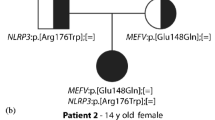
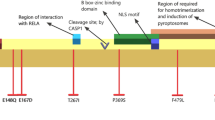
Abstract
Familial Mediterranean fever is an autosomal recessive disease occurring in populations originating from the Mediterranean basin. This autoinflammatory syndrome is caused by mutations in the Mediterranean FeVer (MEFV) gene. MEFV encodes a 781 amino acid protein known as pyrin. Pyrin is an important modulator of apoptosis, inflammation, and cytokine processing. In more recent pilot studies, inherited variant analysis of the MEFV gene in patients with hematologic neoplasm showed an unexpectedly high frequency of these variants in the gene. Here, we summarize the current state of knowledge of the relationship between inherited variants in the MEFV gene and hematologic neoplasms. Although no single underlying defect could be targeted in all hematologic neoplasms, it will be important to fully exploit the mechanisms underlying the neoplasm promoting role of inherited variants in MEFV. However, it is unclear how inherited variants in the MEFV gene are associated with tumor susceptibility or promotion in hematologic neoplasms. Further investigations are needed to determine the actual role of the MEFV gene in pathogenesis of these neoplasms.
Similar content being viewed by others
References
Musabak U, Sengul A, Oktenli C, Pay S, Yesilova Z, Kenar L, et al. Does immune activation continue during an attack-free period in familial Mediterranean fever? Clin Exp Immunol. 2004;138:526–33.
Terekeci HM, Oktenli C, Ozgurtas T, Nalbant S, Top C, Celik S, et al. Increased asymmetric dimethylarginine levels in young men with familial Mediterranean fever (FMF): is it early evidence of interaction between inflammation and endothelial dysfunction in FMF? J Rheum. 2008;35:2024–9.
Terekeci HM, Ulusoy ER, Kucukarslan NM, Nalbant S, Oktenli C. Familial Mediterranean fever attacks do not alter functional and morphologic tissue Doppler echocardiographic parameters. Rheum Int. 2008;28:1239–43.
Touitou I. The spectrum of familial Mediterranean fever (FMF) mutations. Eur J Hum Genet. 2001;9:473–83.
Centola M, Aksentijevich I, Kastner DL. The hereditary periodic fever syndromes: molecular analysis of a new family of inflammatory diseases. Hum Mol Genet. 1998;7:1581–8.
Centola M, Wood G, Frucht DM, Galon J, Arringer M, Farrell C, et al. The gene for familial Mediterranean fever, MEFV, is expressed in early leukocyte development and is regulated in response to inflammatory mediators. Blood. 2000;95:3223–31.
Henry J, Mather IH, McDermott MF, Pontarotti P. B30.2-like domain proteins: update and new insights into a rapidly expanding family of proteins. Mol Biol Evol. 1998;15:1696–705.
Borden KL. RING fingers and B-boxes: zinc-binding protein–protein interaction domains. Biochem Cell Biol. 1998;76:351–8.
The International FMF Consortium. Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. Cell. 1997;90:797–807.
Chae JJ, Aksentijevich I. Kastner DL. Advances in the understanding of familial Mediterranean fever and possibilities for targeted therapy. Br J Haematol. 2009;146:467–78.
The French FMF Consortium. A candidate gene for familial Mediterranean fever. Nature Genet. 1997;17:25–31.
Koc B, Oktenli C, Bulucu F, Karadurmus N, Sanisoglu SY, Gul D. The rate of pyrin mutations in critically ill patients with systemic inflammatory response syndrome and sepsis: a pilot study. J Rheumatol. 2007;34:2070–5.
Tidow N, Chen X, Muller C, Kawano S, Gombart AF, Fischel-Ghodsian N, et al. Hematopoietic-specific expression of MEFV, the gene mutated in familial Mediterranean fever, and subcellular localization of its corresponding protein, pyrin. Blood. 2000;95:1451–5.
Kotone-Miyahara Y, Takaori-Kondo A, Fukunaga K, Goto M, Hayashino Y, Miki M, et al. E148Q/M694I mutation in 3 Japanese patients with familial Mediterranean fever. Int J Hematol. 2004;79:235–7.
Miyoshi T, Yamashita K, Ohno T, Izumi T, Takaori-Kondo A, Sasada M, et al. Familial Mediterranean fever gene as a possible modifier of sweet syndrome with chronic myelogenous leukemia. Acta Haematol. 2008;120:57–62.
Sasaki K, Tahara T, Mitani K. Presentation of familial Mediterranean fever in a heterozygous MEFV mutation triggered by immunosuppressive therapy for myelodysplastic syndrome. Int J Hematol. 2009;90:91–3.
