Abstract
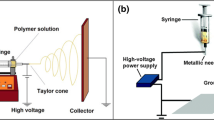
Polyethersulfone (PES) nanofibers are produced by electrospinning solutions of PES/dimethylforamide (DMF), PES/N-methylpyrrolidone (NMP) and PES/(NMP:DMF) (of different NMP:DMF ratios) at temperature of 40 °C and various levels of relative humidity (RH). The influence of environmental conditions on bead formation as well as surface and interior morphologies of electrospun fibers is discussed through the phase diagram of H2O/DMF/PES and H2O/NMP/PES systems. The former case has small miscibility area while the latter one has large of which. The results demonstrate the contribution of RH of operating environment to morphology evolution of nanofibers. If the size of miscibility area increases e.g. H2O/NMP/PES system, a higher values of RH is needed to stabilize the formation of fibers. For this system, low level of humidity leads to develop beads as well as bead-on-string morphology. Adding the second solvent i.e. DMF into the PES/NMP solution shifts the binodal boundary toward the polymer-solvent side meaning a smaller miscibility area. In consequence, formation of fiber can be stabilized under broad range of humidity levels i.e. from low to high level of humidity. Implications regarding formation of surface pores by manipulating phase behavior of ternary system as well as RH of ambient conditions are discussed related to physico-chemical nature of solvent.
Similar content being viewed by others
References
A. Formhals, U. S. Patent, 1975504 (1934).
G. Taylor, Proc. Roy. Soc. Lond. A., 313, 453 (1969).
M. M. Hohman, M. Shin, G. Rutledge, and M. P. Brenner, Phys. Fluids, 13, 2201 (2001).
M. M. Hohman, M. Shin, G. Rutledge, and M. P. Brenner, Phys. Fluids, 13, 2221 (2001).
I. Ahmed, A. Idris, M. Y. Noordin, and R. Rajput, Ind. Eng. Chem. Res., 50, 2272 (2011).
Z. Tang, C. Qiu, J. R. McCutcheon, K. Yoon, H. Ma, D. Fang, E. Lee, C. Kopp, B. S. Hsiao, and B. Chu, J. Polym. Sci. Pol. Phys., 47, 2288 (2009).
S. Sh. Homaeigohar, K. Buhr, and K. Ebert, J. Membrane Sci., 365, 68 (2010).
S. Sh. Homaeigohar and M. Elbahri, J. Colloid. Interf. Sci., 372, 6 (2012).
I. Shahabi, V. Haddadi-Asl, E. Seyedjafari, F. Babaeijandaghi, and M. Soleimani, Biochem. Bioph. Res. Co., 382, 129 (2009).
Y. Zhang, Q. Wei, Q. Zhang, J. Li, J. Yang, and C. Zhao, Sep. Sci. Technol., 46, 1615 (2011).
J. S. Travis, A. Horst, and von Recum, Biomaterials, 29, 1989 (2008).
V. Beachley and X. Wen, Mater. Sci. Eng. C Mater. Biol. Appl., 29, 2448 (2009).
Z. M. Huang, Y. Z. Zhang, M. Kotaki, and S. Ramakrishna, Compos. Sci. Tech., 63, 2223 (2003).
A. Podgorski, A. Bałazy, and L. Gradon, Chem. Eng. Sci., 61, 6804 (2006).
W. W. F. Leung, C. H. Hung, and P. T. Yuen, Sep. Purif. Technol., 71, 30 (2010).
R. Ramaseshan and S. Ramakrishna, J. Am. Ceram. Soc., 90, 1836 (2007).
Y. Miyauchi, B. Ding, and S. Shiratori, Nanotechnology, 17, 5151 (2006).
J. Zheng, A. He, J. Li, J. Xu, and C. C. Han, Polymer, 47, 7095 (2006).
A. C. Patel, Sh. Li, Ce Wang, W. Zhang, and Y. Wei, Chem. Mater., 19, 1231 (2007).
Y. Zhang, J. Li, G. An, and X. He, Sensor. Actuat. B-Chem. 144, 43 (2010).
S. Megelski, J. S. Stephens, D. Bruce Chase, and J. F. Rabolt, Macromolecules, 35, 8456 (2002).
S. A. Theron, E. Zussman, and A. L. Yarin, Polymer, 45, 2017 (2004).
T. Jarusuwannapoom, W. Hongrojjanawiwat, S. Jitjaicham, L. Wannatong, M. Nithitanakul, C. Pattamaprom, P. Koombhongse, R. Rangkupan, and P. Supaphol, Eur. Polym. J., 41, 409 (2005).
