Abstract
The bone marrow (BM) microenvironment (niche) is abnormally altered in acute myeloid leukemia (AML), leading to deficient secretion of proteins, soluble factors, and cytokines by mesenchymal stromal cells (MSC) that modifies the crosstalk between MSC and hematopoietic cells. We focused on a WNT gene/protein family member, WNT5A, which is downregulated in leukemia and correlated with disease progression and poor prognosis. We demonstrated that WNT5A protein upregulated the WNT non-canonical pathway only in leukemic cells, without modulating normal cell behavior. We also introduced a novel WNT5A-mimicking compound, Foxy-5. Our results showed reduction of crucial biological functions that are upregulated in leukemia cells, including ROS generation, cell proliferation, and autophagy, as well as G0/G1 cell cycle arrest. Additionally, Foxy-5 induced early-stage macrophage cell differentiation, a crucial process during leukemia development. At a molecular level, Foxy-5 led to the downregulation of two overexpressed leukemia pathways, PI3K and MAPK, which resulted in a disarrangement of actin polymerization with consequent impairment of CXCL12-induced chemotaxis. Notably, in a novel tri-dimensional bone marrow-mimicking model, Foxy-5 led to reduced leukemia cell growth and similar results were observed in a xenograft in vivo model. Overall, our findings highlight the pivotal role of WNT5A in leukemia and demonstrate that Foxy-5 acts as a specific antineoplastic agent in leukemia, counterbalancing several leukemic oncogenic processes related to the crosstalk in the bone marrow niche, and represents a promising therapeutic option for AML.
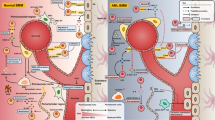
Graphical abstract

WNT5A, a WNT gene/protein family member, is naturally secreted by mesenchymal stromal cells and contributes to the maintenance of the bone marrow microenvironment. WNT5A downregulation is correlated with disease progression and poor prognosis. The treatment with Foxy-5, a WNT5A mimetizing compound, counterbalanced several leukemogenic processes that are upregulated in leukemia cells, including ROS generation, cell proliferation, and autophagy and disruption of PI3K and MAPK signaling pathways.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Asem MS, Buechler S, Wates RB et al (2016) WNT5A signaling in cancer. Cancers (Basel) 8:79. https://doi.org/10.3390/cancers8090079
Bernasconi P, Borsani O (2019) Targeting leukemia stem cell-niche dynamics: a new challenge in AML treatment. J Oncol 2019:8323592. https://doi.org/10.1155/2019/8323592
Braicu C, Buse M, Busuioc C et al (2019) A comprehensive review on MAPK: a promising therapeutic target in cancer. Cancers (Basel) 11:1618. https://doi.org/10.3390/cancers11101618
Canesin G, Evans-Axelsson S, Hellsten R et al (2017) Treatment with the WNT5A-mimicking peptide Foxy-5 effectively reduces the metastatic spread of WNT5A-low prostate cancer cells in an orthotopic mouse model. PLoS ONE 12:e0184418. https://doi.org/10.1371/journal.pone.0184418
Cerami E, Gao J, Dogrusoz U et al (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2:401–404. https://doi.org/10.1158/2159-8290.CD-12-0095
Corrigan PM, Dobbin E, Freeburn RW, Wheadon H (2009) Patterns of Wnt/Fzd/LRP gene expression during embryonic hematopoiesis. Stem Cells Dev 18:759–772. https://doi.org/10.1089/scd.2008.0270
Diaz-Vivancos P, de Simone A, Kiddle G, Foyer CH (2015) Glutathione–linking cell proliferation to oxidative stress. Free Radic Biol Med 89:1154–1164. https://doi.org/10.1016/j.freeradbiomed.2015.09.023
Favaro P, Traina F, Machado-Neto JA et al (2013) FMNL1 promotes proliferation and migration of leukemia cells. J Leukoc Biol 94:503–512. https://doi.org/10.1189/jlb.0113057
Gao J, Aksoy BA, Dogrusoz U et al (2013) Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 6:11. https://doi.org/10.1126/scisignal.2004088
