (2016) includes c. 340 species in Thesium L. (Santalaceae), they are mainly distributed in the old world (including Africa, Asia and Europe), the new world and North America (POWO 2022). Although Thesium is a common and abundant component of the vegetation of the Fynbos Biome which accommodates most of the southern African species, it often goes unnoticed because of its drab morphology. Thesium species are hemiparasitic, annual or perennial herbs, shrublets, shrubs, and small trees, with stems that are sparse to densely branched. The leaves are polymorphic but typically reduced or sparse and adpressed to the stem. Most species in the genus have bisexual flowers and are consistently white except for species in T. subgenus Hagnothesium (A.DC.) Zhigila, Verboom & Muasya whose flowers are unisexual, greenish, and minute (Hill 1925; Zhigila et al. 2019a).
Southern Africa is recognised as a centre of diversity for genera of the family Santalaceae, particularly Thesium (Hill 1925; Moore et al. 2010). In the last decade, Thesium in South Africa has received significant taxonomic attention from several authors (including Nickrent & García 2015; García et al. 2018; Visser et al. 2018; Zhigila et al. 2019a, 2020; Lombard et al. 2021). This attention was triggered by the prioritisation of Thesium as a genus in urgent need of taxonomic revision (Victor et al. 2015). In response to this taxonomic urgency, many new species and new records of genera and species of Thesium have been discovered in South Africa, particularly from the Greater Cape Floristic Region (GCFR) (García et al. 2018; Lombard et al. 2021; Zhigila et al. 2019b; Zhigila & Muasya 2022) and the subtropical northeastern region (e.g. Lombard et al. 2019; Lombard & Le Roux 2023). A number of the newly described species are edaphic specialists with limited geographic distributions, for example T. muasyae Zhigila (Zhigila & Muasya 2022) and the majority are placed phylogenetically within subgenus Frisea (Rchb.) Hendrych (Zhigila et al. 2020). Within subgenus Frisea, the taxonomy of the Annulata clade (sensu Hill 1915) is problematic, with numerous, morphology-based, species complexes. Further, new location records of allied genera have enhanced our understanding of the biogeographic patterns of Thesium. For instance, the segregate genus Lacomucinaea Nickrent & M.A.García has been recorded from the Namaqualand region (Nickrent & García 2015). The discovery of T. ovatifolium N.Lombard & M.M.le Roux and T. hispidifructum N.Lombard & M.M.le Roux in northeastern South Africa (Lombard et al. 2019; Lombard & Le Roux 2023) reveal that more species will undoubtedly be described as research on the genus continues.
In 2017, Ute Schmiedel photographed a Thesium taxon at Knersvlakte Nature Reserve, Namaqualand, whose vegetative and reproductive features were most similar to T. elatius DC., T. ecklonianum Sond. and T. urceolatum A.W.Hill. Subsequent field visits to Knersvlakte and its environs revealed four populations of this taxon. Further assessments of major herbaria holding significant Thesium specimens (BOL, K, NBG, PRE) (acronyms follow Thiers 2023, continuously updated) and iNaturalist (2022), revealed an additional specimen of the taxon collected by N. Helme 7588 (NBG) which had been identified and filed under T. elatius. Based on data from both herbarium and field investigations as well as molecular data, we determined this taxon to be an evolutionarily distinct species. Thus, we used ecological, morphological, and molecular data within a phylogenetic framework, together with multivariate species delimitation techniques, to confirm the new species, and we have called it T. schmiedeliae Zhigila & Muasya.
Materials and Methods
Morphological Observations
For morphological investigations we studied our field collections, herbarium specimens at BOL, NBG (containing SAM and STE vouchers) and PRE, and online voucher materials (JSTOR 2022; K; POWO 2022) of related species. We used a hand lens (×10), or a binocular stereomicroscope fitted with an eyepiece graticule and a Nikon DS-5M camera for micromorphological characterisation. The holotype of Thesium schmiedeliae was deposited in BOL, and duplicates were given to NBG and PRE. Morphological terminology and the species description format follow Hill (1925), as amended by Zhigila et al. (2019a).
