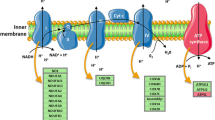
Abstract
Urbanization in low-income countries represents an important inflection point in the epidemiology of disease, with rural populations experiencing high rates of chronic and recurrent infections and urban populations displaying a profile of noncommunicable diseases. To investigate if urbanization alters the expression of genes encoding mitochondrial proteins, we queried gene microarray data from rural and urban populations living in Morocco (GSE17065). The R Bioconductor packages edgeR and limma were used to identify genes with different expression. The experimental design was modeled upon location and sex. Nuclear genes encoding mitochondrial proteins were identified from the MitoCarta2.0 database. Of the 1158 genes listed in the MitoCarta2.0 database, 847 genes (73%) were available for analysis in the Moroccan dataset. The urban-rural comparison with the greatest environmental differences showed that 76.5% of the MitoCarta2.0 genes were differentially expressed, with 97% of the genes having an increased expression in the urban area. Enrichment analysis revealed 367 significantly enriched pathways (adjusted p value < 0.05), with oxidative phosphorylation, insulin secretion and glucose regulations (adj.p values = 6.93E-16) being the top three. Four significantly perturbed KEGG disease pathways were associated with urbanization—three degenerative neurological diseases (Huntington’s, Alzheimer’s, and Parkinson’s diseases) and herpes simplex infection (false discover rate corrected p value (PGFdr) < 0.2). Mitochondrial RNA metabolic processing and translational elongation were the biological processes that had the greatest enrichment (enrichment ratios 14.0 and 14.8, respectively, FDR < 0.5). Our study links urbanization in Morocco with changes in the expression of the nuclear genes encoding mitochondrial proteins.


Similar content being viewed by others
References
Amegah AK, Agyei-Mensah S (2017) Urban air pollution in Sub-Saharan Africa: time for action. Environ Pollut 220(Pt A):738–743. https://doi.org/10.1016/j.envpol.2016.09.042
Amunts A, Brown A, Toots J, Scheres SHW, Ramakrishnan V (2015) Ribosome. The structure of the human mitochondrial ribosome. Science 348:95–98. https://doi.org/10.1126/science.aaa1193
Andersen GB, Tost J (2018) A summary of the biological processes, disease-associated changes, and clinical applications of DNA methylation. Methods Mol Biol 1708:3–30. https://doi.org/10.1007/978-1-4939-7481-8_1
Arnould T, Michel S, Renard, P (2015) Mitochondria Retrograde Signaling and the UPR mt: Where Are We in Mammals? Int J Mol Sci 16(8):18224–18251. https://doi.org/10.3390/ijms160818224
Ayeni FA, Biagi E, Rampelli S, Fiori J, Soverini M, Audu HJ, Turroni S (2018) Infant and adult gut microbiome and metabolome in rural Bassa and urban settlers from Nigeria. Cell Rep 23:3056–3067. https://doi.org/10.1016/j.celrep.2018.05.018
Bickler SW (2000) Non-communicable diseases: is their emergence in industrialized societies related to changes in neuroendocrine function? Med Hypotheses 54:825–828. https://doi.org/10.1054/mehy.1999.0959
Bickler SW, Lizardo RE, De Maio A (2015) The transition from a rural to an urban environment alters expression of the human Ebola virus receptor Neiman-pick C1: implications for the current epidemic in West Africa. Cell Stress Chaperones 20:203–206. https://doi.org/10.1007/s12192-014-0557-9
Bickler SW, Lizardo E, Cauvi DM, De Maio A (2016) The transition from a rural to an urban environment in Africa alters G protein-coupled receptor signaling. Med Hypotheses 95:49–53. https://doi.org/10.1016/j.mehy.2016.08.004
Bickler SW, Wang A, Amin S, Halbach J, Lizardo R, Cauvi DM, De Maio A (2018) Urbanization in sub-Saharan Africa: declining rates of chronic and recurrent infection and their possible role in the origins of non-communicable diseases. World J Surg 42:1617–1628. https://doi.org/10.1007/s00268-017-4389-5
