Abstract
Background
We investigated whether a decrease in the serum zinc level (SZL) among patients with sepsis admitted to the intensive care unit (ICU) was related to sepsis-induced coagulopathy.
Methods
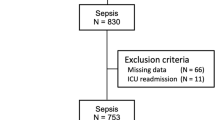
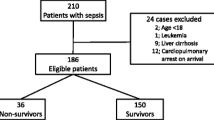
All patients (≥20 years) with a diagnosis of sepsis defined by Sepsis-3 criteria, presenting to the ICU between June 2016 and July 2017, were enrolled. Demographic characteristics and the Sequential Organ Failure Assessment (SOFA) and Japanese Association of Acute Medicine (JAAM) disseminated intravascular coagulation (DIC) scores were recorded. Blood samples were collected upon admission and analyzed for SZL.
Results
One hundred patients with sepsis (median age, 70 years) were enrolled. Patients with SOFA scores ≥8 had a significantly lower SZL compared to those with SOFA scores <8 (p < 0.001). The SZL in the DIC group (JAAM DIC score ≥4) was significantly lower than that in the non-DIC group (JAAM DIC score <4) (p < 0.001). Analysis of receiver operating characteristic (ROC) curves for prediction of sepsis-induced DIC based on SZL in patients with sepsis showed a cut-off value of 25 µg/dL for zinc level and a sensitivity of 63% and a specificity of 72% with AUC of 0.7 (p = 0.0065).
Conclusion
We observed that SZL reflects organ failure, particularly coagulopathy, in patients with sepsis.



Similar content being viewed by others
Availability of data and materials
The dataset generated and/or analyzed during the current study is not publicly available because of patient-related confidentiality, but is available from the corresponding author upon reasonable request.
Abbreviations
- APACHE:
-
Acute physiology and chronic health evaluation
- AUC:
-
area under the curve
- BMI:
-
Body mass index
- DIC:
-
Disseminated intravascular coagulation
- ICU:
-
Intensive care unit
- JAAM:
-
Japanese association of acute medicine
- MODS:
-
Multiple-organ dysfunction syndrome
- PAI:
-
Plasminogen activator inhibitor
- PC:
-
Protein C
- PT:
-
Prothrombin time
- ROC:
-
Receiver operating characteristic
- SOFA:
-
Sequential organ failure assessment
- SIRS:
-
Systemic inflammatory response syndrome
- TNF-α:
-
Tumor necrotic factor alpha
- WBC:
-
White blood cell
References
Walkey AJ, Lagu T, Lindenauer PK. Trends in sepsis and infection sources in the United States. A population-based study. Ann Am Thorac Soc. 2015;12:216–20.
Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369:840–51.
Russell JA. Management of sepsis. N Engl J Med. 2006;355:1699–713.
Lever A, Mackenzie I. Sepsis: definition, epidemiology, and diagnosis. BMJ (Clinical research ed). 2007;335:879–83.
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–10.
Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med. 2003;348:138–50.
Cohen J, Vincent JL, Adhikari NK, Machado FR, Angus DC, Calandra T, et al. Sepsis: a roadmap for future research. Lancet Infect Dis. 2015;15:581–614.
Levi M, van der Poll T. Coagulation and sepsis. Thromb Res. 2017;149:38–44.
Levi M, van der Poll T, Buller HR. Bidirectional relation between inflammation and coagulation. Circulation. 2004;109:2698–704.
Alker W, Haase H. Zinc and sepsis. Nutrients. 2018;10:976.
Mertens K, Lowes DA, Webster NR, Talib J, Hall L, Davies MJ, et al. Low zinc and selenium concentrations in sepsis are associated with oxidative damage and inflammation. Br J Anaesth. 2015;114:990–9.
Iskander KN, Osuchowski MF, Stearns-Kurosawa DJ, Kurosawa S, Stepien D, Valentine C, et al. Sepsis: multiple abnormalities, heterogeneous responses, and evolving understanding. Physiol Rev. 2013;93:1247–88.
Davies MJ. The oxidative environment and protein damage. Biochim Biophys Acta. 2005;1703:93–109.
Prasad AS, Bao B, Beck FW, Kucuk O, Sarkar FH. Antioxidant effect of zinc in humans. Free Radic Biol Med. 2004;37:1182–90.