Phylogenetic Analyses
In a global sampling scheme (Zhigila et al. 2020), we used nuclear ribosomal ITS and three chloroplast DNA regions (matK, rbcL and trnL-trnF) to infer the phylogenetic placement of the new species. Sequences from previous studies (Nickrent & García 2015; Zhigila et al. 2020; Zhigila & Muasya 2022) were retrieved from GenBank and analysed along with six sequences from the newly acquired samples of Thesium schmiedeliae. These newly generated DNA sequences were deposited in GenBank and assigned accession numbers: nrITS OM746332 – OM746333; rbcL OM818243 and OM818244; trnL-trnF OM857948 and OM857949. The procedures outlined in Zhigila et al. (2020) and Zhigila & Muasya (2022) were used for sequence assembly and alignment. Before combining the four data sets, single gene trees were created for each, to determine if there are any well-supported incongruences (bootstrap value of > 70%) between the nrITS and chloroplast topologies. Bootstrap support (BS) values from 71 – 89% were considered weak, while BS values of > 90% were considered strong. Phylogenetic support was considered weak if the posterior probability was ≤ 0 .90 and strong if > 0.90, following Hillis & Bull (1993).
The combined dataset was analysed using maximum likelihood (ML) and Bayesian inference methods. The ML analyses were conducted in RAxML v.8, applying 1,000 bootstrap replicates (Stamatakis 2014). A partitioned Bayesian analysis was conducted in MrBayes 3.2.6 x64 (Ronquist & Huelsenbeck 2003) as implemented on the CIPRES Science Gateway Portal v.3.3 (Miller et al. 2010). Parameter configuration and convergence estimation followed Zhigila et al. (2020). The best-fitting nucleotide substitution model under the Akaike information criterion (AIC), for each of the single partitions, was calculated using jModelTest 2 v.3.7 (Darriba et al. 2012). GTR+ Γ was found to be the most appropriate model for matK, GTR+I for rbcL and trnL-trnF, and GTR+I+ Γ for ITS.
Nomenclature and Conservation Assessments
The International Plant Names Index (IPNI 2023, continuously updated) was used for taxonomic authorities, and the nomenclature follows Turland et al. (2018). The Conservation assessment was conducted using Bachman et al. (2011) for geospatial analysis, following the International Union for Conservation of Nature (IUCN 2022) standard and categories.
Results
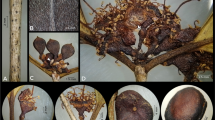
The newly collected specimens from the Knersvlakte Nature Reserve were identified as a new species of Thesium subgenus Frisea section Annulata, based on their alternate leaves, leaves adpressed to the stem, tufts of apical trichomes on the perianth lobes of the flowers, and 5-merous, fruits which are nutlets, and persistent perianth segments (Figs 1 and 2). This taxon was recognised as new to science because it differed from any known Thesium species. It is erect to suberect in growth habit, with terete, glaucous stems, succulent leaves, bracts and bracteoles, an elongated spike of flowers, conspicuous bracts with scars from fallen flowers, old bracts and bracteoles maroon, the presence of elaiosomes on fruits, and veins on the fruits inconspicuous when young but becoming prominent on maturity (Fig. 2; Table 1).
The Bayesian and ML analyses of the combined data set of three chloroplast and the nrITS regions generated congruent topologies. The 50% majority-rule consensus tree obtained from the Bayesian analysis with ML bootstrap (BS) and Bayesian posterior probability (PP) is presented in Fig. 3. As in previous studies, the monophyly of Thesium was strongly supported and T. subgenus Frisea was also reconfirmed as monophyletic (BS = 100%, PP = 1). The two samples of the new species T. schmiedeliae grouped together (BS =100%, PP = 1), forming a grade comprising T. elatius and T. ecklonianum and a clade with species restricted to the GCFR. Of the sequences of 4572 aligned base pairs, 11 nucleotide variation sites were detected that distinguish T. elatius and 14 nucleotide variations distinguished T. ecklonianum.