Oktenli C, Sayan O, Celik S, Erikci AA, Tunca Y, Terekeci HM, et al. High frequency of MEFV gene mutations in patients with myeloid neoplasm. Int J Hematol. 2010;91:758–61.
Celik S, Erikci AA, Tunca Y, Sayan O, Terekeci HM, Umur EE, et al. The rate of MEFV gene mutations in hematolymphoid neoplasms. Int J Immunogenet. 2010;37:387–91.
Yilmaz E, Ozen S, Balci B, Duzova A, Topaloglu R, Besbas N, et al. Mutation frequency of familial Mediterranean fever and evidence for a high carrier rate in the Turkish population. Eur J Hum Genet. 2001;9:553–5.
Sayan O, Kilicaslan E, Celik S, Tangi F, Erikci AA, Ipcioglu O, et al. High frequency of inherited variants in the MEFV gene in acute lymphocytic leukemia. Indian J Hematol Blood Transfus. 2011;27:164–8.
Celik S, Oktenli C, Kilicaslan E, Tangi F, Sayan O, Ozari HO, et al. Frequency of inherited variants in the MEFV gene in myelodysplastic syndrome and acute myeloid leukemia. Int J Hematol. 2012;95:285–90.
Medlej-Hashim M, Serre JL, Corbani S, Saab O, Jalkh N, Delague V, et al. Familial Mediterranean fever (FMF) in Lebanon and Jordan: a population genetics study and report of three novel mutations. Eur J Med Genet. 2004;48:412.
Chen F, Beezhold K, Castranova V. Tumor promoting or tumor suppressing of NF-κB, a matter of cell context dependency. Int Rev Immunol. 2008;27:183–204.
Seshadri S, Duncan MD, Hart JM, Gavrilin MA, Wewers MD. Pyrin levels in human monocytes and monocyte-derived macrophages regulate IL-1β processing and release. J Immunol. 2007;179:1274–81.
Bertin J, DiStefano PS. The PYRIN domain: a novel motif found in apoptosis and inflammation proteins. Cell Death Differ. 2000;7:1273–4.
Martinon F, Hofmann K, Tschopp J. The pyrin domain: a possible member of the death domainfold family implicated in apoptosis and inflammation. Curr Biol. 2001;11:R118–20.
Schaner P, Richards N, Wadhwa A, Aksentijevich I, Kastner DL, Tucker P, et al. Episodic evolution of pyrin in primates: human mutations recapitulate ancestral amino acid states. Nature Genet. 2001;27:318.
Srinivasula SM, Poyet JL, Razmara M, Datta P, Zhang Z, Alnemri ES. The PYRIN-CARD protein ASC is an activating adaptor for caspase-1. J Biol Chem. 2002;277:21119–22.
Fairbrother WJ, Gordon NC, Humke EW, O’Rourke KM, Starovasnik MA, Yin JP, et al. The PYRIN domain: a member of the death domain-fold superfamily. Protein Sci. 2001;10:1911–8.
Liepinsh E, Barbals R, Dahl E, Sharipo A, Staub E, Otting G. The death-domain fold of the ASC PYRIN domain, presenting a basis for PYRIN/PYRIN recognition. J Mol Biol. 2003;332:1155–63.
Richards N, Schaner P, Diaz A, Stuckey J, Shelden E, Wadhwa A, et al. Interaction between pyrin and the apoptotic speck protein (ASC) modulates ASC-induced apoptosis. J Biol Chem. 2001;276:39320–9.
McConnell BB. Vertino PM TMS1/ASC: The cancer connection. Apoptosis. 2004;9:5–18.
Chae JJ, Wood G, Masters SL, Richard K, Park G, Smith BJ, et al. The B30.2 domain of pyrin, the familial Mediterranean fever protein, interacts directly with caspase-1 to modulate IL-1beta production. Proc Natl Acad Sci USA. 2006;103:9982–7.
Masters SL, Yao S, Willson TA, Zhang JG, Palmer KR, Smith BJ, et al. The SPRY domain of SSB-2 adopts a novel fold that presents conserved Par-4-binding residues. Nat Struct Mol Biol. 2006;13:77–84.
Papin S, Cuenin S, Agostini L, Martinon F, Werner S, Beer HD, et al. The SPRY domain of Pyrin, mutated in familial Mediterranean fever patients, interacts with inflammasome components and inhibits proIL-1β processing. Cell Death Differ. 2007;14:1457–66.
Chae JJ, Komarow HD, Cheng J, Wood G, Raben N, Liu PP, et al. Targeted disruption of pyrin, the FMF protein, causes heightened sensitivity to endotoxin and a defect in macrophage apoptosis. Mol Cell. 2003;11:591–604.
