Abstract
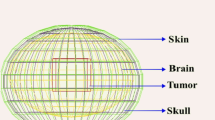
Neutron capture therapy (NCT) with various concentrations of gadolinium (157Gd) is one of the treatment modalities for glioblastoma (GBM) tumors. Current study aims to evaluate how variations of 157Gd concentration and cell oxygen levels can affect the relative biological effectiveness (RBE) of gadolinium neutron capture therapy (GdNCT) technique through a hybrid Monte Carlo (MC) simulation approach. At first, Snyder phantom including a spherical tumor was simulated by Geant4 MC code and relevant energy electron spectra to different 157Gd concentrations including 100, 250, 500, and 1000 ppm were calculated following the neutron irradiation of simulated phantom. Scored energy electron spectra were then imported to Monte Carlo damage simulation (MCDS) code to estimate RBE values (both RBESSB and RBEDSB) at different gadolinium concentrations and oxygen levels from 10 to 100%. The results indicate that variations of 157Gd can affect the energy spectrum of released secondary electrons including Auger electrons. Variation of gadolinium concentration from 100 to 1000 ppm in tumor region can change RBESSB and RBEDSB values by about 0.1% and 0.5%, respectively. Besides, maximum variations of 4.3% and 2% were calculated for RBEDSB and RBESSB when cell oxygen level changed from 10 to 100%. From the results, variations of considered gadolinium and oxygen concentrations during GdNCT can influence RBE values. Nevertheless, due to the not remarkable changes in the intensity of Auger electrons, a slight difference in RBE values would be expected at various 157Gd concentrations, although considerable RBE changes were calculated relevant to the oxygen alternations inside tumor tissue.





Similar content being viewed by others
References
Takagaki M, Hosmane NS. Gadolinium neutron capture therapy for malignant brain tumors. Aino J. 2008;6.
Hottinger AF, Yoon H, DeAngelis LM, Abrey LE. Neurological outcome of long-term glioblastoma survivors. J Neurooncol. 2009;95:301–5.
Farhood B, Samadian H, Ghorbani M, Zakariaee SS, Knaup C. Physical, dosimetric and clinical aspects and delivery systems in neutron capture therapy. Rep Pract Oncol Radiother. 2018;23:462–73.
Takagaki M, Tomaru N, Maguire JA, Hosmane NS. Future applications of Boron and Gadolinium neutron capture therapy. In: Hosmane NS, editor. Boron science, new technologies and applications. Boca Raton: CRC Press; 2011. p. 243–76.
Pozzi EC, Cardoso JE, Colombo LL, Thorp S, Monti Hughes A, Molinari AJ, et al. Boron neutron capture therapy (BNCT) for liver metastasis: therapeutic efficacy in an experimental model. Radiat Environ Biophys. 2012;51:331–9.
Nakagawa N, Akai F, Fukawa N, Fujita Y, Suzuki M, Ono K, Taneda M. Early effects of boron neutron capture therapy on rat glioma models. Brain Tumor Pathol. 2007;24:7–13.
Kanygin VV, Kichigin AI, Krivoshapkin AL, Taskaev SY. Perspectives of boron-neutron capture therapy of malignant brain tumors. In: AIP Conference Proceedings. 2017. p. 020030.
Enger SA, Giusti V, Fortin MA, Lundqvist H, Rosenschöld P. Dosimetry for gadolinium neutron capture therapy (GdNCT). Radiat Measure. 2013;59:233–40.
Safavi-Naeini M, Chacon A, Guatelli S, Franklin DR, Bambery K, Gregoire MC, et al. Opportunistic dose amplification for proton and carbon ion therapy via capture of internally generated thermal neutrons. Sci Rep. 2018;8:16257.
Kulabdullaev GA, Abdullaeva GA, Kim AA, Rakhmonov TT, Kurmantaev A. About radiation innatGd for neutron capture therapy. J Health Sci. 2016;4:35–44.
Masiakowski JT, Horton JL, Peters LJ. Gadolinium neutron capture therapy for brain tumors: a computer study. Med Phys. 1992;19:1277–84.
Zhang DG, Feygelman V, Moros EG, Latifi K, Zhang GG. Monte Carlo study of radiation dose enhancement by gadolinium in megavoltage and high dose rate radiotherapy. PLoS ONE. 2014;9: e109389.
De Stasio G, Rajesh D, Casalbore P, Daniels MJ, Erhardt RJ, Frazer BH, et al. Are gadolinium contrast agents suitable for gadolinium neutron capture therapy? Neurol Res. 2005;27:387–98.
Salt C, Lennox AJ, Takagaki M, Maguire JA, Hosmane NS. Boron and gadolinium neutron capture therapy. Russ Chem Bull. 2004;53:1871–88.
Martin RF, Feinendegen LE. The quest to exploit the auger effect in cancer radiotherapy-a reflective review. Int J Radiat Biol. 2016;92:617–32.
Yasui LS, Andorf C, Schneider L, Kroc T, Lennox A, Saroja KR. Gadolinium neutron capture in glioblastoma multiform cells. Int J Radiat Biol. 2008;84:1130–9.
Chan CC, Chen FH, Hsiao YY. Impact of hypoxia on relative biological effectiveness and oxygen enhancement ratio for a 62-MeV therapeutic proton beam. Cancers. 2021;13:2997.
