Abstract
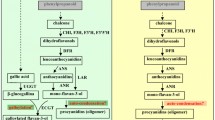
The use of pyrogallol as trapping reagent in proanthocyanidin (PA) analysis was investigated. PAs are antioxidants and disease-preventing agents, the efficiency of which depends on their composition and size. Different nucleophilic compounds are usually used for acid-catalyzed depolymerization of proanthocyanidins: benzyl mercaptan, phloroglucinol, and cysteamine. Each nucleophile has different characteristics and typical reaction temperatures that ranges from 50 to 90 °C. Pyrogallol is more reactive than these compounds, so it was possible to apply a minor reaction temperature that was finally reduced from 50 to 30 °C, compared to phloroglucinol, but maintaining a significant yield. The results, compared to those obtained using phloroglucinol as trapping nucleophile, indicated that pyrogallol is an effective and interesting reagent for this analysis.






Similar content being viewed by others
References
Bordiga M, Travaglia F, Coïsson JD, Locatelli M, Arlorio M, Martelli A (2009) Proceedings of 32nd World Congress of Vine and Wine; Zagreb, Croatia
Bordiga M, Travaglia F, Locatelli M, Coisson JD, Arlorio M (2011) Food Chem 127:180
Del Bas JM, Fernandez-Larrea J, Blay M et al (2005) FASEB J 19:479
Dixon RA, Sharma SB, Xie D (2005) New Phytol 165:9
Escribano-Bailòn MT, Guitiérrez-Fernàndez Y, Rivas-Gonzalo JC, Santos-Buelga C (1992) J Agric Food Chem 40:1794
Fitzpatrick DF, Fleming RC, Bing B, Maggi DA, O’Malley RM (2000) J Agric Food Chem 48:6384
Foo LY (1985) J Chem Soc Chem Commun 1273
Foo LY, Porter LJ (1978) J Chem Soc Perkin Trans 1:1186
Glories Y (1978) These doctorat d’Etat del’Universite de Bordeaux II
Hemingway RW (1989) Chemistry and significance of condensed tannins. Plenum, New York
Hemingway RW, McGraw GW (1983) J Wood Chem Technol 3:421
Jerez M, Tourino S, Sineiro J, Torres JL, Nunez MJ (2007) Food Chem 104:518
Kennedy JA, Jones GP (2001) J Agric Food Chem 49:1740
Kennedy JA, Matthews MA, Waterhouse AL (2000) Phytochemistry 55:77
Lago-Vanzela ES, Da-Silva R, Gomes E, García-Romero E, Hermosín-Gutierrez I (2011) J Agric Food Chem 59:8314
Matthews S, Mila I, Scalbert A et al (1997) J Agric Food Chem 45:1195
Oszmianski J, Wolniak M, Wojdylo A, Wawer I (2007) J Sci Food Agric 87:573
Park JC, Ito H, Yoshida T (2003) Nat Prod Sci 9:49
Pastor del Rio J, Kennedy JA (2006) Am J Enol Vitic 57:125
Prieur C, Rigaud J, Cheynier R, Moutounet M (1994) Phytochemistry 36:781
R Development Core Team (2008) R: a language and environment for statistical computing. RFoundation for Statistical Computing, Vienna
Rasmussen SE, Fredriksen H, Krogholm KS, Poulsen L (2005) Mol Nutr Food Res 49:159
Rodrıguez-Montealegre R, Romero-Peces R, Chacon-Vozmediano JL, Martınez Gascuena J, Garcıa Romero E (2006) J Food Compos Anal 19:687
Scalbert A, Haslam E (1987) Phytochemistry 26:3191
Shao ZH, Becker LB, Vanden Hoek TL et al (2003) Pharmacol Res 47:463
Singleton VL (1992) In: Hemingway RW (ed) Plant polyphenols: synthesis, properties, significance. Plenum, New York, pp 859–880
Sun BS, Leandro C, Ricardo da Silva JM, Spranger I (1998) J Agric Food Chem 46:1390
Thompson RS, Jacques D, Haslam E, Tanner RJN (1972) J Chem Soc Perkin Trans 1:1387
Torres JL, Lozano C (2001) Chromatographia 54:523
Travaglia F, Bordiga M, Locatelli M, Coisson JD, Arlorio M (2011) J Food Sci 76:C742
Acknowledgments
Dr. Matteo Bordiga was a recipient of a fellowship from Italian Politiche Agricole Alimentari e Forestali Ministry (Food-Link project) and Dr. Monica Locatelli from Cariplo Foundation (Nutrial Network 2010 project; cod. 2009-2961).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bordiga, M., Coïsson, J.D., Locatelli, M. et al. Pyrogallol: an Alternative Trapping Agent in Proanthocyanidins Analysis. Food Anal. Methods 6, 148–156 (2013). https://doi.org/10.1007/s12161-012-9427-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-012-9427-1