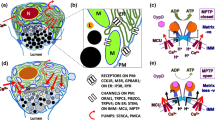
Abstract
Hepatic encephalopathy is described by a broad spectrum of neurological and psychiatric aberrations resulting due to advanced liver dysfunction. It is a neurological disorder due to hepatic insufficiency and/or portosystemic shunts. Its clinical presentation includes neuropsychiatric dysfunction ranging from subclinical changes to comatose state. It is a sign of poor prognosis in cirrhotics with a high 1-year mortality. Each episode of hepatic encephalopathy leads to high hospitalization rate, poor prognosis and raised burden of healthcare. Primary prophylaxis is prevention of initial occurrence and secondary prophylaxis is prevention of reappearance of hepatic encephalopathy in subjects who had prior history. Early detection and management of triggers is very important in the treatment of hepatic encephalopathy. The initial choice of treatment is still lactulose, as it is effective in minimal, overt, and recurrent hepatic encephalopathy. Rifaximin is equally effective as lactulose in managing hepatic encephalopathy and is better tolerated. Branch chain amino acids are beneficial in subjects who are protein intolerant. L-ornithine L-aspartate and probiotics are also useful in the management of hepatic encephalopathy. Rifaximin along with lactulose is effective in managing overt and recurrent hepatic encephalopathy. Large portosystemic shunts embolization and liver transplant is efficacious in certain group of patients. Nutritional therapy and fecal microbiota transplantation are newer therapies for hepatic encephalopathy but the evidences are limited, more research is required to prove their efficacy. Involvement of hospital pharmacists, telemedicine, and providing education are also beneficial in managing hepatic encephalopathy.




Similar content being viewed by others
References
Bajaj JS, Wade JB, Sanyal AJ. Spectrum of neurocognitive impairment in cirrhosis: implications for the assessment of hepatic encephalopathy. Hepatology 2009;50(6):2014–2021
Amodio P, Del Piccolo F, Pettenò E, et al. Prevalence and prognostic value of quantified electroencephalogram (EEG) alterations in cirrhotic patients. J Hepatol 2001;35(1):37–45
Sharma BC, Maharshi S. Prevention of hepatic encephalopathy recurrence. Clin Liver Dis 2015;5(3):64–67
Lauridsen MM, Jepsen P, Wernberg CW, et al. Validation of a simple quality-of-life score for identification of minimal and prediction of overt hepatic encephalopathy. Hepatol Commun 2020;4(9):1353–1361
Sharma BC, Sharma P, Agrawal A, et al. Secondary prophylaxis of hepatic encephalopathy: an open-label randomized controlled trial of lactulose versus placebo. Gastroenterology 2009;137(3):885-891.e1
Bass NM, Mullen KD, Sanyal A, et al. Rifaximin treatment in hepatic encephalopathy. N Engl J Med 2010;362(12):1071–1081
Agrawal A, Sharma BC, Sharma P, et al. Secondary prophylaxis of hepatic encephalopathy in cirrhosis: an open-label, randomized controlled trial of lactulose, probiotics, and no therapy. Am J Gastroenterol 2012;107(7):1043–1050
Shen YC, Chang YH, Fang CJ, et al. Zinc supplementation in patients with cirrhosis and hepatic encephalopathy: a systematic review and meta-analysis. Nutr J 2019;18(1):34
Gluud LL, Dam G, Les I, et al. Branched-chain amino acids for people with hepatic encephalopathy. Cochrane Database Syst Rev 2015;2:CD001939
Butterworth RF. The liver-brain axis in liver failure: neuroinflammation and encephalopathy. Nat Rev Gastroenterol Hepatol 2013;10:522–528
Jain L, Sharma BC, Srivastava S, et al. Serum endotoxin, inflammatory mediators, and magnetic resonance spectroscopy before and after treatment in patients with minimal hepatic encephalopathy. J Gastroenterol Hepatol 2013;28:1187–1193
Bajaj JS, Ridlon JM, Hylemon PB, et al. Linkage of gut microbiome with cognition in hepatic encephalopathy. Am J Physiol Gastrointest Liver Physiol 2012;302:G168–G175
Bajaj JS, Heuman DM, Sanyal AJ, et al. Modulation of the metabiome by rifaximin in patients with cirrhosis and minimal hepatic encephalopathy. PLoS ONE 2013;8: e60042
Bombassaro IZ, Tovo CV, De Mattos ÂZ, et al. Albumin in the management of hepatic encephalopathy: a systematic review and meta-analysis. Ann Hepatol 2021;26: 100541
Laleman W, Simon-Talero M, Maleux G, et al. Embolization of large spontaneous portosystemic shunts for refractory hepatic encephalopathy: a multicenter survey on safety and efficacy. Hepatology 2013;57(6):2448–2457
