Abstract
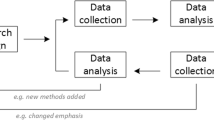
The multiphase optimization strategy (MOST) is a new methodological approach for building, optimizing, and evaluating multicomponent interventions. Conceptually rooted in engineering, MOST emphasizes efficiency and careful management of resources to move intervention science forward steadily and incrementally. MOST can be used to guide the evaluation of research evidence, develop an optimal intervention (the best set of intervention components), and enhance the translation of research findings, particularly type II translation. This article uses an ongoing study to illustrate the application of MOST in the evaluation of diverse intervention components derived from the phase-based framework reviewed in the companion article by Baker et al. (Ann Behav Med, in press, 2011). The article also discusses considerations, challenges, and potential benefits associated with using MOST and similar principled approaches to improving intervention efficacy, effectiveness, and cost-effectiveness. The applicability of this methodology may extend beyond smoking cessation to the development of behavioral interventions for other chronic health challenges.

Similar content being viewed by others
References
Baker TB, Mermelstein R, Collins LM, et al. New methods for tobacco dependence treatment research. Ann Behav Med. 2011; in press.
Fiore MC, Bailey WC, Cohen SJ. Treating tobacco use and dependence: Clinical practice guideline. Rockville: U.S. Department of Health and Human Services, U.S. Public Health Service; 2000.
Fiore MC, Jaen CR, Baker TB, et al. Treating tobacco use and dependence: 2008 update. Rockville: U.S. Department of Health and Human Services, U.S. Public Health Service; 2008.
Collins LM, Murphy SA, Nair VN, Strecher VJ. A strategy for optimizing and evaluating behavioral interventions. Ann Behav Med. 2005;30(1):65–73.
Collins LM, Murphy SA, Strecher V. The Multiphase Optimization Strategy (MOST) and the Sequential Multiple Assignment Randomized Trial (SMART): New methods for more potent eHealth interventions. Am J Prev Med. 2007;32(5 Suppl):S112–8.
Hollis JF, Bills R, Whitlock E, Stevens VJ, Mullooly J, Lichtenstein E. Implementing tobacco interventions in the real world of managed care. Tob Control. 2000;9(Suppl 1):I18–124.
Hung DY, Shelley DR. Multilevel analysis of the Chronic Care Model and 5A services for treating tobacco use in urban primary care clinics. Health Serv Res. 2008;44:103–27.
Kottke TE, Solberg LI, Brekke ML, Cabrera A, Marquez MA. Delivery rates for preventive services in 44 Midwestern clinics. Mayo Clin Proc. 1997;72(6):515–23.
Glasgow RE, Emmons KM. How can we increase translation of research into practice? Types of evidence needed. Annu Rev Public Health. 2007;28:413–33.
Glasgow RE, Lichtenstein E, Marcus AC. Why don't we see more translation of health promotion research to practice? Rethinking the efficacy-to-effectiveness transition. Am J Public Health. 2003;93(8):1261–7.
Box GEP, Hunter, W.G., & Hunter, J.S. Statistics for experimenters: An introduction to design, data analysis, and model building. New York: Wiley; 1978.
Wu CF, Hamada M. Experiments: Planning, analysis, and parameter design optimization. New York: Wiley; 2000.
Shadish WR, Cook TD, Campbell DT. Experimental and quasi-experimental designs for generalized causal inference. Boston: Houghton-Mifflin; 2002.
Campbell MK, Mollison J, Steen N, Grimshaw JM, Eccles M. Analysis of cluster randomized trials in primary care: A practical approach. Fam Pract. 2000;17(2):192–6.
Craig P, Dieppe P, Macintyre S, Michie S, Nazareth I, Petticrew M. Developing and evaluating complex interventions: The new Medical Research Council guidance. BMJ. 2008;337:a1655.
Rose JE, Behm FM, Westman EC, Kukovich P. Precessation treatment with nicotine skin patch facilitates smoking cessation. Nicotine Tob Res. 2006;8(1):89–101.
Moore D, Aveyard P, Connock M, Wang D, Fry-Smith A, Barton P. Effectiveness and safety of nicotine replacement therapy assisted reduction to stop smoking: Systematic review and meta-analysis. BMJ. 2009;338:b1024.
Hughes JR, Carpenter MJ. The feasibility of smoking reduction: An update. Addiction. 2005;100(8):1074–89.
Hollis JF, McAfee TA, Fellows JL, Zbikowski SM, Stark M, Riedlinger K. The effectiveness and cost effectiveness of telephone counselling and the nicotine patch in a state tobacco quitline. Tob Control. 2007;16 Suppl 1:i53–9.
