Abstract
Purpose
We evaluated the prevalence of immune-related adverse events and anti-tumor efficacy in advanced/metastatic urothelial carcinoma following immune-checkpoint inhibitors (ICIs) treatment.
Methods
We conducted a multicenter retrospective study of patients with advanced/metastatic urothelial carcinoma treated with ICIs in four Spanish institutions. irAEs were classified using Common Terminology Criteria for Adverse Event (CTCAE) v.5.0 guidelines. The primary endpoint was overall survival (OS). Other endpoints were overall response rate (ORR) and progression-free survival (PFS). irAEs were evaluated as a time-dependent covariate to avoid immortal time bias.
Results
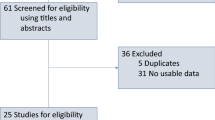
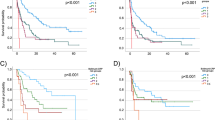
A total of 114 patients were treated with ICIs between May 2013 and May 2019, 105 (92%) of whom received ICIs as monotherapy. irAEs of any grade were experienced in 56 (49%) patients and 21 (18%) patients had grade ≥ 3 toxicity. The most frequent irAEs were gastrointestinal and dermatological toxicities, reported in 25 (22%) and 20 (17%) patients, respectively. Patients with grade 1–2 irAEs had significantly longer OS compared to those without grade 1–2 irAEs (median 18.2 vs. 8.7 months, HR = 0.61 [95% CI 0.39–0.95], p = 0.03). No association with efficacy was observed for patients with grade ≥ 3 irAEs. No difference in PFS was observed after adjusting for the immortal time bias. ORR was higher in patients who developed irAEs (48% vs 17%, p < 0.001).
Conclusions
Our findings suggest that development of irAEs was associated with higher ORR, and patients who developed grade 1–2 irAEs had longer OS. Prospective studies are necessary to confirm our findings.



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, [RMB], upon reasonable request.
References
Bellmunt J, de Wit R, Vaughn D, Fradet Y, Lee JY, Fong L, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med. 2017;376:1015–26.
Powles T, Durán I, van der Heijden MS, Loriot Y, Vogelzang NJ, De Giorgi U, et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastastic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet. 2018;391:748–57.
Sharma P, Retz M, Siefker-Radtke A, Baron A, Necchi A, Bedke J, et al. Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate275): a multicentre single-arm, phase 2 trial. Lancet Oncol. 2017;18:312–22.
Powles T, O’Donnell PH, Massard C, Arkenau HT, Friedlander TW, Hoimes CJ, et al. Efficacy and safety of durvalumab in locally advanced or metastatic urothelial carcinoma: updated results from a phase 1/2 open-label study. JAMA Oncol. 2017;3: e172411.
Apolo AB, Infante JR, Balmanoukian A, Patel MR, Wang D, Kelly K, et al. Avelumab, an anti-programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter phase Ib study. J Clin Oncol. 2017;35:2117–24.
Balar AV, Castellano D, O’Donnell PH, Grivas P, Vuky J, Powles T, et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): a multicentre. Single-arm, phase 2 study. Lancet Oncol. 2017;18:1483–92.
Balar AV, Castellano D, O’Donnell PH, Grivas P, Vuky J, Powles T, et al. Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: a single-arm, multicentre, phase 2 trial. Lancet. 2017;389:67–76.
Powles T, Park SH, Voog E, Caserta C, Valderrama BP, Gurney H, et al. Avelumab maintenance therapy for advanced or metastatic urothelial carcinoma. N Engl J Med. 2020;383:1218–30.
Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378:158–68.
Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, et al. ESMO guidelines management of toxicities from immunotherapy: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(suppl 4):iv119–42.
Kamat AM, Bellmunt J, Galsky MD, Konety BR, Lamm DL, Langham D, et al. Society of immunotherapy of cancer consensus statement on immunotherapy for the treatment of bladder carcinoma. J Immunother Cancer. 2017;5:68.
Das S, Johnson DB. Immune-related adverse events and anti-tumor efficacy of immune checkpoint inhibitors. J Immunother Cancer. 2019;7:306.
Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primers. 2020;6:38.
Maher VE, Fernandes LL, Weinstock C, Tang S, Agarwal S, Brave M, et al. Analysis of the association between adverse events and outcomes in patients receiving a programmed death protein 1 or programmed death ligand 1 antibody. J Clin Oncol. 2019;37:2730–7.
