Abstract
Introduction
Cancer patients receiving chemotherapy are a high risk of VTE, yet the importance of thromboprophylaxis for cancer patients that are at high risk of developing VTE is still controversial.
Aim
To calculate the benefits and harms of thromboprophylaxis, compared to placebo, in ambulatory high-risk cancer patients that are receiving chemotherapy.
Methods
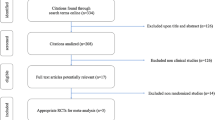
We searched PubMed, Embase, Web of Science, the Cochrane Library, Cochrane Central Register of Controlled Trials, Chinese Biomedical Literature Database, WANFANG Data, Chinese National Knowledge Infrastructure and Chinese Scientific Journal Database for randomized controlled trials (RCTs) describing benefits and harms of thromboprophylaxis. Statistical analysis was performed using Stata software (version 15.1).
Results
We included six studies, which contained a total of 3240 cancer patients with thromboprophylaxis and 2874 cancer patients without thromboprophylaxis. Thromboprophylaxis was effective in high-risk patients with two points or higher (RR 0.51, 95% CI 0.36–0.71, I2 = 0.0%, P = 0.526). It was associated with an increase in bleeding events (RR 1.65, 95% CI 1.14–2.40, I2 = 0.0%, P = 0.498) and was mainly efficient in reducing the risk of pulmonary embolism (RR 0.56, 95% CI 0.33–0.96, I2 = 0.0%, P = 0.263). The risk of major (RR 1.85, 95% CI 0.87–3.94, I2 = 0.0%, P = 0.888) and non-major (RR 1.59, 95% CI 0.96–2.62, I2 = 16.3%, P = 0.303) bleeding showed no significant difference with or without thromboprophylaxis. There was no reduction in all-cause mortality with thromboprophylaxis (RR 0.95, 95% CI 0.78–1.18, I2 = 22.0%, P = 0.277).
Conclusion
Thromoboprophylaxis is effective and safe in cancer patients that are at high risk for developing VTE with chemotherapy.






Similar content being viewed by others
References
Khorana AA, Francis CW, Culakova E, et al. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost. 2007;5:632–4.
Khorana AA, Dalal MR, Lin J, et al. Health care costs associated with venous thromboembolism in selected high-risk ambulatory patients with solid tumors undergoing chemotherapy in the USA. Clinicoecon Outcomes Res. 2013;5:101–8.
Zangari M, Fink LM, Elice F, et al. Thrombotic events in patients with cancer receiving antiangiogenesis agents. J Clin Oncol. 2009;27:4865–73.
Lyman GH, Khorana AA, Falanga A, et al. American society of clinical oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol. 2007;25:5490–505.
National Comprehensive Cancer Network. (NCCN) Clinical practice guidelines in oncology:cancer-associated venous thromboembolic disease. Version 1. 2018. https://www.nccn.org/professionals/physician_gls/default.aspx#vte. Accessed 22 Mar 2018.
Mulder FI, Candeloro M, Kamphuisen PW, et al. The Khorana score for prediction of venous thromboembolism in cancer patients: a systematic review and meta-analysis. Haematologica. 2019;104(6):1277–87.
Carrier M, Abou-Nassar K, Mallick R, et al. Apixaban to prevent venous thromboembolism in patients with cancer. N Engl J Med. 2019;380:711–9.
Khorana AA, Soff GA, Kakkar AK, et al. Rivaroxaban for thromboprophylaxis in high-risk ambulatory patients with cancer. N Engl J Med. 2019;380:720–8.
Khorana AA, Carrier M, Garcia DA, et al. Guidance for the prevention and treatment of cancer-associated venous thromboembolism. J Thromb Thrombolysis. 2016;41:81–91.
Lyman GH, Bohlke K, Khorana AA, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society Of Clinical Oncology clinical practice guideline update 2014. J Clin Oncol. 2015;33:654–6.
Tian J, Zhang J, Ge L, et al. The methodological and reporting quality of systematic reviews from China and the USA are similar. J Clin Epidemiol. 2017;85:50–8.
Yao L, Sun R, Chen YL, et al. The quality of evidence in Chinese meta-analyses needs to be improved. J Clin Epidemiol. 2016;74:73–9.
Xiu-xia L, Ya Z, Yao-long C, et al. The reporting characteristics and methodological quality of Cochrane reviews about health policy research. Health Policy. 2015;119:503–10.