Koekkoek WA, van Zanten AR. Antioxidant vitamins and trace elements in critical illness. Nutr Clin Pract. 2016;31:457–74.
Fischer Walker C, Black RE. Zinc and the risk for infectious disease. Annu Rev Nutr. 2004;24:255–75.
Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14:353–7.
Hamon R, Homan CC, Tran HB, Mukaro VR, Lester SE, Roscioli E, et al. Zinc and zinc transporters in macrophages and their roles in efferocytosis in COPD. PLoS One. 2014;9:e110056.
Fernandes N, Mosnier LO, Tonnu L, Heeb MJ. Zn(2)(+)-containing protein S inhibits extrinsic factor X-activating complex independently of tissue factor pathway inhibitor. J Thromb Haemost. 2010;8:1976–85.
Henderson SJ, Stafford AR, Leslie BA, Kim PY, Vaezzadeh N, Ni R, et al. Zinc delays clot lysis by attenuating plasminogen activation and plasmin-mediated fibrin degradation. Thromb Haemost. 2015;113:1278–88.
Heeb MJ, Prashun D, Griffin JH, Bouma BN. Plasma protein S contains zinc essential for efficient activated protein C-independent anticoagulant activity and binding to factor Xa, but not for efficient binding to tissue factor pathway inhibitor. FASEB J. 2009;23:2244–53.
Hopmeier P, Halbmayer M, Fischer M, Marx G. Zinc modulates thrombin adsorption to fibrin. Thromb Res. 1990;58:293–301.
Watson BR, White NA, Taylor KA, Howes JM, Malcor JD, Bihan D, et al. Zinc is a transmembrane agonist that induces platelet activation in a tyrosine phosphorylation-dependent manner. Metallomics. 2016;8:91–100.
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315:801–10.
Gando S, Iba T, Eguchi Y, Ohtomo Y, Okamoto K, Koseki K, et al. A multicenter, prospective validation of disseminated intravascular coagulation diagnostic criteria for critically ill patients: comparing current criteria. Crit Care Med. 2006;34:625–31.
Funk DJ, Parrillo JE, Kumar A. Sepsis and septic shock: a history. Crit Care Clin. 2009;25:83–101.
Tracey KJ, Beutler B, Lowry SF, Merryweather J, Wolpe S, Milsark IW, et al. Shock and tissue injury induced by recombinant human cachectin. Science. 1986;234:470–4.
Kaukonen KM, Bailey M, Pilcher D, Cooper DJ, Bellomo R. Systemic inflammatory response syndrome criteria in defining severe sepsis. N Engl J Med. 2015;372:1629–38.
Boermeester MA, van Leeuwen PA, Coyle SM, Wolbink GJ, Hack CE, Lowry SF. Interleukin-1 blockade attenuates mediator release and dysregulation of the hemostatic mechanism during human sepsis. Arch Surg. 1995;130:739–48.
Levi M, van der Poll T. Inflammation and coagulation. Crit Care Med. 2010;38:S26-34.
Kushner I. The phenomenon of the acute phase response. Ann N Y Acad Sci. 1982;389:39–48.
Brown KH. Effect of infections on plasma zinc concentration and implications for zinc status assessment in low-income countries. Am J Clin Nutr. 1998;68:425–9.
Hasanzadeh Kiabi F, Alipour A, Darvishi-Khezri H, Aliasgharian A, Emami Zeydi A. Zinc supplementation in adult mechanically ventilated trauma patients is associated with decreased occurrence of ventilator-associated pneumonia: a secondary analysis of a prospective, observational study. Indian J Crit Care Med. 2017;21:34–9.
Wang L, Song Y. Efficacy of zinc given as an adjunct to the treatment of severe pneumonia: a meta-analysis of randomized, double-blind and placebo-controlled trials. Clin Respir J. 2018;12:857–64.
Mohammad MK, Zhou Z, Cave M, Barve A, McClain CJ. Zinc and liver disease. Nutr Clin Prac. 2012;27:8–20.
Fraker PJ, King LE. A distinct role for apoptosis in the changes in lymphopoiesis and myelopoiesis created by deficiencies in zinc. FASEB J. 2001;15:2572–8.
