Abstract
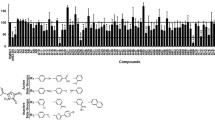
Studies indicate that elevated interleukin-6 (IL-6) levels engage IL6Rα-gp130 receptor complexes to activate signal transducer and activator of transcription 3 (STAT3) that is hyperactivated in many cancers including head and neck squamous cell carcinoma (HNSCC). Our previous HCS campaign identified several hits that selectively blocked IL-6-induced STAT3 activation. This study describes our investigation of the mechanism(s) of action of three of the four chemical series that progressed to lead activities: a triazolothiadiazine (864669), amino alcohol (856350), and an oxazole-piperazine (4248543). We demonstrated that all three blocked IL-6-induced upregulation of the cyclin D1 and Bcl-XL STAT3 target genes. None of the compounds exhibited direct binding interactions with STAT3 in surface plasmon resonance (SPR) binding assays; neither did they inhibit the recruitment and binding of a phospho-tyrosine-gp130 peptide to STAT3 in a fluorescence polarization assay. Furthermore, they exhibited little or no inhibition in a panel of 83 cancer-associated in vitro kinase profiling assays, including lack of inhibition of IL-6-induced Janus kinase (JAK 1, 2, and 3) activation. Further, 864669 and 4248543 selectively inhibited IL-6-induced STAT3 activation but not that induced by oncostatin M (OSM). The compounds 864669 and 4248543 abrogated IL-6-induced phosphorylation of the gp130 signaling subunit (phospho-gp130Y905) of the IL-6-receptor complex in HNSCC cell lines which generate docking sites for the SH2 domains of STAT3. Our data indicate that 864669 and 4248543 block IL-6-induced STAT activation by interfering with the recruitment, assembly, or activation of the hexamer-activated IL-6/IL-6Rα/gp130 signaling complex that occurs after IL-6 binding to IL-6Rα subunits.






Similar content being viewed by others
References
Al Zaid Siddiquee K, Turkson J (2008) STAT3 as a target for inducing apoptosis in solid and hematological tumors. Cell Res 18:254–267. doi:10.1038/cr.2008.18
Avalle L, Pensa S, Regis G, Novelli F, Poli V (2012) STAT1 and STAT3 in tumorigenesis: a matter of balance. JAKSTAT 1:65–72. doi:10.4161/jkst.20045
Brantley EC et al (2008) Loss of protein inhibitors of activated STAT-3 expression in glioblastoma multiforme tumors: implications for STAT-3 activation and gene expression. Clin Cancer Res 14:4694–4704. doi:10.1158/1078-0432.CCR-08-0618
Carpenter JW et al (2002) Configuring radioligand receptor binding assays for HTS using scintillation proximity assay technology. Methods Mol Biol (Clifton, NJ) 190:31–49. doi:10.1385/1-59259-180-9:031
Chang Q et al (2013) The IL-6/JAK/Stat3 feed-forward loop drives tumorigenesis and metastasis. Neoplasia (New York, NY) 15:848–862
Dziennis S, Alkayed NJ (2008) Role of signal transducer and activator of transcription 3 in neuronal survival and regeneration. Rev Neurosci 19:341–361
Frank DA (2007) STAT3 as a central mediator of neoplastic cellular transformation. Cancer Lett 251:199–210. doi:10.1016/j.canlet.2006.10.017
Furqan M, Akinleye A, Mukhi N, Mittal V, Chen Y, Liu D (2013) STAT inhibitors for cancer therapy. J Hematol Oncol 6:90. doi:10.1186/1756-8722-6-90
Gao L et al (2005) Down-regulation of signal transducer and activator of transcription 3 expression using vector-based small interfering RNAs suppresses growth of human prostate tumor in vivo. Clin Cancer Res 11:6333–6341. doi:10.1158/1078-0432.CCR-05-0148
Germain D, Frank DA (2007) Targeting the cytoplasmic and nuclear functions of signal transducers and activators of transcription 3 for cancer therapy. Clin Cancer Res 13:5665–5669. doi:10.1158/1078-0432.CCR-06-2491
Grivennikov S, Karin M (2008) Autocrine IL-6 signaling: a key event in tumorigenesis? Cancer Cell 13:7–9. doi:10.1016/j.ccr.2007.12.020
Guo Y, Xu F, Lu T, Duan Z, Zhang Z (2012) Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat Rev 38:904–910. doi:10.1016/j.ctrv.2012.04.007
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Muller-Newen G, Schaper F (2003) Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J 374:1–20. doi:10.1042/BJ20030407
Heinrich PC, Behrmann I, Muller-Newen G, Schaper F, Graeve L (1998) Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem J 334(Pt 2):297–314
Jing N, Tweardy DJ (2005) Targeting Stat3 in cancer therapy. Anti-Cancer Drugs 16:601–607
