Abstract
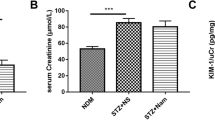
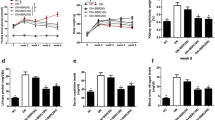
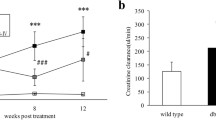
Tropisetron is a 5-HT3 receptor antagonist that exerts protective effect against DN. The aim of this study was to investigate the possible molecular mechanisms associated with the renoprotective effects of tropisetron in STZ-induced diabetic rats. Animals were subdivided into 5 equal groups; control, tropisetron, diabetes, tropisetron + diabetes, and glibenclamide + diabetes (n = 7). For induction of type 1 diabetes, a single injection of STZ (55 mg/kg, i.p.) was administered to the animals. Diabetic rats were treated with tropisetron (3 mg/kg) and glibenclamide (1 mg/kg) for 2 weeks. According to the conducted analysis, diabetes led to renal dysfunction (reduction in glomerular filtration rate and urine urea and creatinine as well as elevation in plasma urea and creatinine) and abnormalities in antioxidant defense system (reduction in TAC and elevation in MDA), compared with the control group, which was prevented by tropisetron treatment. Reverse transcription–quantitative polymerase chain reaction and western blotting analysis demonstrated that SIRT1 gene expression decreased while FOXO3a and NF-κB gene expression as well as phosphorylated FOXO3a/total FOXO3a protein ratios and claudin-1 protein level increased in the kidney of diabetic rats compared with the control group. Herein, the results of this research showed that tropisetron treatment reversed these changes. Besides, all these changes were comparable with those produced by glibenclamide as a positive control. Hence, tropisetron ameliorated renal damage due to diabetic nephropathy possibly by suppressing oxidative stress and alteration of SIRT1, FOXO3a, and claudin-1 levels.



Similar content being viewed by others
References
Accili D, Arden KC (2004) FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell 117:421–426. https://doi.org/10.1016/s0092-8674(04)00452-0
Akbar DH, Hagras MM, Amin HA, Khorshid OA (2013) Comparison between the effect of glibenclamide and captopril on experimentally induced diabetic nephropathy in rats. J Renin-Angiotensin-Aldosterone Syst 14:103–115. https://doi.org/10.1177/1470320312460881
Akhtar S, Siragy HM (2019) Pro-renin receptor suppresses mitochondrial biogenesis and function via AMPK/SIRT-1/PGC-1alpha pathway in diabetic kidney. PLoS One 14:e0225728. https://doi.org/10.1371/journal.pone.0225728
Alzahrani S, Zaitone SA, Said E, El-Sherbiny M, Ajwah S, Alsharif SY, Elsherbiny NM (2020) Protective effect of isoliquiritigenin on experimental diabetic nephropathy in rats: impact on Sirt-1/NFκB balance and NLRP3 expression. Int Immunopharmacol 87:106813. https://doi.org/10.1016/j.intimp.2020.106813
Amini M, Saboory E, Pourheydar B, Bagheri M, Naderi R (2020) Involvement of endocannabinoid system, inflammation and apoptosis in diabetes induced liver injury: role of 5-HT3 receptor antagonist. Int Immunopharmacol 79:106158
Aminzadeh A (2017) Protective effect of tropisetron on high glucose induced apoptosis and oxidative stress in PC12 cells: roles of JNK, P38 MAPKs, and mitochondria pathway. Metab Brain Dis 32:819–826
Asadi F, Razmi A, Dehpour AR, Shafiei M (2016) Tropisetron inhibits high glucose-induced calcineurin/NFAT hypertrophic pathway in H9c2 myocardial cells. J Pharm Pharmacol 68:485–493. https://doi.org/10.1111/jphp.12522