Ma VT, Su CT, Hu M, Taylor JMG, Daignault-Newton S, Kellezi O, et al. Characterization of outcomes in patients with advanced genitourinary malignancies treated with immune checkpoint inhibitors. Urol Oncol. 2021;39(437):e1-437.e9.
Suissa S. Immortal time bias in pharmaco-epidemiology. Am J Epidemiol. 2008;167:492–9.
Schulz GB, Rodler S, Szabados B, Graser A, Buchner A, Stief C, et al. Safety, efficacy and prognostic impact of immune checkpoint inhibitors in older patients with genitourinary cancers. J Geriatr Oncol. 2020;11:1061–6.
Tang SQ, Tang LL, Mao YP, Li WF, Chen L, Zhang Y, et al. The pattern of time to onset and resolution of immune-related adverse events caused by immune checkpoint inhibitors in cancer: a pooled analysis of 23 clinical trials and 8436 patients. Cancer Res Treat. 2021;53:339–54.
Shabto JM, Martini D, Liu Y, Ravindranathan D, Kline MR, Hitron E, et al. Association between immune-related adverse events and clinical outcomes in advanced urothelial cancer patients treated with immunotherapy. J Clin Oncol. 2019;37(7 suppl):472.
Park R, Lopes L, Saeed A. Anti-PD-1/L1-associated immune-related adverse events as harbinger of favorable clinical outcome: systematic review and meta-analysis. Clin Transl Oncol. 2021;23:100–9.
Hussaini S, Chehade R, Boldt RG, Raphael J, Blanchette P, Maleki Vareki S, et al. Association between immune-related side effects and efficacy and benefit of immune checkpoint inhibitors - a systematic review and meta-analysis. Cancer Treat Rev. 2021;92: 102134.
Powles T, van der Heijden MS, Castellano D, Galsky MD, Loriot Y, Petrylak DP, et al. Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2020;21:1574–88.
Powles T, Csőszi T, Özgüroğlu M, Matsubara N, Géczi L, Cheng SY, et al. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22:931–45.
Fan Y, Xie W, Huang H, Wang Y, Li G, Geng Y, et al. Association of immune related adverse events with efficacy of immune checkpoint inhibitors and overall survival in cancers: a systemic review and meta-analysis. Front Oncol. 2021;11:633032. https://doi.org/10.3389/fonc.2021.633032.
Fucà G, Galli G, Poggi M, Lo Russo G, Proto C, Imbimbo M, et al. Modulation of peripheral blood immune cells by early use of steroids and its association with clinical outcomes in patients with metastatic non-small cell lung cancer treated with immune checkpoint inhibitors. ESMO Open. 2019;4:e000457. https://doi.org/10.1136/esmoopen-2018-000457.
Petrelli F, Signorelli D, Ghidini M, Ghidini A, Pizzutilo EG, Ruggieri L, et al. Association of steroids use with survival in patients treated with immune checkpoint inhibitors: a systematic review and meta-analysis. Cancers. 2020;12:546. https://doi.org/10.3390/cancers12030546.
Ricciuti B, Dahlberg SE, Adeni A, Sholl LM, Nishino M, Awad MM. Immune checkpoint inhibitor outcomes for patients with non-small-cell lung cancer receiving baseline corticosteroids for palliative versus nonpalliative indications. J Clin Oncol. 2019;37:1927–34.
Gulati N, Celen A, Johannet P, Mehnert JM, Weber J, Krogsgaard M, et al. Preexisting immune-mediated inflammatory disease is associated with improved survival and increased toxicity in melanoma patients who receive immune checkpoint inhibitors. Cancer Med. 2021;10:7457–65.
Acknowledgements
The authors wish to thank the patients who kindly participated in our study and their families. The authors also thank Sarah MacKenzie PhD for providing medical writing assistance.
