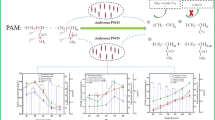
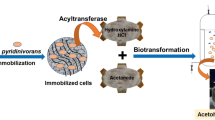
Abstract
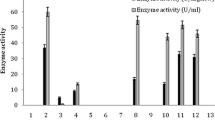
The nitrile hydratase (NHase) of Rhodococcus rhodochrous PA-34 catalyzed the conversion of acrylonitrile to acrylamide. The resting cells (having NHase activity) (8 %; 1 mL corresponds to 22 mg dry cell mass, DCM) were immobilized in polyacrylamide gel containing 12.5 % acrylamide, 0.6 % bisacrylamide, 0.2 % diammonium persulfate and 0.4 % TEMED. The polyacrylamide entrapped cells (1.12 mg DCM/mL) completely converted acrylonitrile in 3 h at 10 °C, using 0.1 mol/L potassium phosphate buffer. In a partitioned fed batch reactor, 432 g/L acrylamide was accumulated after 1 d. The polyacrylamide discs were recycled up to 3×; 405, 210 and 170 g/L acrylamide was produced in 1st, 2nd and 3rd recycling reactions. In four cycles, a total of 1217 g acrylamide was produced by recycling the same mass of entrapped cells.
Similar content being viewed by others
Abbreviations
- DCM:
-
dry cell mass
- NHase(s):
-
nitrile hydratase(s)
- PAA:
-
polyacrylamide
- PPB:
-
potassium phosphate buffer
- PVA:
-
polyvinyl alcohol
- TEMED:
-
N,N,N′,N′-tetramethylethylenediamine
References
Bernet N., Thiery A., Maestracci M., Arnaud A., Rios G.M., Galzy P.: Continuous immobilized cell reactor for amide hydrolysis. J.Ind.Microbiol.2, 129–136 (1987).
Bhalla T.C., Miura A., Wakamoto A., Ohba Y., Furuhashi K.: Asymmetric hydrolysis of α-aminonitriles to optically active amino acids by a nitrilase of Rhodococcus rhodochrous PA-34. Appl.Microbiol.Biotechnol.37, 184–190 (1992).
Cantarella M., Spera A., Leonetti P., Alfani F.: Influence of initial glucose concentration on nitrile hydratase production in Brevibacterium imperialis CBS 498-74. J.Mol.Catal.B Enzym.19-20, 405–414 (2002).
Chang N.H., Choi K.S., Lee C.Y.: Bench scale production of acrylamide using resting cells of Brevibacterium sp. CH2 in a fed batch reactor. Enzyme Microb.Technol.15, 979–984 (1993).
Chaplin M.F., Bucke C.: Enzyme Technology. Cambridge University Press (UK) 1990.
Fradet H., Arnaud A., Rios G., Galzy P.: Hydration of nitriles using a bacterial nitrile hydratase immobilized on DEAE-cellulose. Biotechnol.Bioeng.27, 1581–1585 (1985).
Hijort C.M., Godtfredsen S.E., Emborg C.: Isolation and characterization of nitrile hydratase from Rhodococcus sp. J.Chem.Technol. Biotechnol. 48, 217–226 (1990).
Jallageas J.C., Arnaud A., Galzy P.: Bioconversion of nitriles and their applications. Adv.Biochem.Eng.14, 1–32 (1980).
Kierstan M.P.J., Coughlan M.P.: Immobilization of cells and enzymes by gel entrapment, pp. 43–45 in Immobilized Cells and Enzymes (J. Woodward, Ed.). IRL Press, Oxford (UK) 1985.
Kim S.H., Padamkumar P., Oriel P.: Cobalt activation of Bacillus BR 449 nitrile hydratase expressed in E. coli. Appl.Biochem.Biotechnol.93, 597–603 (2001).
Kobayashi M., Nagasawa T., Yamada H.: Nitrilase of Rhodococcus rhodochrous J1 — purification and characterization. Eur.J.Biochem.182, 349–356 (1989).
Lee C.Y., Hwang Y.B., Chang H.N.: Acrylonitrile adaptation of Brevibacterium sp. CH1 for acrylamide production. Enzyme Microb. Technol.13, 53–58 (1991).
