Abstract
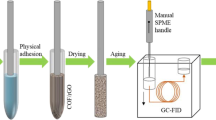
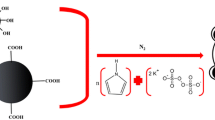
Polyaniline-modified magnetic halloysite nanotube composites (MHNTs@PANI) were evaluated for the first time as an efficient adsorbent for the magnetic micro-solid-phase extraction of polycyclic aromatic hydrocarbons (PAHs) from beer samples prior to gas chromatography-mass spectrometric detection. MHNTs@PANI were prepared from naturally available material halloysite by coating magnetic HNTs with polyaniline. After characterization, MHNTs@PANI were used to extract fluorene, phenanthrene, anthracene, pyrene, benz [a] anthracene, and chrysene via π-π and hydrophobic interaction between PANI and analytes. The effects of the amount of adsorbent, ionic strength, type of eluent, extraction time, and desorption time were investigated. Under optimum conditions, good linearity was obtained for the analytes in the concentration range of 0.05–100 μg L−1 with R2 between 0.9962 and 0.9991. The limits of detection were in the range of 1.64–14.20 ng L−1 and the spiked recoveries ranged from 78.8 to 114.6%. The proposed method proved to be efficient, rapid, sensitive, and reliable, which has been successfully applied to the determination of PAHs in eight brands of beer samples.





Similar content being viewed by others
References
Azari Z, Pourbasheer E, Beheshti A (2016) Mixed hemimicelles solid-phase extraction based on sodium dodecyl sulfate (SDS)-coated nano-magnets for the spectrophotometric determination of fingolomid in biological fluids. Spectrochim Acta A Mol Biomol Spectrosc 153:599–604. https://doi.org/10.1016/j.saa.2015.09.013
Brányik T, Silva DP, Baszczynski M, Lehnert R, Silva JBAE (2012) A review of methods of low alcohol and alcohol-free beer production. J Food Eng 108:493–506. https://doi.org/10.1016/j.jfoodeng.2011.09.020
Cid Martín JJ, Assali M, Fernández-García E, Valdivia V, Sánchez-Fernández EM, Garcia Fernández JM, Wellinger RE, Fernández I, Khiar N (2016) Tuning of glyconanomaterial shape and size for selective bacterial cell agglutination. J Mater Chem B 4:2028–2037. https://doi.org/10.1039/c5tb02488a
Darvishnejad M, Ebrahimzadeh H (2018) Magnetic halloysite nanotube/polyaniline/copper composite coupled with gas chromatography-mass spectrometry: a rapid approach for determination of nitro-phenanthrenes in water and soil samples. J Chromatogr A 1563:1–9. https://doi.org/10.1016/j.chroma.2018.05.051
Dramou P, Fizir M, Taleb A, Itatahine A, Dahiru NS, Mehdi YA, Wei L, Zhang J, He H (2018) Folic acid-conjugated chitosan oligosaccharide-magnetic halloysite nanotubes as a delivery system for camptothecin. Carbohydr Polym 197:117–127. https://doi.org/10.1016/j.carbpol.2018.05.071
Edokpayi JN, Odiyo JO, Popoola OE, Msagati TA (2016) Determination and distribution of polycyclic aromatic hydrocarbons in rivers, sediments and wastewater effluents in Vhembe district, South Africa. Int J Environ Res Public Health 13:387. https://doi.org/10.3390/ijerph13040387
Fan C, Cao XL, Han T, Pei HR, Hu GH, Wang W, Qian C (2019) Selective microextraction of polycyclic aromatic hydrocarbons using a hydrophobic deep eutectic solvent composed with an iron oxide-based nanoferrofluid. Microchim Acta 186:UNSP 560. https://doi.org/10.1007/s00604-019-3651-y
Fasano E, Yebra-Pimentel I, Martínez-Carballo E, Simal-Gándara J (2016) Profiling, distribution and levels of carcinogenic polycyclic aromatic hydrocarbons in traditional smoked plant and animal foods. Food Control 59:581–590. https://doi.org/10.1016/j.foodcont.2015.06.036
