Abstract
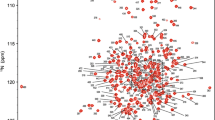
Polyketide cyclase from Mycobacterium tuberculosis (MtPC) is related to the formation of sterol derivatives, which may play a role in immune escape in the initial stage of macrophage infection by Mycobacterium tuberculosis. However, the structure and specific functions of MtPC are still unknown. Here we report the backbone and side-chain NMR resonance assignments for the MtPC. Most resonances were assigned and the secondary structure was predicted according to the assigned backbone resonances by TALOS-N and PECAN. These NMR assignments represent a first step towards researching the structure and function of MtPC.



Similar content being viewed by others
References
Cherney MM, Garen CR, James MN (2008) Crystal structure of Mycobacterium tuberculosis Rv0760c at 1.50 A resolution, a structural homolog of Delta(5)-3-ketosteroid isomerase. Biochim Biophys Acta 1784:1625–1632. https://doi.org/10.1016/j.bbapap.2008.05.012
Delaglio F, Grzesiek S, Vuister GW, Zhu G, Pfeifer J, Bax A (1995) NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J Biomol NMR 6:277–293. https://doi.org/10.1007/BF00197809
Eghbalnia HR, Wang L, Bahrami A, Assadi A, Markley JL (2005) Protein energetic conformational analysis from NMR chemical shifts (PECAN) and its use in determining secondary structural elements. J Biomol NMR 32:71–81. https://doi.org/10.1007/s10858-005-5705-1
Filippova EV, Luan C-H, Dunne SF, Kiryukhina O, Minasov G, Shuvalova L, Anderson WF (2014) Structural characterization of a hypothetical protein: a potential agent involved in trimethylamine metabolism in Catenulispora acidiphila. J Struct Funct Genomics 15:33–40. https://doi.org/10.1007/s10969-014-9176-z
Fritzsche K, Ishida K, Hertweck C (2008) Orchestration of discoid polyketide cyclization in the resistomycin pathway. J Am Chem Soc 130:8307–8316. https://doi.org/10.1021/ja800251m
Gomez JE, McKinney JD (2004) M. Tuberculosis persistence, latency, and drug tolerance. Tuberculosis (edinb) 84:29–44. https://doi.org/10.1016/j.tube.2003.08.003
Gullón S, Olano C, Abdelfattah MS, Braña AF, Rohr J, Méndez C, Salas JA (2006) Isolation, characterization, and heterologous expression of the biosynthesis gene cluster for the antitumor anthracycline steffimycin. Appl Environ Microbiol 72:4172–4183. https://doi.org/10.1128/AEM.00734-06
Iyer LM, Koonin EV, Aravind L (2001) Adaptations of the Helix-Grip fold for ligand binding and catalysis in the START domain superfamily. Proteins 43:134–144. https://doi.org/10.1002/1097-0134(20010501)43:2%3c134::Aid-prot1025%3e3.0.Co;2-i
Kallio P, Sultana A, Niemi J, Mäntsälä P, Schneider G (2006) Crystal structure of the polyketide cyclase AknH with bound substrate and product analogue: implications for catalytic mechanism and product stereoselectivity. J Mol Biol 357:210–220. https://doi.org/10.1016/j.jmb.2005.12.064
Künzel E et al (1997) Tetracenomycin M, a novel genetically engineered tetracenomycin resulting from a combination of mithramycin and tetracenomycin biosynthetic genes. Chemistry 3:1675–1678. https://doi.org/10.1002/chem.19970031017
Lamb DC, Kelly DE, Manning NJ, Kelly SL (1998) A sterol biosynthetic pathway in Mycobacterium. FEBS Lett 437:142–144. https://doi.org/10.1016/s0014-5793(98)01218-6
Lee W, Tonelli M, Markley JL (2014) NMRFAM-SPARKY: enhanced software for biomolecular NMR spectroscopy. Bioinformatics 31:1325–1327
Sb W (1988) NMR of proteins and nucleic acids: Kurt Wüthrich. Wiley-Interscience, New York. 1986. 292 pages. $49.95. J Magnet Reson 79:586–586
Schubert M, Labudde D, Oschkinat H, Schmieder P (2002) A software tool for the prediction of Xaa-Pro peptide bond conformations in proteins based on 13C chemical shift statistics. J Biomol NMR 24:149–154. https://doi.org/10.1023/A:1020997118364
Shen Y, Bax A (2010) Prediction of Xaa-Pro peptide bond conformation from sequence and chemical shifts. J Biomol NMR 46:199–204. https://doi.org/10.1007/s10858-009-9395-y
Shen Y, Bax A (2013) Protein backbone and sidechain torsion angles predicted from NMR chemical shifts using artificial neural networks. J Biomol NMR 56:227–241. https://doi.org/10.1007/s10858-013-9741-y
Silvennoinen L, Sandalova T, Schneider G (2009) The polyketide cyclase RemF from Streptomyces resistomycificus contains an unusual octahedral zinc binding site. FEBS Lett 583:2917–2921. https://doi.org/10.1016/j.febslet.2009.07.061
Sultana A, Kallio P, Jansson A, Wang J-S, Niemi J, Mäntsälä P, Schneider G (2004) Structure of the polyketide cyclase snoal reveals a novel mechanism for enzymatic aldol condensation. EMBO J 23:1911–1921. https://doi.org/10.1038/sj.emboj.7600201
Thompson TB, Katayama K, Watanabe K, Hutchinson CR, Rayment I (2004) Structural and functional analysis of tetracenomycin F2 cyclase from Streptomyces glaucescens. A type II polyketide cyclase. J Biol Chem 279:37956–37963. https://doi.org/10.1074/jbc.M406144200
Ying J, Delaglio F, Torchia DA, Bax A (2017) Sparse multidimensional iterative lineshape-enhanced (SMILE) reconstruction of both non-uniformly sampled and conventional NMR data. J Biomol NMR 68:101–118. https://doi.org/10.1007/s10858-016-0072-7
Zheng S, Zhou Y, Fleming J, Zhou Y, Liu W, Bi L (2017) The putative polyketide cyclase MSMEG_0129 from Mycobacterium smegmatis: purification, crystallization and X-ray crystallographic analysis. Acta Crystallogr F 73:437–442. https://doi.org/10.1107/S2053230X17008937
Acknowledgements
This work was supported by the National Key Research and Development Project of China (No. 2016YFA0500600). All the NMR experiments were performed at the High‐Field Nuclear Magnetic Resonance Centre of Xiamen University
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fan, S., Zhuang, J., Guo, C. et al. 1H, 13C, 15N backbone and side-chain chemical shift assignments of the polyketide cyclase from Mycobacterium tuberculosis. Biomol NMR Assign 15, 397–402 (2021). https://doi.org/10.1007/s12104-021-10036-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-021-10036-5