Zhu SH, Stretch V, Balabanis M, Rosbrook B, Sadler G, Pierce JP. Telephone counseling for smoking cessation: Effects of single-session and multiple-session interventions. J Consult Clin Psychol. 1996;64(1):202–11.
Zhu SH, Tedeschi G, Anderson CM, et al. Telephone counseling as adjuvant treatment for nicotine replacement therapy in a “real-world” setting. Prev Med. 2000;31(4):357–63.
Zhu SH, Anderson CM, Tedeschi GJ, et al. Evidence of real-world effectiveness of a telephone quitline for smokers. New Engl J Med. 2002;347(14):1087–93.
Tonstad S, Tonnesen P, Hajek P, Williams KE, Billing CB, Reeves KR. Effect of maintenance therapy with varenicline on smoking cessation: A randomized controlled trial. JAMA. 2006;296(1):64–71.
Covey LS, Glassman AH, Jiang H, et al. A randomized trial of bupropion and/or nicotine gum as maintenance treatment for preventing smoking relapse. Addiction. 2007;102(8):1292–302.
Hays JT, Hurt RD, Rigotti NA, et al. Sustained-release bupropion for pharmacologic relapse prevention after smoking cessation. A randomized, controlled trial. Ann Intern Med. 2001;135(6):423–33.
Dale Horst W, Klein MW, Williams D, Werder SF. Extended use of nicotine replacement therapy to maintain smoking cessation in persons with schizophrenia. Neuropsychiatr Dis Treat. 2005;1(4):349–55.
Helgason AR, Tomson T, Lund KE, Galanti R, Ahnve S, Gilljam H. Factors related to abstinence in a telephone helpline for smoking cessation. Eur J Public Health. 2004;14(3):306–10.
Fiore MC, Smith SS, Jorenby DE, Baker TB. The effectiveness of the nicotine patch for smoking cessation. A meta-analysis. JAMA. 1994;271(24):1940–7.
Piper ME, Federmen EB, McCarthy DE, et al. Using mediational models to explore the nature of tobacco motivation and tobacco treatment effects. J Abnorm Psychol. 2008;117(1):94–105.
Jorenby DE, Hays JT, Rigotti NA, et al. Efficacy of varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: A randomized controlled trial. JAMA. 2006;296(1):56–63.
Ferguson SG, Shiffman S, Gwaltney CJ. Does reducing withdrawal severity mediate nicotine patch efficacy? A randomized clinical trial. J Consult Clin Psychol. 2006;74(6):1153–61.
Shiffman S, Scharf DM, Shadel WG, et al. Analyzing milestones in smoking cessation: Illustration in a nicotine patch trial in adult smokers. J Consult Clin Psychol. 2006;74(2):276–85.
Collins LM, Dziak JJ, Li R. Design of experiments with multiple independent variables: A resource management perspective on complete and reduced factorial designs. Psychol Methods. 2009;14(3):202–24.
Fisher RA. The arrangement of field experiments. Journal of the Ministry of Agriculture of Great Britain. 1926;33:505–13.
Chakraborty H. The design and analysis aspects of cluster randomized trials. In: Biswas A, Datta S, Fine JP, Segal MR, eds. Statistical Advances in the Biomedical Sciences. Hoboken: Wiley; 2008:67–80.
SAS Institute. SAS/STAT 9.1 User's Guide. Cary: SAS Institute; 2004.
SAS Institute. SAS/QC9.1 User's Guide. Cary: SAS Institute; 2004.
Rivera DE, Pew MD, Collins LM. Using engineering control principles to inform the design of adaptive interventions: A conceptual introduction. Drug Alcohol Depend. 2007;88 Suppl 2:S31–40.
Zafra-Cabeza A, Rivera DE, Collins LM, Riado MA, Camacho EF. A risk-based model predictive control approach to adaptive interventions in behavioral health. Proceedings of the 45th IEEE Conference on Decision and Control. 2006:673–8.
Carroll KM, Rounsaville BJ. Bridging the gap: A hybrid model to link efficacy and effectiveness research in substance abuse treatment. Psychiatr Serv. 2003;54(3):333–9.
Carroll KM, Rounsaville BJ. A vision of the next generation of behavioral therapies research in the addictions. Addiction. 2007;102(6):850–62; discussion 63–9.
Hopkins DP, Briss PA, Ricard CJ, et al. Reviews of evidence regarding interventions to reduce tobacco use and exposure to environmental tobacco smoke. Am J Prev Med. 2001;20(2 Suppl):16–66.
