Abstract
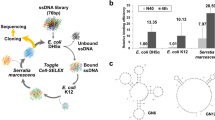
DNA aptamers were developed against lipopolysaccharide (LPS) from E. coli O111:B4 and shown to bind both LPS and E. coli by a colorimetric enzyme-based microplate assay. The polyclonal aptamers were coupled to human C1qrs protein either directly using a bifunctional linker or indirectly using biotinylated aptamers and a streptavidin-C1qrs complex. Both systems significantly reduced colony counts when applied to E. coli O111:B4 and K12 strains across a series of 10× dilutions of the bacteria in the presence of human serum; it was diluted 1: 103 in order to avoid significant bacterial lysis by the competing alternate pathway of complement activation. A number of candidate DNA aptamer sequences were cloned and sequenced from the anti-LPS aptamer library for future screening of antibacterial or “antibiotic” potential and to aid in eventual development of an alternative therapy for antibiotic-resistant bacterial infections.
Similar content being viewed by others
Abbreviations
- 1XBB:
-
single strength binding buffer
- MB(s):
-
magnetic microbead(s)
- Apt(s):
-
aptamer(s)
- P b :
-
p value between treatment groups
- CFU:
-
colony-forming units
- PBS:
-
phosphate-buffered saline
- ds:
-
double-stranded
- PCR:
-
polymerase chain reaction
- EGS:
-
ethane-1,2-diol bis(sulfosuccinimidyl-succinate)
- RT:
-
room temperature
- SELEX:
-
systematic evolution of ligands by exponential enrichment
- GVB:
-
gelatin veronal buffer
- HSCP(s):
-
human serum complement protein(s)
- SMC:
-
sulfosuccinimidyl 4-(N-maleimido-methyl)cyclohexane-1-carboxylate
- IgG, IgM:
-
immunoglobulins G, M
- KDO:
-
3-deoxy-D-manno-octulosonic acid
- ss:
-
single-stranded
- LPS:
-
lipopolysaccharide
- TSA:
-
tryptic soy agar
References
Alanis A.J.: Resistance to antibiotics: are we in the post-antibiotic era? Arch.Med.Res. 36, 697–705 (2005).
Bantroch S., Bühler T., Lam J.S.: Appropriate coating methods and other conditions for enzyme-linked immunosorbent assay of smooth, rough, and neutral lipopolysaccharides of Pseudomonas aeruginosa. Clin.Diagn.Lab.Immunol. 1, 55–62 (1994).
Barnes M.G., Weiss A.A.: BrkA protein of Bordetella pertussis inhibits the classical pathway of complement after C1 deposition. Infect.Immun. 69, 3067–3072 (2001).
Boyle-Vavra S., Daum R.S.: Community-acquired methicillin-resistant Staphylococcus aureus: the role of Panton-Valentine leukocidin. Lab.Invest. 87, 3–9 (2007).
Brody E.N., Gold L.: Aptamers as therapeutic and diagnostic agents. J.Biotechnol. 74, 5–13 (2000).
Bruno J.G., Kiel J.L.: In vitro selection of DNA aptamers to anthrax spores with electrochemiluminescence detection. Biosens.Bioelectron. 14, 457–464 (1999).
Bruno J.G., Kiel J.L.: Use of magnetic beads in selection and detection of biotoxin aptamers by ECL and enzymatic methods. Bio-Techniques 32, 178–183 (2002).
Cassadevall A., Dadachova E., Pirofski L.: Passive antibody therapy for infectious diseases. Nature Rev.Microbiol. 2, 695–703 (2004).
Chu T.C., Marks J.W., Lavery L.A., Faulkner S., Rosenblum M.G., Ellington A.D., Levy M.: Aptamer:toxin conjugates that specifically target prostate tumor cells. Cancer Res. 66, 5989–5992 (2006).
Cload S.T., Mccauley T.G., Keefe A.D., Healy J.M., Wilson C.: Properties of therapeutic aptamers, pp. 398–399 in S. Klussmann (Ed.): The Aptamer Handbook, Chapter 17. Wiley-VCH Verlag, Weinheim (Germany) 2006.
Chopra I.: Research and development of antibacterial agents. Curr.Opin.Microbiol. 1, 495–501 (1998).
Dougan H., Lyster D.M., Vo C.V., Stafford A., Weitz J.I., Hobbs J.B.: Extending the lifetime of anticoagulant oligodeoxynucleotide aptamers in blood. Nucl.Med.Biol. 27, 289–297 (2000).
Dunn D.L., Bogard W.C., Cerra F.B.: Enhanced survival during murine Gram-negative bacterial sepsis by use of a murine monoclonal antibody. Arch.Surg. 120, 50–52 (1985).
Dwarakanath S., Bruno J.G., Shastry A., Phillips T., John A., Kumar A., Stephenson L.D.: Quantum dot-antibody and aptamer conjugates shift fluorescence upon binding bacteria. Biochem.Biophys.Res.Comm. 325, 739–743 (2004).
