Abstract
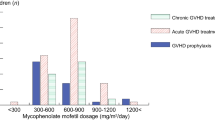
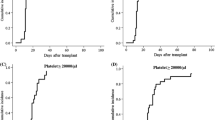
We evaluated the use of mycophenolate mofetil (MMF) after hematopoietic stem cell transplantation (HSCT) in Japan from 1999 to 2008. MMF was administered to 301 patients, including 157 for the prevention of graft-versus-host disease (GVHD), 94 for the treatment of acute GVHD and 50 for the treatment of chronic GVHD. The three most common doses were 500 mg twice daily, 250 mg three times daily and 1,000 mg twice daily, given to 63, 54 and 45 patients, respectively. The incidence of grade II–IV acute GVHD was 30.0% and grade III–IV was 20.0% in the GVHD prevention group. Among treated patients, disappearance or improvement of subjective symptoms occurred in 57.0% of acute GVHD patients and in 52.0% of chronic GVHD patients. With regard to safety, the following major adverse events (grade 3 or more) were recorded: 31 infections, 31 neutropenia, 28 thrombocytopenia, 25 diarrhea and 1 renal disorder. A total of 116 patients developed grade 3 or 4 adverse events, but 79 were successfully treated with supportive treatment. Thus, our findings suggest that MMF is safe and effective for the prevention and treatment of GVHD in patients who have received an allogeneic stem cell transplant.




Similar content being viewed by others
References
Goker H, Haznedaroglu IC, Chao NJ. Acute graft-vs-host disease: pathobiology and management. Exp Hematol. 2001;29:259–77.
Martin PJ, Schoch G, Fisher L, Byers V, Anasetti C, Appelbaum FR, et al. A retrospective analysis of therapy for acute graft-versus-host disease: initial treatment. Blood. 1990;76:1464–72.
Martin PJ, Carpenter PA, Sanders JE, Flowers ME. Diagnosis and clinical management of chronic graft-versus-host disease. Int J Hematol. 2004;79:221–8.
Basara N, Blau WI, Kiehl MG, Romer E, Rudolphi M, Bischoff M, et al. Efficacy and safety of mycophenolate mofetil for the treatment of acute and chronic GVHD in bone marrow transplant recipient. Transplant Proc. 1998;30:4087–9.
Basara N, Blau WI, Kiehl MG, Schmetzer B, Bischoff M, Kirsten D, et al. Mycophenolate mofetil for the prophylaxis of acute GVHD in HLA-mismatched bone marrow transplant patients. Clin Transplant. 2000;14:121–6.
Basara N, Blau WI, Romer E, Rudolphi M, Bischoff M, Kirsten D, et al. Mycophenolate mofetil for the treatment of acute and chronic GVHD in bone marrow transplant patients. Bone Marrow Transplant. 1998;22:61–5.
Bolwell B, Sobecks R, Pohlman B, Andresen S, Rybicki L, Kuczkowski E, et al. A prospective randomized trial comparing cyclosporine and short course methotrexate with cyclosporine and mycophenolate mofetil for GVHD prophylaxis in myeloablative allogeneic bone marrow transplantation. Bone Marrow Transplant. 2004;34:621–5.
Bornhauser M, Schuler U, Porksen G, Naumann R, Geissler G, Thiede C, et al. Mycophenolate mofetil and cyclosporine as graft-versus-host disease prophylaxis after allogeneic blood stem cell transplantation. Transplantation. 1999;67:499–504.
Busca A, Saroglia EM, Lanino E, Manfredini L, Uderzo C, Nicolini B, et al. Mycophenolate mofetil (MMF) as therapy for refractory chronic GVHD (cGVHD) in children receiving bone marrow transplantation. Bone Marrow Transplant. 2000;25:1067–71.
Kim JG, Sohn SK, Kim DH, Lee NY, Suh JS, Lee KS, et al. Different efficacy of mycophenolate mofetil as salvage treatment for acute and chronic GVHD after allogeneic stem cell transplant. Eur J Haematol. 2004;73:56–61.
Mookerjee B, Altomonte V, Vogelsang G. Salvage therapy for refractory chronic graft-versus-host disease with mycophenolate mofetil and tacrolimus. Bone Marrow Transplant. 1999;24:517–20.
Nash RA, Johnston L, Parker P, McCune JS, Storer B, Slattery JT, et al. A phase I/II study of mycophenolate mofetil in combination with cyclosporine for prophylaxis of acute graft-versus-host disease after myeloablative conditioning and allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2005;11:495–505.
Neumann F, Graef T, Tapprich C, Vaupel M, Steidl U, Germing U, et al. Cyclosporine A and mycophenolate mofetil vs cyclosporine A and methotrexate for graft-versus-host disease prophylaxis after stem cell transplantation from HLA-identical siblings. Bone Marrow Transplant. 2005;35:1089–93.
Okamura A, Yamamori M, Shimoyama M, Kawano Y, Kawano H, Kawamori Y, et al. Pharmacokinetics-based optimal dose-exploration of mycophenolate mofetil in allogeneic hematopoietic stem cell transplantation. Int J Hematol. 2008;88:104–10.
Takami A, Mochizuki K, Okumura H, Ito S, Suga Y, Yamazaki H, et al. Mycophenolate mofetil is effective and well tolerated in the treatment of refractory acute and chronic graft-versus-host disease. Int J Hematol. 2006;83:80–5.