Johnson DE (2012) Targeting proliferation and survival pathways in head and neck cancer for therapeutic benefit. Chin J Cancer 31:319–326. doi:10.5732/cjc.011.10404
Johnston PA, Grandis JR (2011) STAT3 signaling: anticancer strategies and challenges. Mol Interv 11:18–26. doi:10.1124/mi.11.1.4
Johnston PA, Sen M, Hua Y, Camarco D, Shun TY, Lazo JS, Grandis JR (2014) High-content pSTAT3/1 imaging assays to screen for selective inhibitors of STAT3 pathway activation in head and neck cancer cell lines. Assay Drug Dev Technol 12:55–79. doi:10.1089/adt.2013.524
Johnston PA et al (2015) HCS campaign to identify selective inhibitors of IL-6-induced STAT3 pathway activation in head and neck cancer cell lines. Assay Drug Dev Technol 13:356–376. doi:10.1089/adt.2015.663
Jones SA, Scheller J, Rose-John S (2011) Therapeutic strategies for the clinical blockade of IL-6/gp130 signaling. J Clin Invest 121:3375–3383. doi:10.1172/JCI57158
Jung IH, Choi JH, Chung YY, Lim GL, Park YN, Park SW (2015) Predominant activation of JAK/STAT3 pathway by interleukin-6 is implicated in hepatocarcinogenesis. Neoplasia (New York, NY) 17:586–597. doi:10.1016/j.neo.2015.07.005
Konnikova L, Kotecki M, Kruger MM, Cochran BH (2003) Knockdown of STAT3 expression by RNAi induces apoptosis in astrocytoma cells. BMC Cancer 3:23. doi:10.1186/1471-2407-3-23
LaPorte M, Wang, Z., Colombo, R., Garzan, A., Peshkov, VA., Liang, M., Johnston, PA., Schurdak, ME., Sen, M., Camarco, DP., Hua, Y., Pollock, NI., Lazo, JS., Grandis, JR., Wipf, P., Huryn, DM. (2016) Optimization of pyrazole-containing 1,2,4-triazolo-[3,4-b] thiadiazines, a new class of STAT3 pathway inhibitors Bioorganic & medicinal chemistry letters
LaPorte MG et al (2014) 2-Guanidinoquinazolines as new inhibitors of the STAT3 pathway. Bioorg Med Chem Lett 24:5081–5085. doi:10.1016/j.bmcl.2014.09.001
Leeman RJ, Lui VW, Grandis JR (2006) STAT3 as a therapeutic target in head and neck cancer. Expert Opin Biol Ther 6:231–241
Li H, Xiao H, Lin L, Jou D, Kumari V, Lin J, Li C (2014) Drug design targeting protein-protein interactions (PPIs) using multiple ligand simultaneous docking (MLSD) and drug repositioning: discovery of raloxifene and bazedoxifene as novel inhibitors of IL-6/GP130 interface. J Med Chem 57:632–641. doi:10.1021/jm401144z
Ling X, Arlinghaus RB (2005) Knockdown of STAT3 expression by RNA interference inhibits the induction of breast tumors in immunocompetent mice. Cancer Res 65:2532–2536. doi:10.1158/0008-5472.CAN-04-2425
Lui VW et al (2014) Frequent mutation of receptor protein tyrosine phosphatases provides a mechanism for STAT3 hyperactivation in head and neck cancer. Proc Natl Acad Sci U S A 111:1114–1119. doi:10.1073/pnas.1319551111
Mihara M, Hashizume M, Yoshida H, Suzuki M, Shiina M (2012) IL-6/IL-6 receptor system and its role in physiological and pathological conditions. Clin Sci (Lond) 122:143–159. doi:10.1042/CS20110340
Mitsuyama K, Matsumoto S, Masuda J, Yamasakii H, Kuwaki K, Takedatsu H, Sata M (2007) Therapeutic strategies for targeting the IL-6/STAT3 cytokine signaling pathway in inflammatory bowel disease. Anticancer Res 27:3749–3756
Nagasaki T, Hara M, Nakanishi H, Takahashi H, Sato M, Takeyama H (2014) Interleukin-6 released by colon cancer-associated fibroblasts is critical for tumour angiogenesis: anti-interleukin-6 receptor antibody suppressed angiogenesis and inhibited tumour-stroma interaction. Br J Cancer 110:469–478. doi:10.1038/bjc.2013.748
Nguyen DP, Li J, Tewari AK (2014) Inflammation and prostate cancer: the role of interleukin 6 (IL-6). BJU Int 113:986–992. doi:10.1111/bju.12452
Park OK, Schaefer TS, Nathans D (1996) In vitro activation of Stat3 by epidermal growth factor receptor kinase. Proc Natl Acad Sci U S A 93:13704–13708
Peyser ND, Freilino M, Wang L, Zeng Y, Li H, Johnson DE, Grandis JR (2016) Frequent promoter hypermethylation of PTPRT increases STAT3 activation and sensitivity to STAT3 inhibition in head and neck cancer. Oncogene 35:1163–1169. doi:10.1038/onc.2015.171
Schafer ZT, Brugge JS (2007) IL-6 involvement in epithelial cancers. J Clin Invest 117:3660–3663. doi:10.1172/JCI34237
Schaper F et al (1998) Activation of the protein tyrosine phosphatase SHP2 via the interleukin-6 signal transducing receptor protein gp130 requires tyrosine kinase Jak1 and limits acute-phase protein expression. Biochem J 335(Pt 3):557–565