Barzegar-Fallah A, Alimoradi H, Mehrzadi S, Barzegar-Fallah N, Zendedel A, Abbasi A, Dehpour AR (2014) The neuroprotective effect of tropisetron on vincristine-induced neurotoxicity. Neurotoxicology 41:1–8. https://doi.org/10.1016/j.neuro.2013.12.002
Barzegar-Fallah A, Alimoradi H, Asadi F, Dehpour AR, Asgari M, Shafiei M (2015) Tropisetron ameliorates early diabetic nephropathy in streptozotocin-induced diabetic rats. Clin Exp Pharmacol Physiol 42:361–368. https://doi.org/10.1111/1440-1681.12373
Beckman KB, Ames BN (1998) The free radical theory of aging matures. Physiol Rev 78:547–581. https://doi.org/10.1152/physrev.1998.78.2.547
Brunet A et al (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science (New York, NY) 303:2011–2015. https://doi.org/10.1126/science.1094637
Chen L, Magliano DJ, Zimmet PZ (2011) The worldwide epidemiology of type 2 diabetes mellitus--present and future perspectives. Nat Rev Endocrinol 8:228–236. https://doi.org/10.1038/nrendo.2011.183
Dong YJ, Liu N, Xiao Z, Sun T, Wu SH, Sun WX, Xu ZG, Yuan H (2014) Renal protective effect of sirtuin 1. J Diabetes Res 2014:843786. https://doi.org/10.1155/2014/843786
Du YG, Wang LP, Qian JW, Zhang KN, Chai KF (2016) Panax notoginseng saponins protect kidney from diabetes by up-regulating silent information regulator 1 and activating antioxidant proteins in rats. Chin J Integrative Med 22:910–917. https://doi.org/10.1007/s11655-015-2446-1
Elmalí E, Altan N, Bukan N (2004) Effect of the sulphonylurea glibenclamide on liver and kidney antioxidant enzymes in streptozocin-induced diabetic rats. Drugs in R&D 5:203–208. https://doi.org/10.2165/00126839-200405040-00003
Elmarakby AA, Sullivan JC (2012) Relationship between oxidative stress and inflammatory cytokines in diabetic nephropathy. Cardiovasc Ther 30:49–59. https://doi.org/10.1111/j.1755-5922.2010.00218.x
Elsherbiny NM, Zaitone SA, Mohammad HMF, El-Sherbiny M (2018) Renoprotective effect of nifuroxazide in diabetes-induced nephropathy: impact on NFκB, oxidative stress, and apoptosis. Toxicol Mech Methods 28:467–473. https://doi.org/10.1080/15376516.2018.1459995
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186:407–421. https://doi.org/10.1016/0076-6879(90)86134-h
Funk JA, Schnellmann RG (2013) Accelerated recovery of renal mitochondrial and tubule homeostasis with SIRT1/PGC-1alpha activation following ischemia-reperfusion injury. Toxicol Appl Pharmacol 273:345–354. https://doi.org/10.1016/j.taap.2013.09.026
Gholizadeh-Ghaleh Aziz S, Naderi R, Mahmodian N (2019) Ameliorative effects of tropisetron on liver injury in streptozotocin-induced diabetic rats. Arch Physiol Biochem:1–6. https://doi.org/10.1080/13813455.2019.1640743
Giacco F, Brownlee M (2010) Oxidative stress and diabetic complications. Circulation Research 107:1058–1070 https://doi.org/10.1161/circresaha.110.223545
Gong Y, Sunq A, Roth RA, Hou J (2017) Inducible expression of claudin-1 in glomerular podocytes generates aberrant tight junctions and proteinuria through slit diaphragm destabilization. J Am Soc Nephrol 28:106–117. https://doi.org/10.1681/asn.2015121324
Gu J et al (2016) Olmesartan prevents microalbuminuria in db/db diabetic mice through inhibition of angiotensin II/p38/SIRT1-induced podocyte apoptosis. Kidney Blood Press Res 41:848–864. https://doi.org/10.1159/000452588