Atsuta Y, Suzuki R, Yoshimi A, Gondo H, Tanaka J, Hiraoka A, et al. Unification of hematopoietic stem cell transplantation registries in Japan and establishment of the TRUMP System. Int J Hematol. 2007;86:269–74.
Alousi AM, Weisdorf DJ, Logan BR, Bolanos-Meade J, Carter S, Difronzo N, et al. Etanercept, mycophenolate, denileukin, or pentostatin plus corticosteroids for acute graft-versus-host disease: a randomized phase 2 trial from the Blood and Marrow Transplant Clinical Trials Network. Blood. 2009;114:511–7.
Lee SJ, Vogelsang G, Gilman A, Weisdorf DJ, Pavletic S, Antin JH, et al. A survey of diagnosis, management, and grading of chronic GVHD. Biol Blood Marrow Transplant. 2002;8:32–9.
Lee SJ, Vogelsang G, Flowers ME. Chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2003;9:215–33.
Furlong T, Martin P, Flowers ME, Carnevale-Schianca F, Yatscoff R, Chauncey T, et al. Therapy with mycophenolate mofetil for refractory acute and chronic GVHD. Bone Marrow Transplant. 2009;44:739–48.
Kanda Y, Chiba S, Hirai H, Sakamaki H, Iseki T, Kodera Y, et al. Allogeneic hematopoietic stem cell transplantation from family members other than HLA-identical siblings over the last decade (1991–2000). Blood. 2003;102:1541–7.
Martin PJ, Storer BE, Rowley SD, Flowers ME, Lee SJ, Carpenter PA, et al. Evaluation of mycophenolate mofetil for initial treatment of chronic graft-versus-host disease. Blood. 2009;113:5074–82.
Arai S, Vogelsang GB. Management of graft-versus-host disease. Blood Rev. 2000;14:190–204.
Bornhauser M, Thiede C, Schuler U, Platzbecker U, Freiberg-Richter J, Helwig A, et al. Dose-reduced conditioning for allogeneic blood stem cell transplantation: durable engraftment without antithymocyte globulin. Bone Marrow Transplant. 2000;26:119–25.
Krejci M, Doubek M, Buchler T, Brychtova Y, Vorlicek J, Mayer J. Mycophenolate mofetil for the treatment of acute and chronic steroid-refractory graft-versus-host disease. Ann Hematol. 2005;84:681–5.
Nash RA, Antin JH, Karanes C, Fay JW, Avalos BR, Yeager AM, et al. Phase 3 study comparing methotrexate and tacrolimus with methotrexate and cyclosporine for prophylaxis of acute graft-versus-host disease after marrow transplantation from unrelated donors. Blood. 2000;96:2062–8.
Ratanatharathorn V, Nash RA, Przepiorka D, Devine SM, Klein JL, Weisdorf D, et al. Phase III study comparing methotrexate and tacrolimus (prograf, FK506) with methotrexate and cyclosporine for graft-versus-host disease prophylaxis after HLA-identical sibling bone marrow transplantation. Blood. 1998;92:2303–14.
Acknowledgments
This work was supported in part by Health and Labour Sciences Research Grants for Clinical Cancer Research from the Ministry of Health, Labour and Welfare, Japan. The authors would like to thank the staff of the Data Center of the Japan Society for Hematopoietic Cell Transplantation and the following collaborating institutions for providing patient data and specimens: Hokkaido University, Sapporo Hokuyu Hospital, Sapporo City General Hospital, Hakodate Municipal Hospital, Aomori Prefectural Central Hospital, Tsukuba University, Gunma University, Saitama Medical University International Medical Center, Chiba University, Jikei University Kashiwa Hospital, National Cancer Center Hospital, Tokyo Metropolitan Cancer and Infectious Diseases Center Komagome Hospital, Keio University, Tokyo Women’s Medical University, Kyorinn University, Toranomon Hospital, Tokai University, St. Marianna University Yokohama City Seibu Hospital, Nagaoka Red Cross Hospital, Kouseiren Takaoka Hospital, Kanazawa University, Yamanashi Prefectural Central Hospital, Shinshu University, Nagano Red Cross Hospital, Gifu University, Hamamatsu Medical University, Nagoya University, National Hospital Organization Nagoya Medical Center, Konan Kosei Hospital, Mie University, Yamada Red Cross Hospital, Shiga University, Kyoto University, Kyoto Prefectural University, Kinki University, Osaka City University, Osaka City General Hospital, Matsushita Memorial Hospital, Osaka Medical College, Kitano Hospital, Hyogo College of Medicine, Kobe University, Kurashiki Central Hospital, Hiroshima University, National Hospital Organization Kure Medical Center, Yamaguchi University, Tokushima University, Tokushima Red Cross Hospital, Kagawa University, Ehime Prefectural Central Hospital, Ehime University, Kyushu University First Department of Internal Medicine, Kyusyu University Third Department of Internal Medicine, Hamanomachi Hospital, National Organization Kyusyu Cancer Center, Nagasaki University, Sasebo City General Hospital, Oita University, Kyusyu University Hospital at Beppu, Imamura Bun-in Hospital and Kagoshima University.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iida, M., Fukuda, T., Ikegame, K. et al. Use of mycophenolate mofetil in patients received allogeneic hematopoietic stem cell transplantation in Japan. Int J Hematol 93, 523–531 (2011). https://doi.org/10.1007/s12185-011-0817-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-011-0817-8