Mukund A, Rajesh S, Arora A, et al. Efficacy of balloon-occluded retrograde transvenous obliteration of large spontaneous lienorenal shunt in patients with severe recurrent hepatic encephalopathy with foam sclerotherapy: initial experience. J Vasc Interv Radiol 2012;23(9):1200–1206
Lv Y, Chen H, Luo B, et al. Concurrent large spontaneous portosystemic shunt embolization for the prevention of overt hepatic encephalopathy after TIPS: a randomized controlled trial. Hepatology 2022;76(3):676–688
Yi F, Guo X, Wang L, et al. Impact of spontaneous splenorenal shunt on liver volume and long-term survival of liver cirrhosis. J Gastroenterol Hepatol 2021;36(6):1694–1702
Greinert R, Zipprich A, Simón-Talero M, et al. Covert hepatic encephalopathy and spontaneous portosystemic shunts increase the risk of developing overt hepatic encephalopathy. Liver Int 2020;40(12):3093–3102
Sharma P, Sharma BC, Agrawal A, et al. Primary prophylaxis of overt hepatic encephalopathy in patients with cirrhosis: an open labeled randomized controlled trial of lactulose versus no lactulose. J Gastroenterol Hepatol 2012;27(8):1329–1335
Bajaj JS, Sanyal AJ, Bell D, et al. Predictors of the recurrence of hepatic encephalopathy in lactulose-treated patients. Aliment Pharmacol Ther 2010;31(9):1012–1017
Leevy CB, Phillips JA. Hospitalizations during the use of rifaximin versus lactulose for the treatment of hepatic encephalopathy. Dig Dis Sci 2007;52(3):737–741
Descombe JJ, Dubourg D, Picard M, et al. Pharmacokinetic study of rifaximin after oral administration in healthy volunteers. Int J Clin Pharmacol Res 1994;14(2):51–56
Sidhu SS, Goyal O, Parker RA, et al. Rifaximin vs. lactulose in treatment of minimal hepatic encephalopathy. Liver Int 2016;36(3):378–385
Riggio O, Ridola L, Pasquale C, et al. Evidence of persistent cognitive impairment after resolution of overt hepatic encephalopathy. Clin Gastroenterol Hepatol 2011;9(2):181–183
Dhiman RK, Rana B, Agrawal S, et al. Probiotic VSL#3 reduces liver disease severity and hospitalization in patients with cirrhosis: a randomized, controlled trial. Gastroenterology 2014;147(6):1327-1337.e3
Vlachogiannakos J, Viazis N, Vasianopoulou P, et al. Long-term administration of rifaximin improves the prognosis of patients with decompensated alcoholic cirrhosis: rifaximin improves prognosis in cirrhosis. J Gastroenterol Hepatol 2013;28(3):450–455
Amodio P. Hepatic encephalopathy: diagnosis and management. Liver Int 2018;38(6):966–975
Zeng X, Sheng X, Wang PQ, et al. Low-dose rifaximin prevents complications and improves survival in patients with decompensated liver cirrhosis. Hepatol Int 2021;15(1):155–165
Abdel Moneim M, Abdelaziz DH, Ibrahim Nagy Y, et al. Rifaximin microbial resistance and its efficacy and safety as a secondary prophylaxis of hepatic encephalopathy in patients with hepatitis C virus-related cirrhosis. Int J Clin Pract 2021;75(11): e14807
Glal KAM, Abd-Elsalam SM, Mostafa TM. Nitazoxanide versus rifaximin in preventing the recurrence of hepatic encephalopathy: a randomized double-blind controlled trial. J Hepatobil Pancreat Sci 2021;28(10):812–824
Bajaj JS, Heuman DM, Wade JB, et al. Rifaximin improves driving simulator performance in a randomized trial of patients with minimal hepatic encephalopathy. Gastroenterology 2011;140(2):478-487.e1
De Jong LA, Van Schoonhoven AV, Hofstra HS, et al. Budget impact of optimizing rifaximin-α use for the prevention of recurrent hepatic encephalopathy in The Netherlands. J Med Econ 2021;24(1):1149–1163
Butterworth RF. Ammonia removal by metabolic scavengers for the prevention and treatment of hepatic encephalopathy in cirrhosis. Drugs R D 2021;21(2):123–132
Varakanahalli S, Sharma BC, Srivastava S, et al. Secondary prophylaxis of hepatic encephalopathy in cirrhosis of liver: a double-blind randomized controlled trial of l-ornithine l-aspartate versus placebo. Eur J Gastroenterol Hepatol 2018;30(8):951–958
Butterworth RF, McPhail MJW. l-Ornithine l-aspartate (LOLA) for hepatic encephalopathy in cirrhosis: results of randomized controlled trials and meta-analyses. Drugs 2019;79(S1):31–37
Goh ET, Stokes CS, Sidhu SS, et al. l-Ornithine l-aspartate for prevention and treatment of hepatic encephalopathy in people with cirrhosis. Cochrane Database Syst Rev 2018;5(5):CD012410
Butterworth RF. Beneficial effects of l-ornithine l-aspartate for prevention of overt hepatic encephalopathy in patients with cirrhosis: a systematic review with meta-analysis. Metab Brain Dis 2020;35(1):75–81