Galinaro CA, Franco DW (2009) Polycyclic aromatic hydrocarbons (PAHs) in cachaça, rum, whiskey and alcohol fuel. Quim Nova 32:1447–1451. https://doi.org/10.1590/S0100-40422009000600016
Garcia-Falcon MS, Simal-Gandara J (2005) Determination of polycyclic aromatic hydrocarbons in alcoholic drinks and the identification of their potential sources. Food Addit Contam A 22:791–797. https://doi.org/10.1080/02652030500198498
Garcia-Londoño VA, Reynoso CM, Resnik SL (2015) Polycyclic aromatic hydrocarbons (PAHs) survey on tea (Camellia sinensis) commercialized in Argentina. Food Control 50:31–37. https://doi.org/10.1016/j.foodcont.2014.07.036
German-Hernandez M, Crespo-Llabres P, Pino V, Ayala JH, Afonso AM (2013) Utilization of an ionic liquid in situ preconcentration method for the determination of the 15 + 1 European Union polycyclic aromatic hydrocarbons in drinking water and fruit-tea infusions. J Sep Sci 36:2496–2506. https://doi.org/10.1002/jssc.201300393
Grover IS, Singh S, Pal B (2013) Priority PAHs in orthodox black tea during manufacturing process. Environ Monit Assess 185:6291–6294. https://doi.org/10.1007/s10661-012-3025-y
Guan XM, Zhao CD, Liu XY, Zhang HX (2013) Hyperbranched polymers containing stereocontorted cores as on-line solid-phase microextraction adsorbent for polycyclic aromatic hydrocarbons. J Chromatogr A 1302:28–33. https://doi.org/10.1016/j.chroma.2013.06.033
Guo L, Tan SF, Li X, Lee HK (2016) Fast automated dual-syringe based dispersive liquid–liquid microextraction coupled with gas chromatography–mass spectrometry for the determination of polycyclic aromatic hydrocarbons in environmental water samples. J Chromatogr A 1438:1–9. https://doi.org/10.1016/j.chroma.2016.02.008
Iwegbue CMA, Agadaga H, Bassey FI, Overah LC, Tesi GO, Nwajei GE (2015) Concentrations and profiles of polycyclic aromatic hydrocarbons in some commercial brands of tea-, coffee-, and cocoa-based food drinks in Nigeria. Int J Food Prop 18:2124–2133. https://doi.org/10.1080/10942912.2014.908906
Li Z, Wang Y, Ni Y, Kokot S (2015) Fluorescence analysis of 6-mercaptopurine with the use of a nano-composite consisting of BSA-capped Au nano-clusters and core–shell Fe3O4–SiO2 nanoparticles. Biosens Bioelectron 70:246–253. https://doi.org/10.1016/j.bios.2015.03.035
Lin DH, Tu YY, Zhu LZ (2005) Concentrations and health risk of polycyclic aromatic hydrocarbons in tea. Food Chem Toxicol 43:41–48. https://doi.org/10.1016/j.fct.2004.08.010
Liu Y, Li HF, Lin JM (2009) Magnetic solid-phase extraction based on octadecyl functionalization of monodisperse magnetic ferrite microspheres for the determination of polycyclic aromatic hydrocarbons in aqueous samples coupled with gas chromatography–mass spectrometry. Talanta 77:1037–1042. https://doi.org/10.1016/j.talanta.2008.08.013
Liu XF, Lu X, Huang Y, Liu CW, Zhao SL (2014) Fe3O4@ionic liquid@methyl orange nanoparticles as a novel nano-adsorbent for magnetic solid-phase extraction of polycyclic aromatic hydrocarbons in environmental water samples. Talanta 119:341–347. https://doi.org/10.1016/j.talanta.2013.11.039
Lvov Y, Abdullayev E (2013) Functional polymer–clay nanotube composites with sustained release of chemical agents. Prog Polym Sci 38:1690–1719. https://doi.org/10.1016/j.progpolymsci.2013.05.009
Massaro M, Colletti CG, Lazzara G, Guernelli S, Noto R, Riela S (2017) Synthesis and characterization of halloysite–cyclodextrin nanosponges for enhanced dyes adsorption. ACS Sustain Chem Eng 5:3346–3352. https://doi.org/10.1021/acssuschemeng.6b03191