Cerretti DP, Kozlosky CJ, Mosley B, Nelson N, Van Ness K, Greenstreet TA, et al. Molecular cloning of the interleukin-1β converting enzyme. Science. 1992;256:97–100.
Fernandes-Alnemri T, Wu J, Yu JW, Datta P, Miller B, Jankowski W, et al. The pyroptosome: a supramolecular assembly of ASC dimers mediating inflammatory cell death via caspase-1 activation. Cell Death Differ. 2007;14:1590–604.
Kobayashi S. Hereditary periodic fever syndromes: autoinflammatory diseases. Int Med. 2005;44:694–5.
Matsushita K, Takeoka M, Sagara J, Itano N, Kurose Y, Nakamura A, et al. A splice variant of ASC regulates IL-1β release and aggregates differently from intact ASC. Mediat Inflamm. 2009;287387:1.
Thornberry NA, Bull HG, Calaycay JR, Chapman KT, Howard AD, Kostura MJ, et al. A novel heterodimeric cysteine protease is required for interleukin-1β processing in monocytes. Nature. 1992;356:768–74.
Stehlik C, Reed JC. The PYRIN connection: novel players in innate immunity and inflammation. J Exp Med. 2004;200:551–8.
Chae JJ, Wood G, Richard K, Jaffe H, Colburn N, Masters SL, et al. The familial Mediterranean fever protein, pyrin, is cleaved by caspase-1 and activates NF-κB through its N-terminal fragment. Blood. 2008;112:1794–803.
Stehlik C, Lee SH, Dorfleutner A, Stassinopoulos A, Sagara J, Reed JC. Apoptosis-associated speck-like protein containing a caspase recruitment domain is a regulator of procaspase-1 activation. J Immunol. 2003;171:6154–63.
Stehlik C, Fiorentino L, Dorfleutner A, Bruey JM, Ariza EM, Sagara J, et al. The PAAD/PYRIN-family protein ASC is a dual regulator of a conserved step in nuclear factor kappaB activation pathways. J Exp Med. 2002;196:1605–15.
Masumoto J, Taniguchi S, Ayukawa K, Sarvotham H, Kishino T, Niikawa N, et al. ASC, a novel 22-kDa protein, aggregates during apoptosis of human promyelocytic leukemia HL-60 cells. J Biol Chem. 1999;274:33835–8.
Gumucio DL, Diaz A, Schaner P, Richards N, Babcock C, Schaller M, et al. Fire and ICE: the role of pyrin domain-containing proteins in inflammation and apoptosis. Clin Exp Rheumatol. 2000;20:S45–53.
Conway KE, McConnell BB, Bowring CE, Donald CD, Warren ST, Vertino PM. TMS1, a novel proapoptotic caspase recruitment domain protein, is a target of methylation-induced gene silencing in human breast cancers. Cancer Res. 2000;60:6236–42.
Martinon F, Burns K, Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol Cell. 2002;10:417–26.
Manji GA, Wang L, Geddes BJ, Brown M, Merriam S, Al-Garawi A, et al. PYPAF1, a PYRIN-containing Apaf1-like protein that assembles with ASC and regulates activation of NF-kappa B. J Biol Chem. 2002;277:11570–5.
Wang L, Manji GA, Grenier JM, Al-Garawi A, Merriam S, Lora JM, et al. PYPAF7, a novel PYRIN-containing Apaf1-like protein that regulates activation of NF-kappa B and caspase-1-dependent cytokine processing. J Biol Chem. 2002;277:29874–80.
Mariathasan S, Newton K, Monack DM, Vucic D, French DM, Lee WP, et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature. 2004;430:213–8.
Yu JW, Wu J, Zhang Z, Datta P, Ibrahimi I, Taniguchi S, et al. Cryopyrin and pyrin activate caspase-1, but not NF-κB, via ASC oligomerization. Cell Death Differ. 2006;13:236–49.
McConnell BB, Vertino PM. Activation of a caspase-9-mediated apoptotic pathway by subcellular redistribution of the novel caspase recruitment domain protein TMS1. Cancer Res. 2000;60:6243–7.
Keutgens A, Robert I, Viatour P, Chariot A. Deregulated NF-κB activity in haematological malignancies. Biochem Pharmacol. 2006;72:1069–80.
Siebenlist U, Brown K, Claudio E. Control of lymphocyte development by nuclear factor-κB. Nature Rev Immunol. 2005;5:435–45.
Grossmann M, Metcalf D, Merryfull J, Beg A, Baltimore D, Gerondakis S. The combined absence of the transcription factors Rel and RelA leads to multiple hemopoietic cell defects. Proc Natl Acad Sci. 1999;96:11848–53.
