Abstract
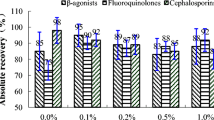
A sensitive liquid chromatography coupled to high-resolution tandem mass spectrometry (LC-IT-TOF MS) with electrospray ionization method for the identification and quantification of five antibiotic residues in milk has been developed. For sample preparation, a simple modification of the QuEChERS method was used with further sample cleanup using dispersive solid phase extraction according to AOAC official standards 2007.01. The method yielded acceptable accuracy values for each drug at each level, with mean recoveries (n = 3) ranging from 83 to 92 %. The recovery of antibiotics of the intra-assay ranged from 85 to 95 % (RSD < 9 %) and the inter-assay from 84 to 95 % (RSD < 11 %). A total of 31 milk samples, either pasteurized or fresh whole milk, were analyzed following the method. Amoxicillin and oxytetracycline (two of the pasteurized milk samples), ampicillin and amoxicillin (3 of 25 fresh milk samples), and tetracycline and oxytetracycline (two other fresh milk samples out of 25) were detected at levels below the current Brazilian maximum residue levels.


Similar content being viewed by others
References
Aguilera-Luiz MM, Vidal JLM, Romero-González R, Frenich AG (2008) J Chromatogr A 1205:10–16
Almeida LP, Vieira RL, Rossi DA, Carneiro AL, Rocha ML (2003) Biosci J 19:83–87
Anastassiades M, Lehotay SJ, Stajnbaher D, Schenck FJ (2003) J AOAC Int 86:412–431
ANVISA (2009) Agência Nacional de Vigilância Sanitária. Programa de Análise de Resíduos de Medicamentos Veterinários em Alimentos de Origem Animal-PAMVET- Relatório 2006-2007, Monitoramento de Resíduos em Leite Expostoao Consumo (5º e 6º ano de atividades) Brasília (DF): http://portal.anvisa.gov.br/wps/wcm/connect/d7ab358047458ad19443d43fbc4c6735/PAMVET.pdf?MOD=AJPERES. Accessed 6 Mar 2013
Capar SG, Cook JM, Bell J, Hickes H, Krynitsky AJ, Lehotay SJ, Ritland CL, Schenck FJ, Soderberg D, Phillips J (2008) J AOAC Int 91:46B–48B
EC (2010) Council Regulation (EC) 37/2010/EC of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin (2010). Off J Eur Comm L15:1–72
Gamba V, Terzano C, Fiorini L, Moretti S, Dusi G, Galarini R (2009) Anal Chim Acta 637:18–23
Junza A, Amatya R, Barrón D, Barbosa J (2011) J Chromatogr B 879:2601–2610
Kennedy DG, Mccracken RJ, Cannavan A, Hewitt SA (1998) J Chromatogr A 812:77–98
Kinsella B, Lehotay SJ, Mastovska K, Lightfield AR, Furey A, Danaher M (2009) Anal Chim Acta 637:196–207
Lehotay SJ, De Kok A, Hienistra M, van Bodegraven P (2005) J AOAC Int 88:595–614
Lehotay SJ, Son KA, Kwon H, Koesukwiwat U, Fu W, Mastovska K, Hoh E (2010) J Chromatogr A 1217:2548–2560
Mamani MCV, Reyes FGR, Rath S (2009) Food Chem 117:545–552
MAPA (2010) Ministério de Agricultura, Pecuária e Abastecimento do Brasil—MAPA—Programa Nacional de Controle de Resíduos e Contaminantes em Leite—PNCRC—Diário oficial da união-seção 1 ISSN 1677-7042, 03 de Maio de 2010
Martín SH, Pinto CG, Pavón JLP, Cordero BM (2010) J Chromatogr A 1217:4883–4889
Mastovska K, Lehotay SJ (2004) J Chromatogr A 1040:259–272
Nebot C, Iglesias A, Regal P, Miranda J, Cepeda A, Gente C (2012) Int Dairy J 22:78–85
O’Keeffe M, Kennedy O (1998) J Food Saf 18:297–319
Ortelli D, Cognard E, Jan P, Edder P (2009) J Chromatogr B 877:2363–2374
Payá P, Anastassiades M, Mack D, Sigalova I, Tasdelen B, Oliva J, Barba A (2007) Anal Bioanal Chem 389:1697–1714
Plossl F, Giero M, Bracher F (2006) J Chromatogr A 1135:19–26
Prestes OD, Friggi CA, Adaime MB, Zanella R (2009) Quím Nova 32:1620–1634
Rønning HT, Einarsen K, Asp TN (2006) J Chromatogr A 1118:226–233
Santos SM, Henriques M, Duarte AC, Esteves VI (2007) Talanta 71:731–737
Sheridan R, Policastro B, Thomas S, Rice D (2008) J Agric Food Chem 56:3509–3516
Stolker AAM, Rutgers P, Oosterink E, Lasaroms JJP, Peters RJB, van Rhijn JA, Nielen NWF (2008) Anal Bioanal Chem 391:2309–2322
Stubbings G, Bigwood T (2009) Anal Chim Acta 637:68–78
van Holthoon F, Mulder PPJ, van Bennekom EO, Heskamp H, Zuidema T, van Rhijn HJA (2010) Anal Bioanal Chem 396:3027–3040
Acknowledgments
Financial supports from CAPES, CNPq, and FACEPE are gratefully acknowledgement. The authors are grateful to Professor Suzana P.V. Nova (UAG-UFRPE) and Professor Lilian S.S. de Andrade (UFRPE) for help in collecting and sending milk samples and for suggestions ans to Severino Granjeiro Junior (LAFEPE), Rogério Ferreira da Silva (UFPE), and Edén Cavalcanti de Albuquerque Júnior (ITEP) for technical support.
Conflict of Interest
Sueny Kêlia Barbosa Freitas declares that she has no conflict of interest. Ana Paula Silveira Paim declares that she has no conflict of interest. Paula Tereza de Souza e Silva declares that she has no conflict of interest. This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Freitas, S.K.B., Paim, A.P.S. & de Souza e Silva, P.T. Development of a LC-IT-TOF MS Procedure to Quantify Veterinary Drug Residues in Milk Employing a QuEChERS Approach. Food Anal. Methods 7, 39–46 (2014). https://doi.org/10.1007/s12161-013-9595-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-013-9595-7