Abstract
Multicopper oxidases (MCOs), such as ascorbic acid oxidase and ceruloplasmin, are multidomain proteins capable of oxidizing many structurally unrelated compounds reducing oxygen to water without ever generating reactive oxygen species. While MCOs show great oxidative versatility, they can only transfer electrons to molecular oxygen, which is the obligate electron acceptor. Therefore, MCOs could also be considered as “O2 consuming enzymes”, thus contributing to create those states of hypoxia that are normally found in tissues, cells and cell compartments. Since hypoxia is also a common feature of many rapidly growing solid tumors, we postulate that the regulation of GPI-ceruloplasmin isoform, present on the surface of the plasma membrane, could be the molecular event in the creation and the maintenance of hypoxia in tumor cells. By silencing the different MCO genes with siRNA, it would appear possible to attempt to overcome tumor hypoxia, thus improving the efficiency of radiotherapy.


Similar content being viewed by others
References
Arrigoni O, Chinni E, Ciraci S, De Tullio MC (2003) In vivo elicitation of ascorbate oxidase activity and its possible role in photosynthesizing leaves. Rendiconti Accademia dei Lincei Scienze Fisiche e Naturali 9:127–134
Asikainen TM, White CW (2005) Antioxidant defenses in the preterm lung: role for hypoxia-inducible factors in BPD? Toxicol Appl Pharmacol 203:177–188
Bento I, Peixoto C, Zaitsev VN, Lindley PF (2007) Ceruloplasmin revisited: structural and functional roles of various metal cation-binding sites. Acta Crystallogr Sect D Biol Crystallogr 63:240–248
Berner RA, Beerling DJ, Dudley R, Robinson JM, Wildman RA (2003) Phanerozoic atmospheric oxygen. Annu Rev Earth Planet Sci 31:105–134
Bielli P, Calabrese L (2002) Structure to function relationships in ceruloplasmin: a “moonlighting” protein. Cell Mol Life Sci 59:1413–1427
Bourhis J (2006) Hypoxia response pathways and radiotherapy for head and neck cancer. J Clin Oncol 24:725–726
Brahimi-Horn MC, Chiche J, Pouyssègur J (2007) Hypoxia and cancer. J Mol Med 85:1301–1307
Braun RD, Beatty AL (2006) Modeling of oxygen transport across tumor multicellular layers. Microvasc Res 73:113–123
Buchler P, Reber HA, Lavey RS, Tomlinson J, Buchler MW, Friess H, Hines OJ (2004) Tumor hypoxia correlates with metastatic tumor growth of pancreatic cancer in orthotopic murine model. J Surg Res 120:295–303
Bui T, Thompson CB (2006) Cancer’s sweet tooth. Cancer Cell 9:419–420
Canfield DE (2005) The early history of atmospheric oxygen. Annu Rev Earth Planet Sci 33:1–36
Cerveza PJ, Mehrbod F, Cotton SJ, Lomeli N, Linder MC, Fonda EG, Wickler SJ (2000) Milk ceruloplasmin and its expression by mammary gland and liver in pigs. Arch Biochem Biophys 373:451–461
Danzeisen R, Ponnambalam S, Lea RG, Page K, Gambling L, McArdle HJ (2000) The effect of ceruloplasmin on iron release from placental (BeWo) cells; evidence for an endogenous Cu oxidase. Placenta 21:805–812
De Cabo RC, Gonzalez-Reyes JA, Navas P (1993) The onset of cell proliferation is stimulated by ascorbate free radical in onion root primordia. Biol Cell 77:231–233
De Tullio MC, Liso R, Arrigoni O (2004) Ascorbic acid oxidase: an enzyme in search of a role. Biol Plant 48:161–166
De Tullio MC, Ciraci S, Liso R, Arrigoni O (2007) Ascorbic acid oxidase is dinamically regulated by lights and oxygen. A tool for oxygen management in plants? J Plant Physiol 164:39–46
Fleming RE, Whitman IP, Gitlin JD (1991) Induction of ceruloplasmin gene expression in rat lung during inflammation and hyperoxia. Am J Physiol 260:68–74
Floris G, Medda R, Padiglia A, Musci G (2000) The physiopathological significance of ceruloplasmin. A possible therapeutic approach. Biochem Pharmacol 60:1735–1741