Mehdinia A, Shoormeij Z, Jabbari A (2017) Trace determination of lead (II) ions by using a magnetic nanocomposite of the type Fe3O4/TiO2/PPy as a sorbent, and FAAS for quantitation. Microchim Acta 184:1529–1537. https://doi.org/10.1007/s00604-017-2156-9
Naing NN, Li SFY, Lee HK (2016) Magnetic micro-solid-phase-extraction of polycyclic aromatic hydrocarbons in water. J Chromatogr A 1440:23–30. https://doi.org/10.1016/j.chroma.2016.02.046
Pan J, Yao H, Xu L, Ou H, Huo P, Li X, Yan Y (2011) Selective recognition of 2,4,6-trichlorophenol by molecularly imprinted polymers based on magnetic halloysite nanotubes composites. J Phys Chem C 115:5440–5449. https://doi.org/10.1021/jp111120x
Pan JM, Wang B, Dai JD, Dai XH, Hang H, Ou HX, Yan YS (2012) Selective recognition of 2,4,5-trichlorophenol by temperature responsive and magnetic molecularly imprinted polymers based on halloysite nanotubes. J Mater Chem 22:3360–3369. https://doi.org/10.1039/c1jm14825g
Paris A, Gaillard JL, Ledauphin J (2019) Rapid extraction of polycyclic aromatic hydrocarbons in apple: ultrasound-assisted solvent extraction followed by microextraction by packed sorbent. Food Anal Methods 12:2194–2204. https://doi.org/10.1007/s12161-019-01568-7
Rascon AJ, Azzouz A, Ballesteros E (2019) Use of semi-automated continuous solid-phase extraction and gas chromatography-mass spectrometry for the determination of polycyclic aromatic hydrocarbons in alcoholic and non-alcoholic drinks from Andalucia (Spain). J Sci Food Agric 99:1117–1125. https://doi.org/10.1002/jsfa.9279
Ricardo AIC, Bernardo FJG, Zougagh M, Martín-Doimeadios RCR, Rios A (2017) Magnetic nanoparticles-carbon nanotubes hybrid composites for selective solid-phase extraction of polycyclic aromatic hydrocarbons and determination by ultra-high performance liquid chromatography. Anal Bioanal Chem 409:5125–5132. https://doi.org/10.1007/s00216-017-0459-7
Russo MV, Avino P, Perugini L, Notardonato I (2016) Fast analysis of nine PAHs in beer by ultrasound-vortex-assisted dispersive liquid-liquid micro-extraction coupled with gas chromatography-ion trap mass spectrometry. RSC Adv 6:13920–13927. https://doi.org/10.1039/c5ra24873f
Singh L, Varshney JG, Agarwal T (2016) Polycyclic aromatic hydrocarbons’ formation and occurrence in processed food. Food Chem 99:768–781. https://doi.org/10.1016/j.foodchem.2015.12.074
Szternfeld P, Marchi J, Malysheva SV, Joly L (2019) modular method for the determination of polycyclic aromatic hydrocarbons in spices and dried herbs by gas chromatography-tandem mass spectrometry. Food Anal Methods 12:2383–2391. https://doi.org/10.1007/s12161-019-01579-4
USEPA (2012) United States Environmental Protection Agency. Appendix A to 40 CFR, Part 423-126. U.S. EPA, Washington, D.C
Wang M, Cui SH, Yang XD, Bi WT (2015) Synthesis of g-C3N4/Fe3O4 nanocomposites and application as a new sorbent for solid phase extraction of polycyclic aromatic hydrocarbons in water samples. Talanta 132:922–928. https://doi.org/10.1016/j.talanta.2014.08.071
Wenzl T, Simon R, Kleiner J, Anklam E (2006) Analytical methods for polycyclic aromatic hydrocarbons (PAHs) in food and the environment needed for new food legislation in the European Union. TrAC Trends Anal Chem 25:716–725. https://doi.org/10.1016/j.trac.2006.05.010