Buchanan FG, DuBois RN (2006) Emerging roles of beta-arrestins. Cell Cycle 5:2060–2063. https://doi.org/10.4161/cc.5.18.3212
Caldwell JC (2001) Population health in transition. Bull World Health Organ 79:159–160 retrieved from https://www.ncbi.nlm.nih.gov/pubmed/11242823
Calvo SE, Clauser KR, Mootha VK (2016) MitoCarta2.0: an updated inventory of mammalian mitochondrial proteins. Nucleic Acids Res 44:D1251–D1257. https://doi.org/10.1093/nar/gkv1003
Chen J, Bardes EE, Aronow BJ, Jegga AG (2009) ToppGene suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res 37:W305–W311. https://doi.org/10.1093/nar/gkp427
Chen M, Zhang H, Liu W, Zhang W (2014) The global pattern of urbanization and economic growth: evidence from the last three decades. PLoS One 9:e103799. https://doi.org/10.1371/journal.pone.0103799
Chiarella AM, Lu D, Hathaway NA (2020) Epigenetic control of a local chromatin landscape. Int J Mol Sci 21:943. https://doi.org/10.3390/ijms21030943
Coker E, Kizito S (2018) A narrative review on the human health effects of ambient air pollution in Sub-Saharan Africa: an urgent need for health effects studies. Int J Environ Res Public Health:15. https://doi.org/10.3390/ijerph15030427
Cowen L, Ideker T., Raphael BJ, Sharan R (2017) Network propagation: a universal amplifier of genetic associations. Nat Rev Genet 18(9):551-562. https://doi.org/10.1038/nrg.2017.38
Dagher Z, Garcon G, Billet S, Gosset P, Ledoux F, Courcot D, Aboukais A, Shirali P (2006) Activation of different pathways of apoptosis by air pollution particulate matter (PM2.5) in human epithelial lung cells (L132) in culture. Toxicology 225:12–24. https://doi.org/10.1016/j.tox.2006.04.038
Davis S, Meltzer PS (2007) GEOquery: a bridge between the gene expression omnibus (GEO) and BioConductor. Bioinformatics 23:1846–1847. https://doi.org/10.1093/bioinformatics/btm254
DiMauro S, Schon EA (2003) Mitochondrial respiratory-chain diseases. N Engl J Med 348:2656–2668. https://doi.org/10.1056/NEJMra022567
Dunning MLA, Eldridge M (2015) illuminaHumanv3.db: Illumina HumanHT12v3 annotation data (chip illuminaHumanv3). R package version 1.26.0. Retrieved from http://bioconductor.org/.
Eckert S, Kohler S (2014) Urbanization and health in developing countries: a systematic review. World Health Popul 15:7–20 Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/24702762
Galea S, Vlahov D (2005) Urban health: evidence, challenges, and directions. Annu Rev Public Health 26:341–365. https://doi.org/10.1146/annurev.publhealth.26.021304.144708
Garaude J (2018) Reprogramming of mitochondrial metabolism by innate immunity. Curr Opin Immunol 56:17–23. https://doi.org/10.1016/j.coi.2018.09.010
Gentleman R, Huber W, Hahne F (2018) genefilter: methods for filtering genes from high-throughput experiments. R package version 1.64.0. Retrieved from http://bioconductor.org/. https://doi.org/10.18129/B9.bioc.genefilter
Gilkerson R (2018) A disturbance in the force: cellular stress sensing by the mitochondrial network. Antioxidants (Basel) 7. https://doi.org/10.3390/antiox7100126
Gong P, Liang S, Carlton EJ, Jiang Q, Wu J, Wang L, Remais JV (2012) Urbanisation and health in China. Lancet 379:843–852. https://doi.org/10.1016/S0140-6736(11)61878-3
Gualtieri M, Ovrevik J, Mollerup S, Asare N, Longhin E, Dahlman HJ, Camatini M, Holme JA (2011) Airborne urban particles (Milan winter-PM2.5) cause mitotic arrest and cell death: effects on DNA, mitochondria, AhR binding and spindle organization. Mutat Res 713:18–31. https://doi.org/10.1016/j.mrfmmm.2011.05.011
Guha M, Avadhani NG. (2013) Mitochondrial retrograde signaling at the crossroads of tumor bioenergetics, genetics and epigenetics. Mitochondrion 13(6):577–591. https://doi.org/10.1016/j.mito.2013.08.007
Hanzelmann S, Castelo R, Guinney J (2013) GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinformatics 14:7. https://doi.org/10.1186/1471-2105-14-7
Harpham T (2009) Urban health in developing countries: what do we know and where do we go? Health Place 15:107–116. https://doi.org/10.1016/j.healthplace.2008.03.004