Taxonomic Treatment
Thesium schmiedeliae Zhigila & Muasya sp. nov. Type: South Africa, Western Cape Province, Moedverloren farm, Knersvlakte, Namaqualand, 13 Sept. 2021, 31.46508S, 18.44977E, alt. 100 m above sea level, D. A. Zhigila & A. M. Muasya 1006 (holotype BOL!; isotypes NBG!, PRE!).
http://www.ipni.org/urn:lsid:ipni.org:names:77323181-1
Erect to suberect perennial shrublet to about 20 cm tall, arising from a woody rootstock, glabrous and glaucous in overall appearance. Stems woody, 2 – 3 mm in diam., terete in transverse section. Branches 4 – 5 in number, mainly from the base, terete, angled from > 45° to ≤ 90°, branching pattern virgate. Leaves terete to triangular, succulent, sessile to subsessile on the stem or branchlets, lanceolate or oblanceolate, 1.5 – 3 × 0.5 – 1.5 cm, basally decurrent, midrib inconspicuous, not keeled but slightly recurved or straight, margins not distinct or entire, apex apiculate. Inflorescences are racemose or elongated axillary spikes in leaf and bract axils. Bracts leaf-like, succulent, slightly adnate to the base of peduncle in racemose inflorescences, broadly lanceolate, 1 – 2 × 0.3 – 0.5 cm, margin entire, apex acute to acuminate, green to reddish-brown or maroon when dry. Bracteoles bract-like but smaller, adpressed to the pedicel in racemose inflorescences and shorter than flower length, with scars of fallen flowers. Flowers hypocrateriform, on long peduncles, 5-merous, 1.5 – 5 × 1 – 2 mm; perianth lobe segments lanceolate to triangular, external glands inconspicuously elongated, alternating with perianth lobe segments, 2 – 2.5 × 1 – 1.2 mm; lobe apex uncinate, obtuse, slightly recurved; apical tuft of trichomes present; lobe margins entire; lobe internal colour white, external colour greyish to golden green; hypanthium distinct, to about 0.3 mm long, length shorter than perianth lobe tube and narrower. Stamens equal flower merosity; 0.2 – 0.3 mm long; staminal filaments exserted, inserted at about the middle of the hypanthium tube, clearly above stigmas, not attached to the perianth lobe walls; with downwardly-directed, golden brown, basal trichomes. Style together with stigma 0.4 – 0.5 mm long; placental column twisted. Fruits nutlets, globose to subglobose, 2 – 5 × 1.5 – 3.5 mm, greyish to golden green, glabrous; without conspicuous longitudinal ribs when young but ribs become prominent when dry; reticulate veins inconspicuous; pedicels enlarging into elaiosomes; persistent perianth segments shorter than to equal to the fruit. Seeds similar in shape to fruits.
recognition. Thesium schmiedeliae is similar to T. elatius in stem architecture, leaves and succulent bracts, and having tufts of trichomes on the perianth lobes. However, T. schmiedeliae has an erect to suberect growth habit, virgate, not twiggy, branching, conspicuous bracts, racemose or elongated axillary spikes of flowers in leaf and bract axils, bracteoles shorter than the flowers, the presence of elaiosomes on fruits and is restricted to quartz fields. T. elatius, by comparison, has a prostrate growth habit, an intricate, twiggy branching pattern, inconspicuous bracts, inflorescences are a spike to globose, bracteoles are equal or longer than flowers, it lacks elaiosomes on its fruits (Fig. 4) and occurs on sandy and coastal flats (Table 1). Thesium schmiedeliae is also similar to T. ecklonianum and T. urceolatum in having a glaucous overall appearance, lacking twiggy branching, having conspicuous bracts and having elaiosomes on its fruits. It differs from both in its erect to suberect growth habit, < 20 cm tall, virgate branching pattern, having all leaves, bracts and bracteoles succulent, and in being restricted to quartz fields. By comparison, T. ecklonianum and T. urceolatum have a prostrate growth habit, decumbent branching patterns, < 50 cm long, lower leaves succulent, upper leaves lanceolate and not succulent, bracts and bracteoles not succulent (Fig. 4), and T. ecklonianum occurs in coastal sand fynbos (Table 1). As the four taxa differ in morphology, are allopatric and occupy both discrete edaphic niches and different positions on the phylogeny, we consider each to be on unique evolutionary trajectories (De Queiroz 2007) and as distinct species.
Type specimens of species Thesium schmiedeliae and closely similar species. A T. elatius; B T. schmiedeliae; C T. ecklonianum http://specimens.kew.org/herbarium/K000431730.