Yang XL, Qiao KX, Ye YR, Yang MY, Li J, Gao HX, Zhang SB, Zhou WF, Lu RH (2016) Facile synthesis of multifunctional attapulgite/Fe3O4/polyaniline nanocomposites for magnetic dispersive solid phase extraction of benzoylurea insecticides in environmental water samples. Anal Chim Acta 934:114–121. https://doi.org/10.1016/j.aca.2016.06.027
Yang XM, Wang JM, Wang WJ, Zhang SH, Wang C, Zhou JH, Wang Z (2019a) Solid phase microextraction of polycyclic aromatic hydrocarbons by using an etched stainless-steel fiber coated with a covalent organic framework. Microchim Acta 186:145. https://doi.org/10.1007/s00604-019-3258-3
Yang XP, Yin YR, Zong YY, Wan T, Liao XJ (2019b) Magnetic nanocomposite as sorbent for magnetic solid phase extraction coupled with high performance liquid chromatography for determination of polycyclic aromatic hydrocarbons. Microchem J 145:26–34. https://doi.org/10.1016/j.microc.2018.10.013
Yazdanpanah M, Nojavan S (2019) Micro-solid phase extraction of some polycyclic aromatic hydrocarbons from environmental water samples using magnetic-cyclodextrin-carbon nano-tube composite as a sorbent. J Chromatogr A 1585:34–45. https://doi.org/10.1016/j.chroma.2018.11.066
Yoshioka T, Nagatomi Y, Harayama K, Bamba T (2018) Development of an analytical method for polycyclic aromatic hydrocarbons in coffee beverages and dark beer using novel high-sensitivity technique of supercritical fluid chromatography/mass spectrometry. J Biosci Bioeng 126:126–130. https://doi.org/10.1016/j.jbiosc.2018.01.014
Yuan P, Tan D, Annabi-Bergaya F (2015) Properties and applications of halloysite nanotubes: recent research advances and future prospects. Appl Clay Sci 112-113:75–93. https://doi.org/10.1016/j.clay.2015.05.001
Zheng HB, Ding J, Zheng SJ, Zhu GT, Yuan BF, Feng YQ (2016) Facile synthesis of magnetic carbon nitride nanosheets and its application in magnetic solid phase extraction for polycyclic aromatic hydrocarbons in edible oil samples. Talanta 148:46–53. https://doi.org/10.1016/j.talanta.2015.10.059
Zhou Q, Lei M, Wu Y, Yuan Y (2017a) Magnetic solid phase extraction of typical polycyclic aromatic hydrocarbons from environmental water samples with metal organic framework MIL-101 (Cr) modified zero valent iron nano-particles. J Chromatogr A 1487:22–29. https://doi.org/10.1016/j.chroma.2017.01.046
Zhou T, Li C, Jin H, Lian Y, Han W (2017b) Effective adsorption/reduction of Cr (VI) oxyanion by halloysite@polyaniline hybrid nanotubes. ACS Appl Mater Interfaces 9:6030–6043. https://doi.org/10.1021/acsami.6b14079
Zhu X, Li H, Zhou H, Zhong S (2015) Fabrication and evaluation of protein imprinted polymer based on magnetic halloysite nanotubes. RSC Adv 5:66147–66154. https://doi.org/10.1039/c5ra09740a
Funding
The authors acknowledge the financial support from Department of Education of Hebei Province (ZD2019089).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Zhihong Shi declares that she has no conflict of interest. Weiyue Pang declares that she has no conflict of interest. Man Chen declares that she has no conflict of interest. Yifan Wu declares that he has no conflict of interest. Hongyi Zhang declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent is not applicable for the nature of this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shi, Z., Pang, W., Chen, M. et al. Polyaniline-Modified Magnetic Halloysite Nanotube-Based Magnetic Micro-Solid-Phase Extraction for the Analysis of Polycyclic Aromatic Hydrocarbons in Beer Samples by Gas Chromatography-Mass Spectrometry. Food Anal. Methods 14, 761–772 (2021). https://doi.org/10.1007/s12161-020-01917-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01917-x