Garg AD, Dudek AM, Ferreira GB et al (2013) ROS-induced autophagy in cancer cells assists in evasion from determinants of immunogenic cell death. Autophagy 9:1292–1307. https://doi.org/10.4161/auto.25399
Giacinti C, Giordano A (2006) RB and cell cycle progression. Oncogene 25:5220–5227. https://doi.org/10.1038/sj.onc.1209615
Jensen HA, Yourish HB, Bunaciu RP et al (2015) Induced myelomonocytic differentiation in leukemia cells is accompanied by noncanonical transcription factor expression. FEBS Open Bio 5:789–800. https://doi.org/10.1016/j.fob.2015.09.008
Liang H, Chen Q, Coles AH et al (2003) WNT5A inhibits B cell proliferation and functions as a tumor suppressor in hematopoietic tissue. Cancer Cell 4:349–360. https://doi.org/10.1016/s1535-6108(03)00268-x
Liou GY, Storz P (2010) Reactive oxygen species in cancer. Free Radic Res 44:479–496. https://doi.org/10.3109/10715761003667554
Luis TC, Ichii M, Brugman MH et al (2012) Wnt signaling strength regulates normal hematopoiesis and its deregulation is involved in leukemia development. Leukemia 26:414–421. https://doi.org/10.1038/leu.2011.387
Martín V, Valencia A, Agirre X et al (2010) Epigenetic regulation of the non-canonical Wnt pathway in acute myeloid leukemia. Cancer Sci 101:425–432. https://doi.org/10.1111/j.1349-7006.2009.01413.x
Mele L, Del Vecchio V, Liccardo D et al (2020) The role of autophagy in resistance to targeted therapies. Cancer Treat Rev 88:102043. https://doi.org/10.1016/j.ctrv.2020.102043
Melo RCC, Longhini AL, Bigarella CL et al (2014) CXCR7 is highly expressed in acute lymphoblastic leukemia and potentiates CXCR4 response to CXCL12. PLoS ONE 9:e85926. https://doi.org/10.1371/journal.pone.0085926
Méndez-Ferrer S, Bonnet D, Steensma DP et al (2020) Bone marrow niches in haematological malignancies. Nat Rev Cancer 20:285–298. https://doi.org/10.1038/s41568-020-0245-2
Mendoza MC, Vilela M, Juarez JE et al (2015) ERK reinforces actin polymerization to power persistent edge protrusion during motility. Sci Signal 8:ra47. https://doi.org/10.1126/scisignal.aaa8859
Nusse R, Clevers H (2017) Wnt/β-catenin signaling, disease, and emerging therapeutic modalities. Cell 169:985–999. https://doi.org/10.1016/j.cell.2017.05.016
Pavlaki K, Pontikoglou CG, Demetriadou A et al (2014) Impaired proliferative potential of bone marrow mesenchymal stromal cells in patients with myelodysplastic syndromes is associated with abnormal WNT signaling pathway. Stem Cells Dev 23:1568–1581. https://doi.org/10.1089/scd.2013.0283
Perillo B, Di Donato M, Pezone A et al (2020) ROS in cancer therapy: the bright side of the moon. Exp Mol Med 52:192–203. https://doi.org/10.1038/s12276-020-0384-2
Povinelli BJ, Nemeth MJ (2014) WNT5A regulates hematopoietic stem cell proliferation and repopulation through the Ryk receptor. Stem Cells 32:105–115. https://doi.org/10.1002/stem.1513
Roversi FM, Bueno MLP, da Silva JAF et al (2022) Novel inhibitor of hematopoietic cell kinase as a potential therapeutic agent for acute myeloid leukemia. Cancer Immunol Immunother 71:1909–1921. https://doi.org/10.1007/s00262-021-03111-2
Säfholm A, Leandersson K, Dejmek J et al (2006) A formylated hexapeptide ligand mimics the ability of Wnt-5a to impair migration of human breast epithelial cells. J Biol Chem 281:2740–2749. https://doi.org/10.1074/jbc.M508386200
Shen YL, Luo Q, Guo YX et al (2014) Bone marrow mesenchymal stem cell-derived WNT5A inhibits leukemia cell progression. Oncol Lett 8:85–90. https://doi.org/10.3892/ol.2014.2117
Sillar JR, Germon ZP, DeIuliis GN, Dun MD (2019) The role of reactive oxygen species in acute myeloid leukaemia. Int J Mol Sci 20:6003. https://doi.org/10.3390/ijms20236003