Yan P, Yao L, Li H, et al. The methodological quality of robotic surgical meta-analyses needed to be improved: a cross-sectional study. J Clin Epidemiol. 2019;109:20–9.
Ge L, Tian JH, Li YN, et al. Association between prospective registration and overall reporting and methodological quality of systematic reviews: a meta-epidemiological study. J Clin Epidemiol. 2018;93:45–55.
Wang X, Chen Y, Yao L, et al. Reporting of declarations and conflicts of interest in WHO guidelines can be further improved. J Clin Epidemiol. 2018;98:1–8.
George D, Agnelli G, Fisher W, et al. Venous thromboembolism (VTE) prevention with semuloparin in cancer patients initiating chemotherapy: benefit–risk assessment by VTE risk in SAVE-ONCO. Blood. 2011;118:206.
Khorana A, Francis C, Kuderer N, et al. Dalteparin thromboprophylaxis in cancer patients at high risk for venous thromboembolism: a randomized trial. Thromb Res. 2017;151:89–99.
Verso M, Agnelli G, Barni S, et al. A modified Khorana risk assessment score for venous thromboembolism in cancer patients receiving chemotherapy: the protecht score. Intern Emerg Med. 2012;7:291–2.
Fei ZXJDLJLYZHZ, et al Early anticoagulant intervention in malignant tumor patients receiving chemothrapy at low-moderate thrombus risk. Tumor. 2016;36:1151–1155
Agnelli G, George DJ, Kakkar AK, et al. Semuloparin for thromboprophylaxis in patients receiving chemotherapy for cancer. N Engl J Med. 2012;366:601–9.
Gould MK, Dembitzer AD, Doyle RL, et al. Low-molecular-weight heparins compared with unfractionated heparin for treatment of acute deep venous thrombosis. A meta-analysis of randomized, controlled trials. Ann Intern Med. 1999;130:800–9.
Sanford D, Naidu A, Alizadeh N, et al. The effect of low molecular weight heparin on survival in cancer patients: an updated systematic review and meta-analysis of randomized trials. J Thromb Haemost. 2014;12:1076–85.
Akl EA, Kahale LA, Hakoum MB, et al. Parenteral anticoagulation in ambulatory patients with cancer. Cochrane Database Syst Rev. 2017;9:Cd006652.
Kinnunen PTT, Murtola TJ, Talala K, et al. Anticoagulants and cancer mortality in the Finnish randomized study of screening for prostate cancer. Cancer Causes Control. 2019;30:877–88.
Levine MN, Gu C, Liebman HA, et al. A randomized phase II trial of apixaban for the prevention of thromboembolism in patients with metastatic cancer. J Thromb Haemost. 2012;10:807–14.
van Es N, Franke VF, Middeldorp S, et al. The Khorana score for the prediction of venous thromboembolism in patients with pancreatic cancer. Thromb Res. 2017;150:30–2.
Martin AJM, Alfonso PG, Blanco ABR, et al. Incidence of venous thromboembolism (VTE) in ambulatory pancreatic cancer patients receiving chemotherapy and analysis of Khorana’s predictive model. Clin Transl Oncol. 2014;16:927–30.
Rupa-Matysek J, Gil L, Kazmierczak M, et al. Prediction of venous thromboembolism in newly diagnosed patients treated for lymphoid malignancies: validation of the Khorana risk score. Med Oncol. 2017;35:5.
Pishko AM, Smith KJ, Ragni MV. Anticoagulation in ambulatory cancer patients with no indication for prophylactic or therapeutic anticoagulation: a cost-effectiveness analysis from a USA perspective. Thromb Haemost. 2012;108:303–10.
Funding
This work was not sponsored by any findings.
Author information
Authors and Affiliations
Contributions
KHY and LMT was involved in the design of the study. YB and BG collected the data. YB and BG performed the meta-analysis and wrote the first draft of the manuscript, which was critically revised by YB, BG, PJY, LMT and KHY.
Corresponding authors
Ethics declarations
Conflict of interest
None of the authors has anything to declare. The views expressed are those of the authors.
Ethical approval
This meta-analysis was based on a literature review and modeling techniques, so this study did not require approval by an Institutional Research Ethics Board.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Data sharing statement
No additional data are available.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Rights and permissions
About this article
Cite this article
Bao, Y., Gao, B., Yan, P. et al. The effectiveness and safety of thromboprophylaxis in cancer patients based on Khorana score: a meta-analysis and systematic review of randomized controlled trials. Clin Transl Oncol 22, 1992–2001 (2020). https://doi.org/10.1007/s12094-020-02336-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02336-4