Dwarakanath S., Satyanarayana S., Bruno J.G., Vattem D., Rao P.M., Ikanovic M., Phillips T.: Ultrasensitive fluorescent nanoparticle-based binding assays for foodborne and waterborne pathogens of clinical interest. J.Clin.Ligand Assay 29, 136–142 (2006).
Dwarakanath S., Bruno J.G., Athmaram T.N., Bali G., Vattem D., Rao P.: Antibody-quantum dot conjugates exhibit enhanced antibacterial effect vs. unconjugated quantum dots. Folia Microbiol. 52, 31–34 (2007).
Hofacker I.L.: Vienna RNA secondary structure server. Nucl.Acids Res. 31, 3429–3431 (2003).
Horstman A.L., Bauman S.J., Kuehn M.J.: Lipopolysaccharide 3-deoxy-D-manno-octulosonic acid (KDO) core determines bacterial association of secreted toxins. J.Biol.Chem. 279, 8070–8075 (2004).
Ikanovic M., Rudzinski W.E., Bruno J.G., Allman A., Carrillo M.P., Dwarakanath S., Bhahdigadi S., Rao P., Kiel J.L., Andrews C.J.: Fluorescence assay based on aptamer-quantum dot binding to Bacillus thuringiensis spores. J.Fluoresc. 17, 193–199 (2007).
Jayasena S.D.: Aptamers: an emerging class of molecules that rival antibodies in diagnostics. Clin.Chem. 45, 1628–1650 (1999).
Joiner K.A., Schmetz M.A., Sanders M.E., Murray T.G., Hammer C.H., Dourmashkin R., Frank M.M.: Multimeric complement component C9 is necessary for killing of Escherichia coli J5 by terminal attack complex C5b-9. Proc.Nat.Acad.Sci.USA 82, 4808–4812 (1985).
Kastowsky M., Gutberlet T., Bradaczek H.: Molecular modeling of the three-dimensional structure and conformational flexibility of bacterial lipopolysaccharide. J.Bact. 174, 4789–4806 (1992).
Keller M.A., Stiehm E.R.: Passive immunity in prevention and treatment of infectious diseases. Clin.Microbiol.Rev. 13, 602–614 (2000).
Linksmüller-Loennies S., Brade L., Brade H.: Neutralizing and cross-reactive antibodies against enterobacterial lipopolysaccharide. Internat.J.Med.Microbiol. 297, 321–340 (2007).
Lodowska J., Wolny D., Weglarz L., Dzierzewicz Z.: The structural diversity of lipid A from Gram-negative bacteria. Postepy Hig.Med.Dosw. 61, 106–121 (2007).
Mann D., Reinemann C., Stoltenberg R., Strehlitz B.: In vitro selection of DNA aptamers binding ethanolamine. Biochem.Biophys. Res.Comm. 338, 1928–1934 (2005).
Pan Q., Zhang X.L., Wu H.L., He P.W., Wang F., Zhang M.S., Hu J.M., Xia B., Wu J.: Aptamers that preferentially bind type IVB pilli and inhibit human monocytic-cell invasion by Salmonella enterica serovar Typhi. Antimicrob.Agents Chemother. 49, 4052–4060 (2005).
Santalucia J.: A unified view of polymer, dumbbell, and oligonucleotide DNA nearest-neighbor thermodynamics. Proc.Nat.Acad. Sci.USA 95, 1460–1465 (1998).
Vivekananda J., Kiel J.L.: Anti-Francisella tularensis DNA aptamers detect tularemia antigen from different subspecies by aptamerlinked immobilized sorbent assay. Lab.Invest. 86, 610–618 (2006).
Wlotzka B., Leva S., Eschgfäller B., Burmeister J., Kleinjung F., Kaduk C., Muhn P., Hess-Stumpp H., Klussmann S.: In vivo properties of an anti-GnRH spiegelmer: an example of an oligonucleotide-based therapeutic substance class. Proc. Nat.Acad.Sci.USA 99, 8898–8902 (2002).
Zhen B., Song Y.J., Guo Z.B., Wang J., Zhang M.L., Yu S.Y., Yang R.F.: In vitro selection and affinity function of the aptamers to Bacillus anthracis spores by SELEX. Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao (Shanghai) 34, 635–642 (2002).
Zhong Z., Burns T., Chang Q., Carroll M., Pirofski L.: Molecular and functional characteristics of a protective human monoclonal antibody to serotype 8 Streptococcus pneumoniae capsular polysaccharide. Infect.Immun. 67, 4119–4127 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bruno, J.G., Carrillo, M.P. & Phillips, T. In Vitro antibacterial effects of antilipopolysaccharide DNA aptamer-C1qrs complexes. Folia Microbiol 53, 295–302 (2008). https://doi.org/10.1007/s12223-008-0046-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-008-0046-6