Hopkins DP, Husten CG, Fielding JE, Rosenquist JN, Westphal LL. Evidence reviews and recommendations on interventions to reduce tobacco use and exposure to environmental tobacco smoke: A summary of selected guidelines. Am J Prev Med. 2001;20(2 Suppl):67–87.
Glasgow RE, Klesges LM, Dzewaltowski DA, Bull SS, Estabrooks P. The future of health behavior change research: What is needed to improve translation of research into health promotion practice? Ann Behav Med. 2004;27(1):3–12.
Glasgow RE, Orleans CT, Wagner EH. Does the chronic care model serve also as a template for improving prevention? Milbank Q. 2001;79(4):579–612, iv–v.
McLellan A, Lewis, DC, O'Brein, CP, Kleber, HD. Drug dependence, a chronic medical illness: Implications for treatment, insurance, and outcomes evaluation. JAMA. 2000;284(13):1689–95.
Hung DY, Rundall TG, Tallia AF, Cohen DJ, Halpin HA, Crabtree BF. Rethinking prevention in primary care: Applying the chronic care model to address health risk behaviors. Milbank Q. 2007;85(1):69–91.
Kreps GL, Neuhauser L. New directions in eHealth communication: Opportunities and challenges. Patient Educ Couns. 2010;78(3):329–36.
Friend K, Levy DT. Smoking treatment interventions and policies to promote their use: A critical review. Nicotine Tob Res. 2001;3(4):299–310.
Evaluation resources. Available at: www.dhs.ca.gov/tobacco. 2000.
Centers for Disease Control and Prevention. Use of FDA-approved pharmacological treatments for tobacco dependence—United States, 1984–1998. MMWR. 2000;49:665–8.
Rothemich SF, Woolf SH, Johnson RE, et al. Effect on cessation counseling of documenting smoking status as a routine vital sign: An ACORN study. Ann Fam Med. 2008;6(1):60–8.
Fiore MC, Bailey WC, Cohen SJ. Smoking cessation: Clinical practice guideline no. 18. Rockville: U.S. Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research; 1996.
Japuntich SJ, Zehner ME, Smith SS, et al. Smoking cessation via the internet: A randomized clinical trial of an internet intervention as adjuvant treatment in a smoking cessation intervention. Nicotine Tob Res. 2006;8 Suppl 1:S59–67.
Strecher VJ, McClure J, Alexander G, et al. Web-based smoking cessation components and tailoring depth: Results of a randomized trial. Am J Prev Med. 2008;34:373–81.
Kenford SL, Fiore MC, Jorenby DE, Smith SS, Wetter D, Baker TB. Predicting smoking cessation. Who will quit with and without the nicotine patch. JAMA. 1994;271(8):589–94.
Collins LM, Murphy SA, Bierman KL. A conceptual framework for adaptive preventive interventions. Prev Sci. 2004;5(3):185–96.
Murphy SA, Lynch KG, Oslin D, McKay JR, TenHave T. Developing adaptive treatment strategies in substance abuse research. Drug Alcohol Depend. 2007;88 Suppl 2:S24–30.
Acknowledgment
This research was supported by a grant 9P50CA143188-11 from the National Cancer Institute. Dr. Collins was supported via grant P50DA10075, from the National Institute on Drug Abuse. Dr. Baker was supported via NCI 1K05CA139871. Dr. Piper was supported by grant 1UL1RR025011 from the Clinical and Translational Science Award (CTSA) program of the National Center for Research Resources (NCRR), National Institutes of Health (NIH). Dr. Cook was supported by K08DA021311.
Conflicts of Interest
Linda M. Collins, Timothy B. Baker, Robin Mermelstein, Megan E. Piper, Stevens S. Smith, Bruce A. Christiansen, Tanya R. Schlam, and Jessica W. Cook have no potential conflicts of interest to disclose. Douglas E. Jorenby has received research support from the National Institute on Drug Abuse, the National Cancer Institute, Pfizer, Inc., Sanofi-Synthelabo, and Nabi Biopharmaceuticals. He has received support for educational activities from the National Institute on Drug Abuse and the Veterans Administration and consulting fees from Nabi Biopharmaceuticals. Over the last 3 years, Michael C. Fiore served as an investigator on research studies at the University of Wisconsin that were funded by Nabi Biopharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Collins, L.M., Baker, T.B., Mermelstein, R.J. et al. The Multiphase Optimization Strategy for Engineering Effective Tobacco Use Interventions. ann. behav. med. 41, 208–226 (2011). https://doi.org/10.1007/s12160-010-9253-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12160-010-9253-x