Ito A, Nakano H, Kusano Y, Hirayama R, Furusawa Y, Murayama C, et al. Contribution of indirect action to radiation-induced mammalian cell inactivation: dependence on photon energy and heavy-ion LET. Radiat Res. 2006;165:703–12.
Nickoloff JA, Taylor L, Sharma N, Kato TA. Exploiting DNA repair pathways for tumor sensitization, mitigation of resistance, and normal tissue protection in radiotherapy. Cancer Drug Resist. 2021;4:244.
Snyder WS, Ford MR, Warner GG, Fisher HL. Estimates for absorbed fractions for monoenergetic photon sources uniformly distributed in various organs of a heterogeneous phantom. J Nucl Med Suppl. 1969;3:47.
White DR, Griffith RV, Wilson IJ. Report 46- Photon, electron, proton and neutron interaction data for body tissues. J ICRU. 1992;28:171.
Goorley T, Zamenhof R, Nikjoo H. Calculated DNA damage from gadolinium Auger electrons and relation to dose distributions in a head phantom. Int J Radiat Biol. 2004;80:933–40.
Avagyan R, Avetisyan R, Ivanyan V, Kerobyan I. GEANT4 simulations of a beam shaping assembly design and optimization for thermal/epithermal neutrons. Acta Phys Pol B. 2017;48:10.
Karger CP, Peschke P. RBE and related modeling in carbon-ion therapy. Phys Med Biol. 2017;63:01TR02.
Stewart R, Yu V, Georgakilas A, Koumenis C, Park J, Carlson D. Effects of radiation quality and oxygen on clustered DNA lesions and cell death. Radiat Res. 2011;176:587–602.
Semenenko VA, Stewart RD. A fast Monte Carlo algorithm to simulate the spectrum of DNA damages formed by ionizing radiation. Radiat Res. 2004;161:451–7.
Semenenko VA, Stewart RD. Fast Monte Carlo simulation of DNA damage formed by electrons and light ions. Phys Med Biol. 2006;51:1693–706.
Hsiao YY, Hung TH, Tu SJ, Tung CJ. Fast Monte Carlo simulation of DNA damage induction by Auger-electron emission. Int J Radiat Biol. 2014;90:392–400.
Kalospyros SA, Gika V, Nikitaki Z, Kalamara A, Kyriakou I, Emfietzoglou D, et al. Monte Carlo simulation-based calculations of complex DNA damage for incidents of environmental ionizing radiation exposure. Appl Sci. 2021;11:8985.
Stewart RD, Streitmatter SW, Argento DC, Kirkby C, Goorley JT, Moffitt G, et al. Rapid MCNP simulation of DNA double strand break (DSB) relative biological effectiveness (RBE) for photons, neutrons, and light ions. Phys Med Biol. 2015;60:8249–74.
Wang CC, Hsiao Y, Lee CC, Chao TC, Wang CC, Tung CJ. Monte Carlo simulations of therapeutic proton beams for relative biological effectiveness of double-strand break. Int J Radiat Biol. 2012;88:158–63.
Chatzipapas KP, Papadimitroulas P, Emfietzoglou D, Kalospyros SA, Hada M, Georgakilas AG, et al. Ionizing radiation and complex DNA damage: quantifying the radiobiological damage using Monte Carlo simulations. Cancers. 2020;12:799.
Ackerman NL, de la Fuente RL, Falzone N, Vallis KA, Bernal MA. Targeted alpha therapy with 212Pb or 225Ac: Change in RBE from daughter migration. Phys Med. 2018;51:91–8.
Duan D, Han Y, Tu Z, Guo H, Zhang Z, Shi Y, et al. Gadolinium neutron capture reaction-induced nucleodynamic therapy potentiates antitumor immunity. CCS Chem. 2023. https://doi.org/10.31635/ccschem.023.202202488.
Cerullo N, Bufalino D, Daquino G. Progress in the use of gadolinium for NCT. Appl Radiat Isot. 2009;67:S157–60.
Reniers B, Liu D, Rusch T, Verhaegen F. Calculation of relative biological effectiveness of a low-energy electronic brachytherapy source. Phys Med Biol. 2008;53:7125–35.
Shamsabadi R, Baghani HR, Azadegan B, Mowlavi AA. Impact of spherical applicator diameter on relative biologic effectiveness of low energy IORT X-rays: a hybrid Monte Carlo study. Phys Med. 2020;80:297–307.
Yasui L, Owens K. Necrosis is not induced by gadolinium neutron capture in glioblastoma multiforme cells. Int J Radiat Biol. 2012;88:980–90.
Humm JL, Howell RW, Rao DV. Dosimetry of Auger-electron-emitting radionuclides: report no. 3 of AAPM Nuclear Medicine Task Group No. 6. Med Phys. 1995;21:1901–15.
Matsumura A, Zhang T, Nakai K, Endo K, Kumada H, Yamamoto T, et al. Combination of boron and gadolinium compounds for neutron capture therapy. An in vitro study. J Exp Clin Cancer Res. 2005;24:93–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interest to declare that are relevant to the content of this article. No funds, grants or other support was received for conducting this study.
Ethical approval
No approval of research ethics committee was required to accomplish the goals of this study because of the review nature of conducted study.
Informed consent
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Shamsabadi, R., Baghani, H.R. Impact of gadolinium concentration and cell oxygen levels on radiobiological characteristics of gadolinium neutron capture therapy technique in brain tumor treatment. Radiol Phys Technol 17, 135–142 (2024). https://doi.org/10.1007/s12194-023-00758-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12194-023-00758-7