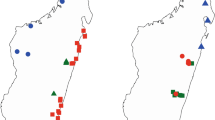
distribution. The new species is only known from the vicinity of the type locality, Knersvlakte Nature Reserve, over a range of < 10 km2 in Vanrhynsdorp Division, Western Cape, South Africa (Map 1).
specimens examined. south africa. Western Cape Province: Moedverloren farm, Knersvlakte, Namaqualand, 13 Sept. 2021, 31.46484S, 18.45069E, elev. 120 m above sea level, D. A. Zhigila & A. M. Muasya 1023 (BOL); Moedverloren farm, Knersvlakte, Namaqualand, 14 Sept. 2021, 31.47324S, 18.43673E, elev. 150 m asl, D. A. Zhigila & A. M. Muasya 1085 (BOL); Biodiversity Observatory S28, Moedverloren farm, Knersvlakte, Namaqualand, 14 Sept. 2021, 31.46753S, 18.44761E, elev. 150 m asl, D. A. Zhigila & A. M. Muasya 1088 (BOL); Dr Ute Schmiedel’s observatory site, Moedverloren farm, Knersvlakte, Namaqualand, 14 Sept. 2021, 31.34099S, 18.4751E, elev. 120 m asl, D. A. Zhigila & A. M. Muasya 1132 (BOL).
habitat. Thesium schmiedeliae is endemic to the quartz fields of the Knersvlakte, at elevations between 100 and 200 m asl. The species occurs singly on slightly acidic soils (pH 4.5 – 6.5) with low to moderate salinity (up to 2500 mS/cm).
conservation status. Vulnerable V D2 (IUCN 2022). Four populations of Thesium schmiedeliae have been observed at Moedverloren farm, Knersvlakte Nature Reserve. This area is a Nature Reserve with no immediate threat at present. However, the extent of occurrence of T. schmiedeliae is relatively small, with Extent of Occurrence = 0.5 km2, and it is only known from a few collections in four localities, with Area of Occupancy = 10.5 km2. Due to the narrow distribution of the species, its habitat specialisation to a very harsh environment, and the sparse occurrence per site, the species could be threatened by the frequency and severity of droughts that are increasingly experienced in the area.
phenology. Flowering between August and November; fruiting from September to January.
etymology. The name Thesium schmiedeliae honours Dr Ute Schmiedel, who called the attention of the authors to this species. Further, she has contributed extensively to the understanding of the ecology of the southern African quartz fields which is the type locality of the species.
notes.Thesium schmiedeliae is here described from the Knersvlakte Nature Reserve, Vanrhynsdorp Division, Western Cape, South Africa. It is an erect perennial shrublet that grows on quartz patches. These quartz patches are unusually arid habitats with extremely low pH levels (Schmiedel & Jürgens 1999; Eibes et al. 2022). The southern African quartz habitats have c. 155 local to regional endemic vascular plants, of which 67% belong to the Aizoaceae (Schmiedel 2004) and are characterised by perennial, leaf-succulent, dwarf shrubs (Schmiedel 2004). The dwarf growth habit and leaf succulence observed in T. schmiedeliae are hypothesised to be a convergent evolutionary response to the specialised habitat conditions of the quartz fields (Schmiedel & Jürgens 1999). The discovery of this new species suggests that there may be further undescribed diversity in the Santalaceae in South Africa that requires further field surveys.
In the context of molecular phylogenetic relationships, Thesium schmiedeliae is placed in T. subgenus Frisea (Zhigila et al. 2020), section Annulata (Hill 1925), and ‘leafy clade’ sensu Moore et al. (2010). This phylogenetic position is supported by several morphological apomorphies which unite species in this clade. For example, species in subgenus Frisea are united by the presence of dense apical trichomes on the perianth segments and the presence of downwardly facing tufts of golden yellow trichomes on perianth lobes behind the anthers (Fig. 2; Zhigila et al. 2020).
References
Angiosperm Phylogeny Group IV (2016). An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot. J. Linn. Soc. 181: 1 – 20. https://doi.org/10.1111/boj.12385.
Bachman, S., Moat, J., Hill, A. W., de la Torre, J. & Scott, B. (2011). Supporting Red List threat assessments with GeoCAT: geospatial conservation assessment tool. In: V. Smith & L. Penev (eds), e-Infrastructures for data publishing in biodiversity science. ZooKeys 150: 117 – 126. https://doi.org/10.3897/zookeys.150.2109.