Voll RE, Jimi E, Phillips RJ, Barber DF, Rincon M, Hayday AC, et al. NF-kappa B activation by the pre-T cell receptor serves as a selective survival signal in T lymphocyte development. Immunity. 2000;13:677–89.
Feng B, Cheng S, Pear WS, Liou HC. NF-κB inhibitor blocks B cell development at two checkpoints. Med Immunol. 2004;3:1.
Pasparakis M, Schmidt-Supprian M, Rajewsky K. IkappaB kinase signaling is essential for maintenance of mature B cells. J Exp Med. 2002;196:743–52.
Blonska M, Lin X. CARMA1-mediated NF-κB and JNK activation in lymphocytes. Immunol Rev. 2009;228:199–211.
Packham G. The role of NF-κB in lymphoid malignancies. Br J Haematol. 2008;143:3–15.
Gilmore TD. Introduction to NF-kappaB: players, pathways, perspectives. Oncogene. 2006;25:6680–4.
Lucas PC, McAllister-Lucas LM, Nunez G. NF-κB signaling in lymphocytes: a new cast of Characters. J Cell Sci. 2004;117:31–9.
Bassères DS, Baldwin AS. Nuclear factor-kappaB and inhibitor of kappaB kinase pathways in oncogenic initiation and progression. Oncogene. 2006;25:6817–30.
Jost PJ, Ruland J. Aberrant NF-kappaB signaling in lymphoma: mechanisms, consequences, and therapeutic implications. Blood. 2007;109:2700–7.
Cilloni D, Martinelli G, Messa F, Baccarani M, Saglio G. Nuclear factor κB as a target for new drug development in myeloid malignancies. Haematologica. 2007;92:1224–9.
Braun T, Carvalho G, Fabre C, Grosjean J, Fenaux P, Kroemer G. Targeting NF-κB in hematologic malignancies. Cell Death Differ. 2006;13:748–58.
Braun T, Carvalho G, Coquelle A, Vozenin MC, Lepelley P, Hirsch F, et al. NF-kappaB constitutes a potential therapeutic target in high-risk myelodysplastic syndrome. Blood. 2006;107:1156–65.
Kordes U, Krappmann D, Heissmeyer V, Ludwig WD, Scheidereit C. Transcription factor NF-kappaB is constitutively activated in acute lymphoblastic leukemia cells. Leukemia. 2000;14:399–402.
Guzman ML, Neering SJ, Upchurch D, Grimes B, Howard DS, Rizzieri DA, et al. Nuclear factor-kappaB is constitutively activated in primitive human acute myelogenous leukemia cells. Blood. 2001;98:2301–7.
Ni H, Ergin M, Huang Q, Qin JZ, Amin HM, Martinez RL, et al. Analysis of expression of nuclear factor kappa B (NF-kappa B) in multiple myeloma: downregulation of NF-kappa B induces apoptosis. Br J Haematol. 2001;115:279–86.
Bueso-Ramos CE, Rocha FC, Shishodia S, Medeiros LJ, Kantarjian HM, Vadhan-Raj S, et al. Expression of constitutively active nuclear-κB RelA transcription factor in blasts of acute myeloid leukaemia. Hum Pathol. 2003;35:246–53.
Munzert G, Kirchner D, Ottmann O, Bergmann L, Schmid RM. Constitutive NF-κB/Rel activation in Philadelphia chromosome positive (Ph+) acute lymphoblastic leukemia (ALL). Leuk Lymphoma. 2004;45:1181–4.
Bharti AC, Shishodia S, Reuben JM, Weber D, Alexanian R, Raj-Vadhan S, et al. Nuclear factor-kappaB and STAT3 are constitutively active in CD138+ cells derived from multiple myeloma patients, and suppression of these transcription factors leads to apoptosis. Blood. 2004;103:3175–84.
Reuther JY, Reuther GW, Cortez D, Pendergast AM, Baldwin AS Jr. A requirement for NF-kappaB activation in Bcr-Abl mediated transformation. Genes Dev. 1998;12:968–81.
Kirchner D, Duyster J, Ottmann O, Schmid RM, Bergmann L, Munzert G. Mechanisms of Bcr-Abl-mediated NF-kappB/Rel activation. Exp Hematol. 2003;31:504–11.
Conflict of interest
The authors of the present work have no interests, which might be perceived as posing a conflict or bias.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Oktenli, C., Celik, S. High frequency of inherited variants in the MEFV gene in patients with hematologic neoplasms: a genetic susceptibility?. Int J Hematol 95, 380–385 (2012). https://doi.org/10.1007/s12185-012-1061-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-012-1061-6