Haj-Mirzaian A et al (2016) Attenuation of oxidative and nitrosative stress in cortical area associates with antidepressant-like effects of tropisetron in male mice following social isolation stress. Brain Res Bull 124:150–163. https://doi.org/10.1016/j.brainresbull.2016.04.018
Hasegawa K et al (2008) Sirt1 protects against oxidative stress-induced renal tubular cell apoptosis by the bidirectional regulation of catalase expression. Biochem Biophys Res Commun 372:51–56. https://doi.org/10.1016/j.bbrc.2008.04.176
Hasegawa K, Wakino S, Simic P, Sakamaki Y, Minakuchi H, Fujimura K, Hosoya K, Komatsu M, Kaneko Y, Kanda T, Kubota E, Tokuyama H, Hayashi K, Guarente L, Itoh H (2013) Renal tubular Sirt1 attenuates diabetic albuminuria by epigenetically suppressing claudin-1 overexpression in podocytes. Nat Med 19:1496–1504
Hayden MS, Ghosh S (2012) NF-κB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev 26:203–234. https://doi.org/10.1101/gad.183434.111
Heimes K, Feistel B, Verspohl EJ (2009) Impact of the 5-HT 3 receptor channel system for insulin secretion and interaction of ginger extracts. Eur J Pharmacol 624:58–65
Hori YS, Kuno A, Hosoda R, Horio Y (2013) Regulation of FOXOs and p53 by SIRT1 modulators under oxidative stress. PLoS One 8:e73875. https://doi.org/10.1371/journal.pone.0073875
Huby AC, Rastaldi MP, Caron K, Smithies O, Dussaule JC, Chatziantoniou C (2009) Restoration of podocyte structure and improvement of chronic renal disease in transgenic mice overexpressing renin. PLoS One 4:e6721. https://doi.org/10.1371/journal.pone.0006721
Jaimes EA, Hua P, Tian RX, Raij L (2010) Human glomerular endothelium: interplay among glucose, free fatty acids, angiotensin II, and oxidative stress. Am J Physiol Ren Physiol 298:F125–F132. https://doi.org/10.1152/ajprenal.00248.2009
Kanwar YS et al (2008) Diabetic nephropathy: mechanisms of renal disease progression. Ex Biol Med (Maywood, NJ) 233:4–11. https://doi.org/10.3181/0705-mr-134
Kim DH et al (2011) SIRT1 activation by resveratrol ameliorates cisplatin-induced renal injury through deacetylation of p53. Am J Physiol Ren Physiol 301:F427–F435. https://doi.org/10.1152/ajprenal.00258.2010
Kiritoshi S et al (2003) Reactive oxygen species from mitochondria induce cyclooxygenase-2 gene expression in human mesangial cells: potential role in diabetic nephropathy. Diabetes 52:2570–2577. https://doi.org/10.2337/diabetes.52.10.2570
Kobayashi Y, Furukawa-Hibi Y, Chen C, Horio Y, Isobe K, Ikeda K, Motoyama N (2005) SIRT1 is critical regulator of FOXO-mediated transcription in response to oxidative stress. Int J Mol Med 16:237–243
Kops GJ et al (2002) Forkhead transcription factor FOXO3a protects quiescent cells from oxidative stress. Nature 419:316–321. https://doi.org/10.1038/nature01036
Kuhad A, Chopra K (2009) Attenuation of diabetic nephropathy by tocotrienol: involvement of NFkB signaling pathway. Life Sci 84:296–301. https://doi.org/10.1016/j.lfs.2008.12.014
Kumar GS, Kulkarni A, Khurana A, Kaur J, Tikoo K (2014) Selenium nanoparticles involve HSP-70 and SIRT1 in preventing the progression of type 1 diabetic nephropathy. Chem Biol Interact 223:125–133. https://doi.org/10.1016/j.cbi.2014.09.017
Kume S, Kitada M, Kanasaki K, Maegawa H, Koya D (2013) Anti-aging molecule, Sirt1: a novel therapeutic target for diabetic nephropathy. Arch Pharm Res 36:230–236
Langley E et al (2002) Human SIR2 deacetylates p53 and antagonizes PML/p53-induced cellular senescence. EMBO J 21:2383–2396. https://doi.org/10.1093/emboj/21.10.2383