Darriba D., Taboada G. L., Doallo, R. & Posada, D. (2012). jModelTest 2: more models, new heuristics and parallel computing. Nature Methods 9 (8): 772. https://doi.org/10.1038/nmeth.2109.
De Queiroz, K. (2007). Species concepts and species delimitation. Syst. Biol. 56 (6): 879 – 886. https://doi.org/10.1080/10635150701701083.
Eibes, P., Schaffrath, F., Oldeland, J., Thormählen, W., Schmiedel, U. & Irl, S. (2022). Testing the concept of edaphism for the quartz island flora of the Knersvlakte, South Africa. S. African J. Bot. 151: 555 – 564. https://doi.org/10.1016/j.sajb.2022.10.015.
García, M. A., Nickrent, D. L. & Mucina, L. (2018). Thesium nautimontanum, a new species of Thesiaceae (Santalales) from South Africa. PhytoKeys 109: 41 – 51. https://doi.org/10.3897/phytokeys.109.28607.
Hill, A. W. (1915). The genus Thesium in South Africa, with a key and descriptions of new species. Bull. Misc. Inform., Kew 1915 (1): 1 – 43. https://doi.org/10.2307/4115447.
Hill, A. W. (1925). Order CXX. Santalaceae. In: W. T. Thiselton-Dyer (ed), Flora Capensis 5: 135 – 212. Reeve, London. https://doi.org/10.5962/bhl.title.821.
Hillis, D. M. & Bull, J. J. (1993). An Empirical Test of Bootstrapping as a Method for Assessing Confidence in Phylogenetic Analysis. Syst. Biol. 42: 182 – 192. https://doi.org/10.1093/sysbio/42.2.182.
iNaturalist (2022). iNaturalist Home Page. Available from https://www.inaturalist.org.
IPNI (2023, continuously updated). International Plant Names Index. Published on the Internet http://www.ipni.org, The Royal Botanic Gardens, Kew, Harvard University Herbaria & Libraries and Australian National Herbarium. [Accessed 2 Feb. 2023].
IUCN (2022). Guidelines for using the IUCN Red List categories and criteria. Version 15.1. Standards and Petitions Subcommittee. http://www.iucnredlist.org/documents/RedListGuidelines.pdf [Accessed Nov. 2022].
JSTOR Global Plants (2022). Global Plants on JSTOR. https://plants.jstor.org/compilation/Thesium.[Accessed 7 Oct. 2022].
Lombard, N. & Le Roux, M. M. (2023). Thesium hispidifructum (Santalaceae), a New Hispidulous Species from Limpopo, South Africa and Notes on Enigmatic T. celatum. Taxonomy 3: 95 – 108. https://doi.org/10.3390/taxonomy3010009
Lombard, N. & Le Roux, M. M. & van Wyk, B. (2019). Thesium ovatifolium (Santalaceae), a new species with ovate leaves from KwaZulu-Natal, South Africa. Phytotaxa 405 (5). https://doi.org/10.11646/phytotaxa.405.5.5
Lombard, N., Le Roux, M. M. & van Wyk, B. (2021). A taxonomic revision of the Thesium scirpioides species complex (Subgenus Frisea, Santalaceae) near endemic to South Africa. S. African J. Bot. 138: 193 – 208. https://doi.org/10.1016/j.sajb.2020.12.005
Miller, M. A., Pfeiffer, W. & Schwartz, T. (2010). Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop (GCE), 14 Nov. 2010, New Orleans, LA. Pp 1 – 8. https://doi.org/10.1109/GCE.2010.5676129.
Moore, T. E., Verboom, G. A. & Forest, F. (2010). Phylogenetics & biogeography of the parasitic genus Thesium L. (Santalaceae), with an emphasis on the Cape of South Africa. Bot. J. Linn. Soc. 162: 435 – 452. https://doi.org/10.1111/j.1095-8339.2010.01032.x.
Nickrent, D. L. & García, M. A. (2015). Lacomucinaea, a new monotypic genus in Thesiaceae (Santalales). Phytotaxa 224 (2): 173 – 184. https://doi.org/10.11646/phytotaxa.224.2.4.