C. J. Luo, M. Nangrejo, and M. Edirisinghe, Polymer, 51, 1654 (2010).
J. H. Yu, S. V. Fridrikh, and G. C. Rutledge, Polymer, 47, 4789 (2006).
C. L. Casper, J. S. Stephens, N. G. Tassi, D. B. Chase, and J. F. Rabolt, Macromolecules, 37, 573 (2004).
C. L. Pai, M. C. Boyce, and G. C. Rutledge, Macromolecules, 42, 2102 (2009).
S. D. Vrieze, T. V. Camp, A. Nelvig, B. Hagstrom, P. Westbroek, and K. D. Clerck, J. Mater. Sci., 44, 1357 (2009).
S. Tripatanasuwan, Z. Zhong, and D. H. Reneker, Polymer, 48, 5742 (2007).
J. Zheng, H. Zhang, Z. Zhao, and C. C. Han, Polymer, 53, 546 (2012).
Z. Qi, H. Yu, Y. Chen, and M. Zhu, Mater. Lett., 63, 415 (2009).
X. Yu, H. Xiang, Y. Long, N. Zhao, X. Zhang, and J. Xu, Mat. Lett., 64, 2407 (2010).
H. Tompa, “Polymer Solution”, London, Butterworth, 1956.
L. Yilmaz and A. J. McHugh, J. Appl. Polym. Sci., 31, 997 (1986).
F. W. Altena and C. A. Smolders, Macromolecules, 15, 1491 (1982).
M. Karimi, W. Albrecht, M. Heuchel, M. H. Kish, J. Frahn, T. H. Weigel, D. Hofmann, H. Modarress, and A. Lendlein, J. Membrane Sci., 265, 1 (2005).
M. Karimi, W. Albrecht, M. Heuchel, T. H. Weigel, and A. Lendlein, Polymer, 49, 2587 (2008).
R. M. Boom, Ph. D. Dissertation, University of Twente, Enschede, The Netherlands, 1992.
M. Karimi, M. Heuchel, W. Albrecht, and D. Hofmann, J. Membrane Sci., 292, 80 (2007).
C. Barth, R. Horst, and B. A. Wolf, J. Chem. Thermodyn, 30, 641 (1998).
H. C. Park, Y. P. Kim, H. Y. Kim, and Y. S. Kang, J. Membrane Sci., 156, 169 (1999).
S. P. Nunes and T. Inoue, J. Membrane Sci., 111, 93 (1996).
C. Barth and B. A. Wolf, Macromol. Chem. Phys., 201, 365 (2000).
K. H. Lee, H. Y. Kim, H. J. Bang, Y. H. Jung, and S. G. Lee, Polymer, 44, 4029 (2003).
G. Eda and S. Shivkumar, J. Appl. Polym. Sci., 106, 475 (2007).
S. A. Theron, E. Zussman, and A. L. Yarin, Polymer, 45, 2017 (2004).
A. L. Yarin, “Free Liquid Jets and Films: Hydrodynamics and Rheology”, Longman Scientific and Technical, New York, 1993.
L. Wannatong, A. Sirivat, and P. Supaphol, Polym. Int., 53, 1851 (2004).
J. A. Riddick and W. B. Bunger, “Organic Solvents”, 3rd ed., Vol. II, pp.454–475, Wiley-Interscience, New York, 1970.
G. P. Johari, A. Hallbrucker, and E. Mayer, Nature, 330, 552 (1987).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fashandi, H., Karimi, M. Evidence for the impression of phase behavior of nonsolvent/solvent/polymer ternary system on morphology of polyethersulfone electrospun nanofibers. Fibers Polym 15, 1375–1386 (2014). https://doi.org/10.1007/s12221-014-1375-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12221-014-1375-5