Mark H.F., Gaylord G.N., Bikales N.M.: Acrylamide: Encyclopedia of Polymer Science and Technology, Vol. 1, pp. 177–195. Interscience Publishers 1962.
Mitsubishi R.: Enzymatic production of acrylamide, pp. 71–75 in The Application of Biotechnology to Industrial Sustainability. OECD (France) 2001.
Nagasawa T., Takeuchi K., Yamada H.: Occurrence of a cobalt-induced and cobalt-containing nitrile hydratase in Rhodococcus rhodochrous J1. Biochem.Biophys.Res.Commun.155, 1008–1016 (1988).
Nagasawa T., Takeuchi K., Nardidei V., Mihara Y., Yamada H.: Optimum culture conditions for the production of cobalt-containing nitrile hydratase by Rhodococcus rhodochrous J1. Appl.Microbiol.Biotechnol.34, 783–788 (1991).
Nagasawa T., Shimizu H., Yamada H.: The superiority of the third-generation catalyst Rhodococcus rhodochrous J1 nitrile hydratase for industrial production of acrylamide. Appl.Microbiol.Biotechnol.40, 189–195 (1993).
Precigou S., Wieser M., Pommares P., Goulas P., Duran R.: Rhodococcus pyridinovorans MW 3, a bacterium producing a nitrile hydratase. Biotechnol.Lett.26, 1379–1384 (2004).
Raj J., Prasad S., Bhalla T.C.: Rhodococcus rhodochrous PA-34, a potential catalyst for acrylamide synthesis. Process Biochem.41, 1359–1363 (2006).
Raj J., Seth A., Prasad S., Bhalla T.C.: Bioconversion of butyronitrile to butyramide using whole cells of Rhodococcus rhodochrous PA-34. Appl.Microbiol.Biotechnol.74, 535–539 (2007).
Raj J., Sharma N.N., Prasad S., Bhalla T.C.: Acrylamide synthesis using agar entrapped cells of Rhodococcus rhodochrous PA-34 in a partitioned fed batch reactor. J.Industr.Microbiol.Technol.35, 35–40 (2008).
Ramakrishna C., Desai J.D.: Bioconversion of acrylonitrile to acrylamide by Arthrobacter sp. IPCB-3. Indian J.Exp.Biol.31, 173–177 (1993).
Ramakrishna C., Dave H., Ravindranathan M.: Microbial metabolism of nitriles and its biotechnological potential. J.Sci.Ind.Res.58, 925–947 (1999).
Sankhian U.D., Kumar H., Chand D., Kumar D., Bhalla T.C.: Nitrile hydratase of Rhodococcus rhodochrous NHB-2: optimization of conditions for production of enzyme and conversion of acrylonitrile to acrylamide. Asian J.Microbiol.Biotech.Env. Sci.5, 217–223 (2003).
Watanabe I., Satoh Y., Enomoto K., Seki S., Sakashita K.: Optimal conditions for cultivation of Rhodococcus sp. N-774 and for conversion of acrylonitrile to acrylamide by resting cells. Agric.Biol.Chem.51, 3201–3206 (1987).
Yamada H., Ryuno K., Nagasawa T., Enomoto K., Watanabe I.: Optimum culture conditions for production by Pseudomonas chloraraphis B23 of nitrile hydratase. Agric.Biol.Chem.50, 2859–2865 (1986).
Yamada H., Kobayashi M.: Nitrile hydratase and its application to industrial production of acrylamide. Biosci.Biotech.Biochem.60, 1391–1400 (1996).
Youn K.B., Hwan H.G.: Production of acrylamide using immobilized cells of Rhodococcus rhodochrous M33. Biotechnol.Bioprocess. Eng.7, 194–200 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raj, J., Prasad, S., Sharma, N.N. et al. Bioconversion of acrylonitrile to acrylamide using polyacrylamide entrapped cells of Rhodococcus rhodochrous PA-34. Folia Microbiol 55, 442–446 (2010). https://doi.org/10.1007/s12223-010-0074-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-010-0074-x