Fortna RR, Watson HA, Nyquist SE (1999) Glycosyl phosphatidylinositol-anchored ceruloplasmin is expressed by rat Sertoli cells and is concentrated in detergent-insoluble membrane fractions. Biol Reprod 61:1042–1049
Fukumura D, Jain RK (2006) Tumor microenvironment abnormalities: causes, consequences, and strategies to normalize. J Cell Biochem 101:937–949
Ganz T (2005) Cellular iron: ferroportin is the only way out. Cell Metab 1:155–157
Gnaiger E, Mendez G, Hand SC (2000) High phosphorylation efficiency and depression of uncoupled respiration in mitochondria under hypoxia. Proc Natl Acad Sci USA 97:11080–11085
Gray MW, Burger G, Lang BF (1999) Mitochondrial evolution. Science 283:1476–1481
Gwak GY, Yoon JH, Kim KM, Lee HS, Chung JW, Gores GJ (2005) Hypoxia stimulates proliferation of human hepatome cells through the induction of hexokinase II expression. J Hepatol 42:358–364
Gyulikhandanova NE, Tsymbalenko NV, Platonova NA, Babich VS, Puchkova LV (2004) Regulation of ceruloplasmin gene in mammals. Bull Exp Biol Med 137:485–489
Harris ED (1999) Ceruloplasmin and iron: vindication after 30 years. Nutrition 15:72–73
Healy J, Tipton K (2007) Ceruloplasmin and what it might do. J Neural Trasnm 114:777–781
Hidalgo A, Garcia-Herdugo G, Gonzales-Reyes JA, Morrè DJ, Navas P (1991) Ascorbate free radical stimulates onion root growth by increasing cell elongation. Bot Gazette 152:282–288
Hochstrasser H, Tomiuk J, Walter U, Behnke S, Spiegel J, Kruger R, Becker G, Riess O, Berg D (2005) Functional relevance of ceruloplasmin mutations in Parkinson’s disease. FASEB J 19:1851–1853
Hockel M, Vaupel P (2001) Tumor hypoxia: definitions and current clinical, biologic, and molecular aspects. J Natl Cancer Inst 93:266–276
Hoegger PJ, Kilaru S, James TY, Thacker JR, Kues U (2006) Phylogenetic comparison and classification of laccase and related multicopper oxidase protein sequences. FEBS J 273:2308–2326
Hoopes JT, Dean JF (2004) Ferroxidase activity in a laccase-like multicopper oxidase from Liriodendron tulipifera. Plant Physiol Biochem 42:27–33
James JL, Stone PR, Chamley LW (2005) The regulation of trophoblast differentiation by oxygen in the first trimester of pregnancy. Hum Reprod Update 12:137–144
Jeong SY, David S (2003) Glycosylphosphatidylinositol-anchored ceruloplasmin is required for iron efflux from cells in the central nervous system. J Biol Chem 278:27144–27148
Kato N, Esaka M (1999) Changes in ascorbate oxidase gene expression and ascorbate levels in cell division and cell elongation in tobacco cells. Physiol Plant 105:321–329
Klomp LWJ, Gitlin JD (1996) Expression of the ceruloplasmin gene in the human retina and brain: implications for a pathogenic model in aceruloplasminemia. Hum Mol Genet 5:1989–1996
Kolmeier KH, Silverstein MN, Fleisher GA (2006) Anaerobic glycolysis in normal and leukemic bone-marrow leukocytes: effect of methyl-glyoxal-bis-guanylhydrazone dihydrochloride. Cancer 19:1195–1199
Lee JH, Horak CE, Khanna C, Meng Z, Yu LR, Veenstra TD, Steeg PS (2008) Alterations in Gemin5 expression contribute to alternative mRNA splicing patterns and tumor cell motility. Cancer Res 68:639–644
Li XF, O’Donoghue JA (2008) Hypoxia in microcospic tumors. Cancer Lett 264:172–180
Li D, Marks JD, Schumacker PT, Young RM, Brorson JR (2005) Physiological hypoxia promotes survival of cultured cortical neurons. Eur J Neurosci 22:1319–1326
Li YW, Li L, Zhao JY (2008) An inhibition of ceruloplasmin expression induced by cerebral ischemia in the cortex and hippocampus of rats. Neurosci Bull 24:13–20
Liso R, De Tullio MC, Ciraci S, Balestrini R, La Rocca N, Bruno L, Chiappetta A, Bitonti MB, Bonfante P, Arrigoni O (2004) Localization of ascorbic acid, ascorbic acid oxidase, and glutathione in roots of Cucurbita maxima, L. J Exp Bot 55:2589–2597