Schust J, Sperl B, Hollis A, Mayer TU, Berg T (2006) Stattic: a small-molecule inhibitor of STAT3 activation and dimerization. Chem Biol 13:1235–1242
Sen M et al (2014) Systemic administration of a cyclic signal transducer and activator of transcription 3 (STAT3) decoy oligonucleotide inhibits tumor growth without inducing toxicological effects. Mol Med 20:46–56. doi:10.2119/molmed.2013.00104
Sen M et al (2012) First-in-human trial of a STAT3 decoy oligonucleotide in head and neck tumors: implications for cancer therapy. Cancer Discov 2:694–705. doi:10.1158/2159-8290.CD-12-0191
Shariat SF, Andrews B, Kattan MW, Kim J, Wheeler TM, Slawin KM (2001) Plasma levels of interleukin-6 and its soluble receptor are associated with prostate cancer progression and metastasis. Urology 58:1008–1015
Siddiquee K et al (2007) Selective chemical probe inhibitor of Stat3, identified through structure-based virtual screening, induces antitumor activity. Proc Natl Acad Sci U S A 104:7391–7396. doi:10.1073/pnas.0609757104
Stanam A, Love-Homan L, Joseph TS, Espinosa-Cotton M, Simons AL (2015) Upregulated interleukin-6 expression contributes to erlotinib resistance in head and neck squamous cell carcinoma. Mol Oncol 9:1371–1383. doi:10.1016/j.molonc.2015.03.008
Taga T, Kawanishi Y, Hardy RR, Hirano T, Kishimoto T (1987) Receptors for B cell stimulatory factor 2. Quantitation, specificity, distribution, and regulation of their expression. J Exp Med 166:967–981
Tawara K, Oxford JT, Jorcyk CL (2011) Clinical significance of interleukin (IL)-6 in cancer metastasis to bone: potential of anti-IL-6 therapies. Cancer Manag Res 3:177–189. doi:10.2147/CMR.S18101
Turkson J et al (2001) Phosphotyrosyl peptides block Stat3-mediated DNA binding activity, gene regulation, and cell transformation. J Biol Chem 276:45443–45455. doi:10.1074/jbc.M107527200
Vultur A et al (2004) Cell-to-cell adhesion modulates Stat3 activity in normal and breast carcinoma cells. Oncogene 23:2600–2616. doi:10.1038/sj.onc.1207378
Waldner MJ, Foersch S, Neurath MF (2012) Interleukin-6—a key regulator of colorectal cancer development. Int J Biol Sci 8:1248–1253. doi:10.7150/ijbs.4614
Wang SW, Sun YM (2014) The IL-6/JAK/STAT3 pathway: potential therapeutic strategies in treating colorectal cancer (review). Int J Oncol 44:1032–1040. doi:10.3892/ijo.2014.2259
Wang YZ, Wharton W, Garcia R, Kraker A, Jove R, Pledger WJ (2000) Activation of Stat3 preassembled with platelet-derived growth factor beta receptors requires Src kinase activity. Oncogene 19:2075–2085. doi:10.1038/sj.onc.1203548
Wilks AF (2008) The JAK kinases: not just another kinase drug discovery target. Semin Cell Dev Biol 19:319–328. doi:10.1016/j.semcdb.2008.07.020
Yadav A, Kumar B, Datta J, Teknos TN, Kumar P (2011) IL-6 promotes head and neck tumor metastasis by inducing epithelial-mesenchymal transition via the JAK-STAT3-SNAIL signaling pathway. Mol Cancer Res: MCR 9:1658–1667. doi:10.1158/1541-7786.MCR-11-0271
Yue P, Turkson J (2009) Targeting STAT3 in cancer: how successful are we? Expert Opin Investig Drugs 18:45–56
Zhang HF, Lai R (2014) STAT3 in cancer-friend or foe? Cancers (Basel) 6:1408–1440. doi:10.3390/cancers6031408
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding sources
This project has been funded in part with Federal Funds from the National Cancer Institute, National Institutes of Health, under Contract No. HSN261200800001E. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government. This study was supported by NExT-CBC Project ID #1015, S08-221 Task Order 6 “STAT3 Pathway Inhibitor HCS” (Grandis, PI), NCI Chemical Biology Consortium, Pittsburgh Specialized Application Center (PSAC) (Lazo and Johnston co-PIs), and University of Pittsburgh Chemical Diversity Center (Huryn, PI). The project was also supported in part by funds from the American Cancer Society (Grandis) and a Head and Neck Spore P50 award (Grandis, CA097190).
Rights and permissions
About this article
Cite this article
Sen, M., Johnston, P.A., Pollock, N.I. et al. Mechanism of action of selective inhibitors of IL-6 induced STAT3 pathway in head and neck cancer cell lines. J Chem Biol 10, 129–141 (2017). https://doi.org/10.1007/s12154-017-0169-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12154-017-0169-9