Funding
This work was supported by the Vall d´Hebron Institute of Oncology. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Study Concept and design: RM-B and JC. Acquisition of data; ND, MG, NV, MF and JG. Organization of the data: GV. Analysis of the database: RM-B and GV. Interpretation of data: All authors. Drafting of the manuscript: All authors. Critical revision of the manuscript for important intellectual content: All authors. Statistical analysis: GV. Obtaining funding: None.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare the following financial interests/personal. RMB: (all unrelated in the last 3 years): Consulting or advisory and/or speakers bureaus for Sanofi Aventis, AstraZeneca, Merck Sharp & Dohme, Astellas, BMS and received travel and accommodations expenses from Roche, Sanofi Aventis, Astellas, Janssen, Merck Sharp & Dohme, Bayer, and Pfizer. GV: has received research honoraria for speaker activities from MSD and advisory role from AstraZeneca. NV: Speaker bureaus for Sanofi, AstraZeneca, Astellas, Janssen, Roche, MSD, Bristol. MF: Advisory and/or speakers bureaus for BMS, Ipsen, Merck and Pfizer. JG: nothing to declare. TB: Advisory and/or speakers bureaus for Merck, Roche, BMS, Pfizer, Astellas and received travel and accommodations expenses from Roche, MSD, Ipsen, Merck and Bayer. CS: Consulting or advisory and/or speakers bureaus for Astellas, Bayer, BMS, Roche, Ipsen, Merck Sharp & Dohme, Novartis, Pfizer, Sanofi Aventis. ND: Travel and accommodations expenses from Sanofi Aventis and Pfizer. JM: has received honoraria and/or acted as consulting or advisory role for AstraZeneca, Roche, MSD, Amgen and Pfizer; JM is also the PI of grants by AstraZeneca and Pfizer to VHIO as institution. MG: Travel and accommodations expenses from Roche, Astellas, Ipsen and Pfizer. MD: Consulting or advisory and/or speakers bureaus for Sanofi Aventis, Bristol-Myers Squibb, Pfizer and received travel and accommodations expenses from Sanofi and Lilly. JP: Honoraria from Pfizer, Bristol-Myers Squibb, Ipsen, AstraZeneca, Roche, MSD Oncology, Janssen-Cilag, Astellas Pharma, EUSA Pharma, Eisai, Pierre Fabre, Sanofi, and Bayer; consulting or advisory role from Pfizer, Astellas Pharma, Janssen-Cilag, Merck Sharpe & Dohme, Bayer, Roche, Bristol-Myers Squibb, Clovis Oncology, Ipsen, Eisai, and Sanofi; research funding from Astellas Pharma and Pfizer; travel/accommodations/expenses from Pfizer, Roche, Janssen-Cilag, Bristol-Myers Squibb, and MSD Oncology. JC: Consulting: Astellas Pharma, AstraZeneca, Bayer, Bristol-Myers Squibb, Johnson & Johnson, MSD Oncology, Novartis (AAA), Pfizer, Roche, Sanofi. Speakers bureaus: Bayer, Asofarma, Astellas, Janssen Fundings / Grants to out institution: AB Science, Aragon Pharmaceuticals, Arog Pharmaceuticals, INC, Astellas Pharma., AstraZeneca AB, Aveo Pharmaceuticals INC, Bayer AG, Blueprint Medicines Corporation, BN Immunotherapeutics INC, Boehringer Ingelheim España, S.A., Bristol-Myers Squibb International Corporation (BMS), Clovis Oncology, INC, Cougar Biotechnology INC, Deciphera Pharmaceuticals LLC, Exelixis INC, F. Hoffmann-La Roche LTD, Genentech INC, Glaxosmithkline, SA, Incyte Corporation, Janssen-Cilag International NV, Karyopharm Therapeutics INC., Laboratories Leurquin Mediolanum SAS, Lilly, S.A., Medimmune, Millennium Pharmaceuticals, INC., Nanobiotix SA, Novartis Farmacéutica, S.A., Pfizer, S.L.U, Puma Biotechnology, INC, Sanofi-Aventis, S.A., SFJ Pharma LTD. II, Teva Pharma S.L.U.
Ethical approval (Research involving human participants and/or animals), Informed consent
The study was approved by the Ethics Committee at Vall d’Hebron University Hospital (PR[AG]490/2021). All the patients who were alive at the time of the analysis signed the informed consent form.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
See Table 3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morales-Barrera, R., Villacampa, G., Vidal, N. et al. Prevalence of immune-related adverse events and anti-tumor efficacy in advanced/metastatic urothelial carcinoma following immune-checkpoint inhibitor treatment. Clin Transl Oncol 25, 3556–3564 (2023). https://doi.org/10.1007/s12094-023-03213-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03213-6