Li Z, Zhang H, Chen Y, Fan L, Fang J (2012) Forkhead transcription factor FOXO3a protein activates nuclear factor kappaB through B-cell lymphoma/leukemia 10 (BCL10) protein and promotes tumor cell survival in serum deprivation. J Biol Chem 287:17737–17745. https://doi.org/10.1074/jbc.M111.291708
Liu R et al (2014) Role of transcription factor acetylation in diabetic kidney disease. Diabetes 63:2440–2453. https://doi.org/10.2337/db13-1810
Mirshafa A, Mohammadi H, Shokrzadeh M, Mohammadi E, Talebpour Amiri F, Shaki F (2020) Tropisetron protects against brain aging via attenuating oxidative stress, apoptosis and inflammation: the role of SIRT1 signaling. Life Sci 248:117452. https://doi.org/10.1016/j.lfs.2020.117452
Motta MC et al (2004) Mammalian SIRT1 represses forkhead transcription factors. Cell 116:551–563. https://doi.org/10.1016/s0092-8674(04)00126-6
Naderi R, Shirpoor A, Samadi M, Pourheydar B, Moslehi A (2020) Tropisetron improves pancreas function and increases insulin synthesis and secretion in the STZ-induced diabetic rats: involvement of UCP2/ZnT8 pathway. J Pharm Pharmacol 72:1082–1091. https://doi.org/10.1111/jphp.13278
Nakamura T, Ushiyama C, Shimada N, Hayashi K, Ebihara I, Koide H (2000) Comparative effects of pioglitazone, glibenclamide, and voglibose on urinary endothelin-1 and albumin excretion in diabetes patients. J Diabetes Complicat 14:250–254. https://doi.org/10.1016/s1056-8727(00)00124-0
Ohse T et al (2009) A new function for parietal epithelial cells: a second glomerular barrier. Am J Physiol -Renal Physiol 297:F1566–F1574
Papadimitriou A, Silva KC, Peixoto EB, Borges CM, Lopes de Faria JM, Lopes de Faria JB (2015) Theobromine increases NAD+/Sirt-1 activity and protects the kidney under diabetic conditions. Am J Physiol -Renal Physiol 308:F209–F225
Park HS et al (2016) Resveratrol increases AdipoR1 and AdipoR2 expression in type 2 diabetic nephropathy. J Transl Med 14:176. https://doi.org/10.1186/s12967-016-0922-9
Peluso I, Raguzzini A (2016) Salivary and urinary total antioxidant capacity as biomarkers of oxidative stress in humans. Pathol Res Int 2016:5480267–5480214. https://doi.org/10.1155/2016/5480267
Pieme CA et al (2017) Relationship between hyperglycemia, antioxidant capacity and some enzymatic and non-enzymatic antioxidants in African patients with type 2 diabetes. BMC Res Notes 10:141. https://doi.org/10.1186/s13104-017-2463-6
Rashidi M, Bazi A (2020) Tropisetron attenuates tumor growth and progression in an experimental model of mouse lung. Cancer 121:1610–1622. https://doi.org/10.1002/jcb.29395
Rincon-Choles H et al (2006) ZO-1 expression and phosphorylation in diabetic nephropathy. Diabetes 55:894–900. https://doi.org/10.2337/diabetes.55.04.06.db05-0355
Ritz E, Schmieder RE, Pollock CA (2010) Renal protection in diabetes: lessons from ONTARGET®. Cardiovasc Diabetol 9:60. https://doi.org/10.1186/1475-2840-9-60
Sengupta A, Molkentin JD, Paik J-H, DePinho RA, Yutzey KE (2011) FoxO transcription factors promote cardiomyocyte survival upon induction of oxidative stress. J Biol Chem 286:7468–7478
Sharma S, Kulkarni SK, Chopra K (2006) Curcumin, the active principle of turmeric (Curcuma longa), ameliorates diabetic nephropathy in rats. Clin Exp Pharmacol Physiol 33:940–945. https://doi.org/10.1111/j.1440-1681.2006.04468.x