Htet AS, Kjollesdal MK et al (2017) Lipid profiles and determinants of total cholesterol and hypercholesterolaemia among 25-74 year-old urban and rural citizens of the Yangon Region, Myanmar: a cross-sectional study. BMJ Open 7:e017465. https://doi.org/10.1136/bmjopen-2017-017465
Idaghdour Y, Storey JD, Jadallah SJ, Gibson G (2008) A genome-wide gene expression signature of environmental geography in leukocytes of Moroccan Amazighs. PLoS Genet 4:e1000052. https://doi.org/10.1371/journal.pgen.1000052
Idaghdour Y, Czika W, Shianna KV, Lee SH, Visscher PM, Martin HC, Miclaus K, Jadallah SJ, Goldstein DB, Wolfinger RD, Gibson G (2010) Geographical genomics of human leukocyte gene expression variation in southern Morocco. Nat Genet 42:62–67. https://doi.org/10.1038/ng.495
Jamison DT, Mosley WH (1991) Disease control priorities in developing countries: health policy responses to epidemiological change. Am J Public Health 81(1):15–22. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/1983911
Jha AR, Davenport ER, Gautam Y, Bhandari D, Tandukar S, Ng KM, Fragiadakis GK, Holmes S, Gautam GP, Leach J, Sherchand JB, Bustamante CD, Sonnenburg JL (2018) Gut microbiome transition across a lifestyle gradient in Himalaya. PLoS Biol 16:e2005396. https://doi.org/10.1371/journal.pbio.2005396
Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K (2017) KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res 45:D353–D361. https://doi.org/10.1093/nar/gkw1092
Kavishe B, Vanobberghen F, Katende D, Kapiga S, Munderi P, Baisley K, Biraro S, Mosha N, Mutungi G, Mghamba J, Hughes P, Smeeth L, Grosskurth H, Peck R (2019) Dyslipidemias and cardiovascular risk scores in urban and rural populations in north-western Tanzania and southern Uganda. PLoS One 14:e0223189. https://doi.org/10.1371/journal.pone.0223189
Lane N (2005) Power, sex, suicide: mitochondria and the meaning of life: Oxford University Press
Li Z, Hyseni X, Carter JD, Soukup JM, Dailey LA, Huang YC (2006) Pollutant particles enhanced H2O2 production from NAD(P)H oxidase and mitochondria in human pulmonary artery endothelial cells. Am J Physiol Cell Physiol 291:C357–C365. https://doi.org/10.1152/ajpcell.00365.2005
Liberzon A, Birger C, Thorvaldsdottir H, Ghandi M, Mesirov JP, Tamayo P (2015) The molecular signatures database (MSigDB) hallmark gene set collection. Cell Syst 1:417–425. https://doi.org/10.1016/j.cels.2015.12.004
Liew CC, Ma J, Tang HC, Zheng R, Dempsey AA (2006) The peripheral blood transcriptome dynamically reflects system wide biology: a potential diagnostic tool. J Lab Clin Med 147:126–132. https://doi.org/10.1016/j.lab.2005.10.005
Lokmer A, Aflalo S, Amougou N, Lafosse S, Froment A, Tabe FE, Poyet M, Groussin M, Said-Mohamed R, Ségurel L (2020) Response of the human gut and saliva microbiome to urbanization in Cameroon. Sci Rep 10:2856. https://doi.org/10.1038/s41598-020-59849-9
Lowell BB, Shulman GI (2005) Mitochondrial dysfunction and type 2 diabetes. Science 307:384–387. https://doi.org/10.1126/science.1104343
Monlun M, Hyernard C, Blanco P, Lartigue L, Faustin B (2017) Mitochondria as molecular platforms integrating multiple innate immune signalings. J Mol Biol 429:1–13. https://doi.org/10.1016/j.jmb.2016.10.028
Mortality, GBD, Causes of Death C (2016) Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388(10053):1459–1544. https://doi.org/10.1016/S0140-6736(16)31012-1
Murray CJ, Lopez AD (1997) Global mortality, disability, and the contribution of risk factors: Global Burden of Disease Study. Lancet 349(9063):1436–1442. https://doi.org/10.1016/S0140-6736(96)07495-8
Naquet P, Giessner C, Galland F (2016) Metabolic adaptation of tissues to stress releases metabolites influencing innate immunity. Curr Opin Immunol 38:30–38. https://doi.org/10.1016/j.coi.2015.10.005
Nath AP, Arafat D, Gibson G (2012) Using blood informative transcripts in geographical genomics: impact of lifestyle on gene expression in Fijians. Front Genet 3:243. https://doi.org/10.3389/fgene.2012.00243