POWO (2022). Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. http://www.plantsoftheworldonline.org/. [Accessed 17 Nov. 2022]
Ronquist, F. & Huelsenbeck, J. P. (2003). MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19: 1572 – 1574. https://doi.org/10.1093/bioinformatics/btg180.
Schmiedel, U. (2004). The phytogeography of the obligate quartz field flora of southern Africa. Schumannia 4 (Biodiversity & Ecology 2): 181 – 205. University of Hamburg.
Schmiedel, U. & Jürgens, N. (1999). Community structure on unusual habitat islands: quartz-fields in the Succulent Karoo, South Africa. Pl. Ecol. 142: 57 – 69. https://doi.org/10.1023/A:1009818210799.
Stamatakis, A. (2014). RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30 (9): 1312 – 1313. https://doi.org/10.1093/bioinformatics/btu033.
Thiers, B. (2023, continuously updated). Index Herbariorum: a Global Directory of Public Herbaria and Associated Staff. New York Botanical Gardens Virtual Herbarium. Available at: <http://sweetgum.nybg.org/ih>. [Accessed 15 Jan. 2023].
Turland, N. J., Wiersema, J. H., Barrie, F. R., Greuter, W., Hawksworth, D. L., Herendeen, P. S., Knapp, S., Kusber, W.-H., Li, D.-Z., Marhold, K., May, T. W., McNeill, J., Monro, A. M., Prado, J., Price, M. J. & Smith, G. F. (eds) (2018). International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. Regnum Veg. 159. Koeltz Botanical Books, Glashütten. https://doi.org/10.12705/Code.2018.
Victor, J. E., Smith, G. F. & Van Wyk, A. E. (2015). Strategy for plant taxonomic research in South Africa 2015–2020. SANBI Biodiversity Series 26. South African National Biodiversity Institute, Pretoria. ISBN: 978-1-928224-09-9.
Visser, N., le Roux, M. M. & van Wyk, B. E. (2018). A taxonomic revision of the Thesium goetzeanum species complex (Santalaceae) from Lesotho, South Africa and Swaziland. S. African J. Bot. 119: 45 – 62. https://doi.org/10.1016/j.sajb.2018.08.005.
Zhigila, D. A. & Muasya, A. M. (2022). Thesium muasyae (Santalaceae), a new species from the limestone fynbos of the Overberg, South Africa. PhytoKeys 201: 1 – 14. https://doi.org/10.3897/phytokeys.201.80774.
Zhigila, D. A., Verboom, G. A. & Muasya, A. M. (2020). An infrageneric classification of Thesium (Santalaceae) based on molecular phylogenetic data. Taxon 69 (1): 100 – 123. https://doi.org/10.1002/tax.12202.
Zhigila, D. A., Verboom, G. A., Stirton, C. H. & Muasya, A. M. (2019a). A taxonomic revision of Thesium section Hagnothesium (Santalaceae) and description of a new species, T. quartzicolum. S. African J. Bot. 124: 280 – 303. https://doi.org/10.1016/j.sajb.2019.05.016.
Zhigila, D. A., Verboom, G. A., Stirton, C. H., Smith, H. J. & Muasya, A. M. (2019b). Six new Thesium (Santalaceae) species endemic to the Greater Cape Floristic Region, South Africa, and one new name. Phytotaxa 423 (4): 215 – 237. https://doi.org/10.11646/phytotaxa.423.4.1.
Acknowledgements
We thank the Smuts Memorial Botanical Fellowship at the Department of Biological Sciences, University of Cape Town, South Africa for providing funding to DAZ for this project. Additional financial support was received from the National Research Foundation (Foundational Biodiversity Information Programme, Grant 136337 to AMM). We acknowledge the support by the International Association of Plant Taxonomists with a grant to fund the molecular lab work. We appreciate the Western Cape Nature Conservation Board for permits to collect herbarium specimens (permit number CN35-28-17379 – DAZ). We thank Pia M. Eibes for the line drawings. We are also grateful to the curators of BOL, K, NBG and PRE for providing access to herbarium materials.
Funding
Open access funding provided by University of Cape Town.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhigila, D.A., Schmiedel, U. & Muasya, A.M. Thesium schmiedeliae (Santalaceae), a new species from Knersvlakte quartz fields, South Africa. Kew Bull 78, 509–518 (2023). https://doi.org/10.1007/s12225-023-10114-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12225-023-10114-y