Shi JX, Huang Q (2018) Glucagonlike peptide1 protects mouse podocytes against high glucoseinduced apoptosis, and suppresses reactive oxygen species production and proinflammatory cytokine secretion, through sirtuin 1 activation in vitro. Mol Med Rep 18:1789–1797. https://doi.org/10.3892/mmr.2018.9085
Singh RM, Howarth FC, Adeghate E, Bidasee K, Singh J, Waqar T (2018) Type 1 diabetes mellitus induces structural changes and molecular remodelling in the rat kidney. Mol Cell Biochem 449:9–25. https://doi.org/10.1007/s11010-018-3338-4
Song J, Yang B, Jia X, Li M, Tan W, Ma S, Shi X, Feng L (2018) Distinctive roles of Sirtuins on diabetes, protective or detrimental? Front Endocrinol 9:724. https://doi.org/10.3389/fendo.2018.00724
Tanaka J et al (2009) Foxo1 links hyperglycemia to LDL oxidation and endothelial nitric oxide synthase dysfunction in vascular endothelial cells. Diabetes 58:2344–2354. https://doi.org/10.2337/db09-0167
Tang Y et al (2018) Isoliquiritigenin attenuates LPS-induced AKI by suppression of inflammation involving NF-κB pathway. Am J Transl Res 10:4141–4151
Vikram A, Tripathi DN, Kumar A, Singh S (2014) Oxidative stress and inflammation in diabetic complications. Int J Endocrinol 2014:679754. https://doi.org/10.1155/2014/679754
Wang X, Hu S, Liu L (2017a) Phosphorylation and acetylation modifications of FOXO3a: independently or synergistically? Oncol Lett 13:2867–2872. https://doi.org/10.3892/ol.2017.5851
Wang X, Meng L, Zhao L, Wang Z, Liu H, Liu G, Guan G (2017b) Resveratrol ameliorates hyperglycemia-induced renal tubular oxidative stress damage via modulating the SIRT1/FOXO3a pathway. Diabetes Res Clin Pract 126:172–181. https://doi.org/10.1016/j.diabres.2016.12.005
Yaribeygi H, Mohammadi MT, Rezaee R, Sahebkar A (2018) Crocin improves renal function by declining Nox-4, IL-18, and p53 expression levels in an experimental model of diabetic nephropathy. J Cell Biochem 119:6080–6093. https://doi.org/10.1002/jcb.26806
Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW (2004) Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J 23:2369–2380. https://doi.org/10.1038/sj.emboj.7600244
Yun JM, Chien A, Jialal I, Devaraj S (2012) Resveratrol up-regulates SIRT1 and inhibits cellular oxidative stress in the diabetic milieu: mechanistic insights. J Nutr Biochem 23:699–705. https://doi.org/10.1016/j.jnutbio.2011.03.012
Zhang F et al (2015) Protective effects of icariin-mediated SIRT1/FOXO3 signaling pathway on intestinal ischemia/reperfusion-induced acute lung injury. Mol Med Rep 11:269–276. https://doi.org/10.3892/mmr.2014.2679
Funding
This study was supported by the Nephrology and Kidney Transplant Research Center, Urmia University of Medical Sciences, Urmia, Iran.
Author information
Authors and Affiliations
Contributions
Mahrokh Samadi: data curation, formal analysis, and writing (original draft). Shiva Gholizadeh -Ghaleh Aziz: data curation. Roya Naderi: conceptualization, data curation, formal analysis, methodology, project administration, supervision, validation, visualization, writing (original draft), writing (review), and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Samadi, M., Aziz, S.GG. & Naderi, R. The effect of tropisetron on oxidative stress, SIRT1, FOXO3a, and claudin-1 in the renal tissue of STZ-induced diabetic rats. Cell Stress and Chaperones 26, 217–227 (2021). https://doi.org/10.1007/s12192-020-01170-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-020-01170-5