Mayer LS, Uciechowski P, Meyer S, Schwerdtle T, Rink L, Haase H. Differential impact of zinc deficiency on phagocytosis, oxidative burst, and production of pro-inflammatory cytokines by human monocytes. Metallomics. 2014;6:1288–95.
Moshage H. Cytokines and the hepatic acute phase response. J Pathol. 1997;181:257–66.
Gando S, Levi M, Toh CH. Disseminated intravascular coagulation. Nat Rev Dis Primers. 2016;2:16037.
Iba T, Levy JH. Inflammation and thrombosis: Roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis. J Thromb Haemost. 2018;16:231–41.
Iba T, Levy JH, Thachil J, Wada H, Levi M, Scientific and Standardization Committee on DIC of the International Society on Thrombosis and Haemostasis. The progression from coagulopathy to disseminated intravascular coagulation in representative underlying diseases. Thromb Res. 2019;179:11–4.
Levy JH, Sniecinski RM, Welsby IJ, Levi M. Antithrombin: anti-inflammatory properties and clinical applications. Thromb Haemost. 2016;115:712–28.
Bajaj SP, Schmidt AE, Agah S, Bajaj MS, Padmanabhan K. High resolution structures of p-aminobenzamidine- and benzamidine-VIIa/soluble tissue factor: unpredicted conformation of the 192–193 peptide bond and mapping of Ca2+, Mg2+, Na+, and Zn2+ sites in factor VIIa. J Biol Chem. 2006;281:24873–88.
Esmon CT. The protein C pathway. Chest. 2003;124:26s–32s.
Burstyn-Cohen T, Heeb MJ, Lemke G. Lack of protein S in mice causes embryonic lethal coagulopathy and vascular dysgenesis. J Clin Invest. 2009;119:2942–53.
Li B, Cui W, Tan Y, Luo P, Chen Q, Zhang C, et al. Zinc is essential for the transcription function of Nrf2 in human renal tubule cells in vitro and mouse kidney in vivo under the diabetic condition. J Cell Mol Med. 2014;18:895–06.
Prasad AS, Bao B, Beck FW, Sarkar FH. Zinc activates NF-kappaB in HUT-78 cells. J Lab Clin Med. 2001;138:250–6.
Liuzzi JP, Lichten LA, Rivera S, Blanchard RK, Aydemir TB, Knutson MD, et al. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to the hypozincemia of the acute-phase response. Proc Natl Acad Sci U S A. 2005;102:6843–8.
Bahtouee M, Eghbali SS, Maleki N, Rastgou V, Motamed N. Acute physiology and chronic health evaluation II score for the assessment of mortality prediction in the intensive care unit: a single-centre study from Iran. Nurs Crit Care. 2019;24:375–80.
Cander B, Dundar ZD, Gul M, Girisgin S. Prognostic value of serum zinc levels in critically ill patients. J Crit Care. 2011;26:42–6.
Fukada T, Yamasaki S, Nishida K, Murakami M, Hirano T. Zinc homeostasis and signaling in health and diseases: Zinc signaling. J Biol Inorg Chem. 2011;16:1123–34.
Sugiura T, Nakamura H. Metallothionein in platelets. Int Arch Allergy Immunol. 1994;103:341–8.
Taylor KA, Pugh N. The contribution of zinc to platelet behaviour during haemostasis and thrombosis. Metallomics. 2016;8:144–55.
Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991;43:109–42.
Jarosz M, Olbert M, Wyszogrodzka G, Młyniec K, Librowski T. Antioxidant and anti-inflammatory effects of zinc Zinc-dependent NF-κB signaling. Inflammopharmacology. 2017;25:11–24.
Acknowledgements
We appreciate the assistance of the nursing staff at the Emergency and Critical Care Center at Fukuoka University Hospital, Fukuoka, Japan. We thank Arshad Makhdum, PhD, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Institutional Research Ethics Committee of Fukuoka University Hospital (No. 18-3-02). It complied with the principles of Helsinki Declaration.
Consent to participate
Informed consent was waived based on the study’s retrospective, observational design, which preserves the confidentiality of personal information.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Irie, Y., Hoshino, K., Kawano, Y. et al. Relationship between serum zinc level and sepsis-induced coagulopathy. Int J Hematol 115, 87–95 (2022). https://doi.org/10.1007/s12185-021-03225-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03225-4