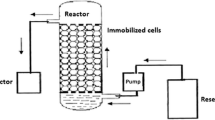
Abstract
The accumulation of chitin waste from the seafood industry is a serious environmental problem. However, this residue can be degraded by chitinases and its subproducts, such as chitosan, economically exploited. In this study, a chitinase producer bacteria, identified as Paenibacillus illinoisensis, was isolated from the Brazilian coastal city of Terra de Areia – Rio Grande Do Sul (RS) and was immobilized within alginate beads to evaluate its chitinase production. The alginate beads containing cells presented an average size of 4 mm, 99% of immobilization efficiency and increased the enzymatic activity in 40.71% compared to the free cells. The biomass during enzymatic production increased 62.01% and the total cells leaked from the alginate beads corresponded to 6.46% after 96 h. Immobilized cells were reused in a sequential batch system and remained stable for production for up to four 96-h cycles, decreasing only 21.04% of the initial activity at the end of the fourth cycle. Therefore, the methodology used for cell immobilization resulted in adequate beads to maintain cell viability during the enzymatic production, increasing enzymatic activity, showing low cell leakage from the support and appropriate recyclable capacity.






Similar content being viewed by others
References
Berlowska J, Kregiel D, Ambroziak W (2013) Physiological tests for yeast brewery cells immobilized on modified chamotte carrier. Anton Leeuw Int J G 104:703–714. https://doi.org/10.1007/s10482-013-9978-1
Cao L (2005) Introduction: Immobilized enzymes: past, present and prospects. 1st ed. In: Cao L (ed) Carrier-bound immobilized enzymes. Wiley-VCH, Weinheim, pp 1–52
Chandrasekaran R, Revathi K, Nisha S, Kirubakaran SA, Sathish-Narayanan S, Senthil-Nathan S (2012) Physiological effect of chitinase purified from Bacillus subtilis against the tobacco cutworm Spodoptera litura Fab. Pestic Biochem Physiol 104(1):65–71. https://doi.org/10.1016/j.pestbp.2012.07.002
Doan CT, Tran TN, Wang SL (2021) Production of thermophilic chitinase by Paenibacillus sp. TKU052 by bioprocessing of chitinous fishery wastes and its application in N-acetyl-D-glucosamine production. Polymers (Basel) 13(18):3048. https://doi.org/10.3390/polym13183048
Eş I, Ribeiro MC, Santos Júnior SR, Khaneghah AM, Rodriguez AG, Amaral AC (2016) Production of cyclodextrin glycosyltransferase by immobilized Bacillus sp. on chitosan matrix. Bioprocess Biosyst Eng 39:1487–1500. https://doi.org/10.1007/s00449-016-1625-6
Eş I, Vieira JDG, Amaral AC (2015) Principles, techniques, and applications of biocatalyst immobilization for industrial application. Appl Microbiol Biotechnol 99:2065–2082. https://doi.org/10.1007/s00253-015-6390-y
Gabardo S, Pereira GF, Rech R, Ayub MAS (2015) The modelling of ethanol production by Kluyveromyces marxianus using whey as substrate in continuous A-Stat bioreactors. J Ind Microbiol Biotechnol 42:1243–1253. https://doi.org/10.1007/s10295-015-1661-2
Gao H, Khera E, Lee JK, Wen F (2016) Immobilization of multi-biocatalysts in alginate beads for cofactor regeneration and improved reusability. J vis Exp 110:53994. https://doi.org/10.3791/53944
Gohel V, Singh A, Vimal M, Ashwini P, Chatpar HS (2006) Bioprospecting and antifungal potential of chitinolytic microorganisms. AJB 5(2):54–72
Halder SK, Maity C, Jana A, Ghosh K, das A PT, Mohapatra PKD, Pati BR, Mondal KC (2014) Chitinases biosynthesis by immobilized Aeromonas hydrophila SBK1 by prawn shells valorisation and application of enzyme cocktail for fungal protoplast preparation. J Biosci Bioeng 117(2):170–177. https://doi.org/10.1016/j.jbiosc.2013.07.011
Hamid R, Khan MA, Ahmad M, Ahmad MM, Abdin MZ, Musarrat J, Javed S (2013) Chitinases: an update. J Pharm Bioallied Sci 5:21–40. https://doi.org/10.4103/0975-7406.106559
Jahromi ST, Barzkar N (2018) Marine bacterial chitinase as sources of energy, eco-friendly agent, and industrial biocatalyst. Int J Biol Macromol 120(B):2147–2154. https://doi.org/10.1016/j.ijbiomac.2018.09.083
Jung WJ, Jung SJ, An KN, Jin YL, Park RD, Kim KY, Shon BK, Kim TH (2002) Effect of chitinase-producing Paenibacillus illinoisensis KJA-424 on egg hatching of root-knot nematode (Meloidogyne incognita). J Microbiol Biotechnol 12(6):865–871
Jung WJ, Mabood F, Souleimanov A, Park RD, Smith DL (2008) Chitinases produced by Paenibacillus illinoisensis and Bacillus thuringiensis subsp. pakistani degrade Nod factor from Bradyrhizobium japonicum. Microbiol Res 163:345–349. https://doi.org/10.1016/j.micres.2006.06.013