Pearce SF, Rebelo-Guiomar P, D'Souza AR, Powell CA, Van Haute L, Minczuk M (2017) Regulation of mammalian mitochondrial gene expression: recent advances. Trends Biochem Sci 42:625–639. https://doi.org/10.1016/j.tibs.2017.02.003
Pigneret M, Roussel D, Hervant F (2018) Anaerobic end-products and mitochondrial parameters as physiological biomarkers to assess the impact of urban pollutants on a key bioturbator. Environ Sci Pollut Res Int 25:27225–27234. https://doi.org/10.1007/s11356-018-2756-x
Quiros PM., Mottis A, Auwerx J (2016) Mitonuclear communication in homeostasis and stress. Nat Rev Mol Cell Biol 17(4):213–226. https://doi.org/10.1038/nrm.2016.23
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47. https://doi.org/10.1093/nar/gkv007
Rosenthal SB, Len J, Webster M, Gary A, Birmingham A, Fisch KM (2018) Interactive network visualization in Jupyter notebooks: visJS2jupyter. Bioinformatics 34:126–128. https://doi.org/10.1093/bioinformatics/btx581
Sander LE, Garaude J (2018) The mitochondrial respiratory chain: a metabolic rheostat of innate immune cell-mediated antibacterial responses. Mitochondrion 41:28–36. https://doi.org/10.1016/j.mito.2017.10.008
Satterthwaite D, McGranahan G, Tacoli C (2010) Urbanization and its implications for food and farming. Philos Trans R Soc Lond Ser B Biol Sci 365:2809–2820. https://doi.org/10.1098/rstb.2010.0136
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Smith AC, Robinson AJ (2016) MitoMiner v3.1, an update on the mitochondrial proteomics database. Nucleic Acids Res 44:D1258–D1261. https://doi.org/10.1093/nar/gkv1001
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos A, Tsafou KP, Kuhn M, Bork P, Jensen LJ, von Mering C (2015) STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43(Database issue):D447–D452. https://doi.org/10.1093/nar/gku1003
Tarca AL KP, Draghici S (2018) SPIA: Signaling Pathway Impact Analysis (SPIA) using combined evidence of pathway over-representation and unusual signaling perturbations. R package version 2.34.0. Retrieved from https://www.bioconductor.org/
United Nations (2019) World Urbanization Prospects: The 2018 Revision. Retrieved from https://population.un.org/wup/Publications/Files/WUP2018-Report.pdf
Vasunilashorn S, Crimmins EM, Kim JK, Winking J, Gurven M, Kaplan H, Finch CE (2010) Blood lipids, infection, and inflammatory markers in the Tsimane of Bolivia. Am J Hum Biol 22:731–740. https://doi.org/10.1002/ajhb.21074
Wallace DC (2005) Mitochondria and cancer: Warburg addressed. Cold Spring Harb Symp Quant Biol 70:363–374. https://doi.org/10.1101/sqb.2005.70.035
Wang J, Vasaikar S, Shi Z, Greer M, Zhang B (2017) WebGestalt 2017: a more comprehensive, powerful, flexible and interactive gene set enrichment analysis toolkit. Nucleic Acids Res 45:W130–W137. https://doi.org/10.1093/nar/gkx356
Funding
This study was supported by the Fogarty International Center, National Institutes of Health (NIH), grant number R25TW011216 and the University of California San Diego (UCSD), Clinical and Translational Research Institute (CTRI) grant number UL1TR001442. The authors thank Barbara Rho for her excellent editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Table S1
Differentially expressed MitoCarta2.0 genes in the Agadir-Boutrouch (urban-rural) geographic comparison. (CSV 119 kb).
Table S2
Gene Set Enrichment Analysis (GSEA) of differentially expressed MitoCarta2.0 genes in the Agadir-Boutrouch (urban-rural) geographic comparison. (CSV 37 kb).
Table S3
Signaling Pathway Impact Analysis (SPIA) of differentially expressed MitoCarta2.0 genes in the Agadir-Boutrouch (urban-rural) geographic comparison. (CSV 12 kb).
Rights and permissions
About this article
Cite this article
Bickler, S.W., Prieto, J.M., Cauvi, D.M. et al. Differential expression of nuclear genes encoding mitochondrial proteins from urban and rural populations in Morocco. Cell Stress and Chaperones 25, 847–856 (2020). https://doi.org/10.1007/s12192-020-01108-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-020-01108-x