Martinive P, Defresne F, Bouzin C, Saliez J, Lair F, Gregoire V, Michiels C, Dessy C, Feron O (2006) Preconditioning of tumor vasculature and tumor cells by intermittent hypoxia: implications for anticancer therapies. Cancer Res 66:11736–11744
Mei G, Di Venere A, Nicolai E, Rosato N, Finazzi-Agrò A (2003) Probing the conformational stability of two different copper proteins: a dynamic fluorescence study on azurin and ascorbate oxidase. J Fluoresc 13:33–39
Messerschmidt A (1997) Spatial structures of ascorbic oxidases, laccase and related proteins: implications for the catalytic mechanism. In: World Scientific Publishing Co. Pte. Ltd (ed) Multi-copper oxidases, chap 2. pp 23–80
Messerschmidt A, Huber R (1990) The blue oxidases, ascorbate oxidase, laccase and ceruplasmin. Eur J Biochem 187:341–352
Musci G (2001) Ceruloplasmin, the unique multi-copper oxidase of vertebrates. Prot Pept Lett 8:159–169
Nakamura K, Go N (2005) Function and molecular evolution of multicopper blue proteins. Cell Mol Life Sci 62:2050–2066
Nordsmark M, Bentzen SM, Rudat V, Brizel D, Lartigau E, Stadler P, Becker A, Adam M, Molls M, Dunst J, Terris DJ, Overgaard J (2005) Prognostic value of tumor oxygenation in 397 head and neck tumors after primary radiation therapy. An international multi-center study. Radiother Oncol 77:18–24
Patel BN, David S (1997) A novel GPI_anchored form of ceruloplasmin expressed by astrocytes. J Biol Chem 272:20185–20190
Ribatti D, Nico B, Crivellato E, Vacca A (2007) The structure of the vascular network of tumors. Cancer Lett 248:18–23
Roberts SA, Weichsel A, Grass G, Thakali K, Hazzard JT, Tollin G, Rensing C, Montfort WR (2002) Crystal structure and electron transfer kinetics of CueO, a multicopper oxidase required for copper homeostasis in Escherichia coli. Proc Natl Acad Sci USA 99:2766–2771
Spencer HJ (1939) The effect of puncturing individual latex tubes of Euphorbia Wulfenii. Ann Bot 3:4–9
Thomas T, Macpherson A, Rogers P (1995) Ceruloplasmin gene expression in the rat uterus. Biochim Biophys Acta 1261:77–82
Vasin AV, Platonova NA, Klotchenko SA, Tsymbalenko NV, Puchkova LV (2004) Expression of ceruloplasmin pseudogene in cultured human cells. Doklady Biochem Biophys 397:254–257
Vasin AV, Platonova NA, Povalihin RG, Klotchenko SA, Samsonov SA, Tsymbalenko NV, Puchkova LV (2005) Mitochondrial ceruplasmin of mammals. Mol Biol 39:42–52
Vizàn P, Alcarraz-Vizàn G, Diaz-Moralli S, Solovjeva ON, Frederiks WM, Cascante M (2009) Modulation of pentose phosphate pathway during cell cycle progression in human colon adenocarcinoma cell line HT29. Int J Cancer 124:2789–2796
Young SD, Marshall RS, Hill RP (1988) Hypoxia induces DNA overreplication and enhances metastatic potential of murine tumor cells. Proc Natl Acad Sci USA 85:9533–9537
Zowczak M, Iskra M, Paszkowski J, Manczak M, Torlinski L, Wysocka E (2001) Oxidase activity of ceruloplasmin and concentrations of copper and zinc in serum of cancer patients. J Trace Elem Med Biol 15:193–196
Acknowledgments
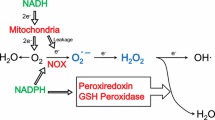
We are grateful to Prof. H. J. McArdle for the gift of Fig. 2 [reprinted from Danzeisen R, Ponnambalam S, Lea RG, Page K, Gambling L, McArdle HJ (2000) The effect of ceruloplasmin on iron release from placental (BeWo) cells: evidence for an endogenous Cu oxidase. Placenta 21:805–812. With permission from Elsevier].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arrigoni, R., Arrigoni, O. Multicopper oxidases: an innovative approach for oxygen management of aerobic organisms. Rend. Fis. Acc. Lincei 21, 71–80 (2010). https://doi.org/10.1007/s12210-009-0071-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12210-009-0071-7