Kumar MNVR (2000) A review of chitin and chitosan applications. React Function Polym 46:1–27. https://doi.org/10.1016/S1381-5148(00)00038-9
Kurbanoglu EB, Zibeyaz K, Ozdal M, Taskin M, Kurbanoglu NI (2010) Asymmetric reduction of substituted acetophenones using once immobilized Rhodotorula glutinis cells. Bioresour Technol 101(11):3825–3829. https://doi.org/10.1016/j.biortech.2010.01.016
Maity C, Samanta S, Halder SK, Mohapatra PKD, Pati BR, Jana M, Mondal KC (2011) Isozymes of α-amylases from newly isolated Bacillus thuringiensis CKB19: production from immobilized cells. Biotechnol Bioprocess Eng 16:312–319. https://doi.org/10.1007/s12257-010-0218-5
Manaf SA, Fuzi SFZM, Manas NHA, Illias RM, Oon LK, Hegde G, Man RC, Azelee NIW, Matias-Peralta HM (2021) Emergence of nanomaterial as potential immobilization support for whole cell biocatalyst and cell toxicity effects. Biotechnol Appl Biochem 68(6):1128–1138. https://doi.org/10.1002/bab.2034
Matsuo Y, Kurita M, Park JK, Tanaka K, Nakagawa T, Kawamukai M, Matsuda H (1999) Purification, characterization, and gene analysis of N-acetylglucosaminidase from Enterobacter sp. G-1. Biosci Biotechnol Biochem 63(7):1261–1268. https://doi.org/10.1271/bbb.63.1261
Miller GL (1959) Use of dinitrosaIicyIic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Nakatsu CH, Byappanahalli MN, Nevers MB (2019) Bacterial community 16S rRNA gene sequencing characterizes riverine microbial impact on lake Michigan. Front Microbiol 10:996. https://doi.org/10.3389/fmicb.2019.00996
Nurdebyandaru N, Mubarik NR, Prawasti TS (2010) Chitinolytic bacteria isolated from chili rhizosphere: chitinase characterization and application as biocontrol for Aphis gossypii. Microbial Indones 4(3):103–107. https://doi.org/10.5454/mi.4.3.1
Obradović NS, Kruni TC, Trifkovi TK, Bulatovi ML, Rakin MP, Rakin MB, Bugarski BM (2015) Influence of chitosan coating on mechanical stability of biopolymer carriers with probiotic starter culture in fermented whey beverages. Int J Polym Sci 2015:1–8. https://doi.org/10.1155/2015/732858
Okamoto Y, Yano R, Miyatake K, Tomohiro I, Shigemasa Y, Minami S (2003) Effects of chitin and chitosan on blood coagulation. Carbohydr Polym 53(3):337–342. https://doi.org/10.1016/S0144-8617(03)00076-6
Pillai CKS, Paul W, Sharma CP (2009) Chitin and chitosan polymers: chemistry, solubility and fiber formation. Prog Polym Sci 34:641–678. https://doi.org/10.1016/j.progpolymsci.2009.04.001
Sáez-Nieto JA, Medina-Pascual MJ, Carrasco G, Garrido N, Fernandez-Torres MA, Villalón P, Valdezate S (2017) Paenibacillus spp. isolated from human and environmental samples in Spain: detection of 11 new species. New Microbes New Infect 19:19–27. https://doi.org/10.1016/j.nmni.2017.05.006
Seo DJ, Jang YH, Park RD, Jung WJ (2012) Immobilization of chitinases from Streptomyces griseus and Paenibacillus illinoisensis on chitosan beads. Carbohydr Polym 88:391–394. https://doi.org/10.1016/j.carbpol.2011.12.009
Suresh PV (2012) Biodegradation of shrimp processing bio-waste and concomitant production of chitinase enzyme and N-acetyl-D-glucosamine by marine bacteria: production and process optimization. World J Microbiol Biotechnol 28:2945–2962. https://doi.org/10.1007/s11274-012-1106-2
Yadav M, Goswami P, Paritosh K, Kumar M, Pareek N, Vivekanand V (2019) Seafood waste: a source for preparation of commercially employable chitin/ chitosan materials. Bioresour Bioprocess 6:8. https://doi.org/10.1186/s40643-019-0243-y
Yang J, Zhang KQ (2019) Chitin synthesis and degradation in fungi: biology and enzymes. pp 153–167 In: Q. Yang and T. Fukamizo (eds.) Targeting Chitin-containing Organisms, Springer Nature, Singapore
Zur J, Wojcieszyńska D, Guzik U (2016) Metabolic responses of bacterial cells to immobilization. Molecules 21:1–15. https://doi.org/10.3390/molecules21070958
Acknowledgements
The authors would like to thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the scholarship to Francenya Kelley Lopes da Silva.
Funding
This research was supported by Conselho Nacional de Desenvolvimento Tecnológico (CNPq) Universal process #459678/2014–0.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study does not contain any experiments concerning human or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva, F.K.L., de Sa Alexandre, A.R., Casas, A.A. et al. Increased production of chitinase by a Paenibacillus illinoisensis isolated from Brazilian coastal soil when immobilized in alginate beads. Folia Microbiol 67, 935–945 (2022). https://doi.org/10.1007/s12223-022-00992-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-022-00992-3