Skopek R, Palusińska M, Kaczor-Keller K et al (2023) Choosing the right cell line for acute myeloid leukemia (AML) research. Int J Mol Sci 24:5377. https://doi.org/10.3390/ijms24065377
Tyner JW, Tognon CE, Bottomly D et al (2018) Functional genomic landscape of acute myeloid leukaemia. Nature 562:526–531. https://doi.org/10.1038/s41586-018-0623-z
Valencia A, Román-Gómez J, Cervera J et al (2009) Wnt signaling pathway is epigenetically regulated by methylation of Wnt antagonists in acute myeloid leukemia. Leukemia 23:1658–1666. https://doi.org/10.1038/leu.2009.86
Yadav V, Jobe N, Mehdawi L, Andersson T (2021) Targeting Oncogenic WNT Signalling with WNT Signalling-Derived Peptides. Handb Exp Pharmacol 269:279–303. https://doi.org/10.1007/164_2021_528
Zang S, Liu N, Wang H et al (2014) Wnt signaling is involved in 6-benzylthioinosine-induced AML cell differentiation. BMC Cancer 14:886. https://doi.org/10.1186/1471-2407-14-886
Zhang Y, Choksi S, Chen K et al (2013) ROS play a critical role in the differentiation of alternatively activated macrophages and the occurrence of tumor-associated macrophages. Cell Res 23:898–914. https://doi.org/10.1038/cr.2013.75
Acknowledgements
The authors would like to thank WntResearch AB, Sweden for providing the WNT5A mimicking peptide Foxy-5 and valuable information as to how it should be used; Raquel S Foglio (Hematology and Transfusion Medicine Center, University of Campinas, São Paulo, Brazil) for the English revision; Irene Santos and Nadia Ghinelli Âmor (Hematology and Transfusion Medicine Center, University of Campinas, São Paulo, Brazil) for flow cytometry assistance; Karla Priscila Ferro for in vivo assay assistance; and Mariana Ozello Baratti for microscopy assistance. We also thank the National Institute of Science and Technology on Photonics Applied to Cell Biology (INFABIC) at UNICAMP for the access to equipment and confocal assistance and analysis (FAPESP grants 2014/50938-8 and CNPq grants 465699/2014-6) and the Hemostasis Laboratory of Hematology and Transfusion Medicine Center (FAPESP grants 2016/14172-6) at UNICAMP for cell proliferation analysis by IncuCyte S3 System (Sartorius). The authors also thank CNPq and FAPESP for the financial support.
Funding
This study was funded by grants from the Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP grants 2017/21801-2, 2019/25247-5 and 2021/05320-0) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Grants 303405/2018-0).
Author information
Authors and Affiliations
Contributions
FMR and MLPB designed and performed the experiments, collecting, analyzing and interpreting data, and writing the manuscript; STOS provided financial support, revised and gave final approval of the manuscript. All authors have read, commented, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Ethical approval
The study was approved by the Human Ethics Committee of the University (CAAE 45254321.7.0000.5404 and CAAE 1110.0.146.000- 11), in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Human and animal rights
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article. All patients signed informed consent forms. The study was approved by the Institutional Animal Experimentation Ethics Committee of the University of Campinas (5750-1/2021). All procedures were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Consent for publication
All authors agreed with the content and gave explicit consent. A consent from the responsible authorities at the Hematology and Transfusion Medicine Center/University of Campinas has been carried out.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bueno, M.L.P., Saad, S.T.O. & Roversi, F.M. The antitumor effects of WNT5A against hematological malignancies. J. Cell Commun. Signal. 17, 1487–1499 (2023). https://doi.org/10.1007/s12079-023-00773-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-023-00773-8