Alvares-da-Silva MR, de Araujo A, Vicenzi JR, et al. Oral l-ornithine-l-aspartate in minimal hepatic encephalopathy: a randomized, double-blind, placebo-controlled trial. Hepatol Res 2014;44(9):956–963
Mittal VV, Sharma BC, Sharma P, et al. A randomized controlled trial comparing lactulose, probiotics, and l-ornithine l-aspartate in treatment of minimal hepatic encephalopathy. Eur J Gastroenterol Hepatol 2011;23(8):725–732
Bajaj JS, Saeian K, Christensen KM, et al. Probiotic yogurt for the treatment of minimal hepatic encephalopathy. Am J Gastroenterol 2008;103(7):1707–1715
Saji S, Kumar S, Thomas V. A randomized double blind placebo controlled trial of probiotics in minimal hepatic encephalopathy. Trop Gastroenterol 2011;32(2):128–132
Lunia MK, Sharma BC, Sharma P, et al. Probiotics prevent hepatic encephalopathy in patients with cirrhosis: a randomized controlled trial. Clin Gastroenterol Hepatol 2014;12(6):1003-1008.e1
Lichter-Konecki U, Diaz GA, Merritt JL, et al. Ammonia control in children with urea cycle disorders (UCDs); phase 2 comparison of sodium phenylbutyrate and glycerol phenylbutyrate. Mol Genet Metab 2011;103(4):323–329
Rockey DC, Vierling JM, Mantry P, et al. Randomized, double-blind, controlled study of glycerol phenylbutyrate in hepatic encephalopathy. Hepatology 2014;59(3):1073–1083
Les I, Doval E, García-Martínez R, et al. Effects of branched-chain amino acids supplementation in patients with cirrhosis and a previous episode of hepatic encephalopathy: a randomized study. Am J Gastroenterol 2011;106(6):1081–1088
Park JG, Tak WY, Park SY, et al. Effects of branched-chain amino acids (BCAAs) on the progression of advanced liver disease: a Korean nationwide, multicenter, retrospective, observational, cohort study. Medicine 2017;96(24): e6580
Shaw J, Tate V, Hanson J, et al. What diet should I recommend my patient with hepatic encephalopathy? Curr Hepatol Rep 2020;19(1):13–22
Bischoff SC, Bernal W, Dasarathy S, et al. ESPEN practical guideline: clinical nutrition in liver disease. Clin Nutr 2020;39(12):3533–3562
Maharshi S, Sharma BC, Sachdeva S, et al. Efficacy of nutritional therapy for patients with cirrhosis and minimal hepatic encephalopathy in a randomized trial. Clin Gastroenterol Hepatol 2016;14(3):454-460.e3
Ahuja H, Sharma BC, Sachdeva S, et al. A double blind randomized controlled trial to assess efficacy of nutritional therapy for prevention of recurrence of hepatic encephalopathy in patients with cirrhosis. J Gastroenterol Hepatol 2023;38(3):433–440
Frenette CT, Levy C, Saab S. Hepatic encephalopathy-related hospitalizations in cirrhosis: transition of care and closing the revolving door. Dig Dis Sci 2022;67(6):1994–2004
Hassouneh R, Bajaj JS. Gut microbiota modulation and fecal transplantation: an overview on innovative strategies for hepatic encephalopathy treatment. JCM 2021;10(2):330
Bajaj JS, Salzman NH, Acharya C, et al. Fecal microbial transplant capsules are safe in hepatic encephalopathy: a phase 1, randomized placebo-controlled trial. Hepatology 2019;70(5):1690–1703
Caraceni P, Riggio O, Angeli P, et al. Long-term albumin administration in decompensated cirrhosis (ANSWER): an open-label randomised trial. Lancet 2018;391(10138):2417–2429
China L, Freemantle N, Forrest E, et al. A randomized trial of albumin infusions in hospitalized patients with cirrhosis. N Engl J Med 2021;384:808–817
Pockros P, Hassanein T, Vierling J, et al. Phase 2, multicenter, randomized study of AST-120 (spherical carbon adsorbent) vs. lactulose in the treatment of low-grade hepatic encephalopathy (HE). J Hepatol 2009;50:S43–S44
Bajaj J, Sheikh M, Chojkier M, et al. AST-120 (spherical carbon adsorbent) in covert hepatic encephalopathy: results of the Astute trial. J Hepatol 2013;58:S84
Malaguarnera M. Acetyl-l-carnitine in hepatic encephalopathy. Metab Brain Dis 2013;28:193–199
Malaguarnera M, Vacante M, Giordano M, et al. Oral acetyl-l carnitine therapy reduces fatigue in overt hepatic encephalopathy: a randomized, double-blind, placebo-controlled study. Am J Clin Nutr 2011;93:799–808
Martí-Carvajal A, Gluud C, Arevalo-Rodriguez I. Acetyl-l-carnitine for patients with hepatic encephalopathy. Cochrane Database Syst Rev 2019;1(1):CD011451
Amrein R, Leishman B, Bentzinger C, et al. Flumazenil in benzodiazepine antagonism. Med Toxicol 1987;2:411–429
Ahboucha S, Butterworth R. The neurosteroid system: implication in the pathophysiology of HE. Neurochem Int 2008;52:575–587
Goh E, Andersen M, Morgan M, et al. Flumazenil versus placebo or no intervention for people with cirrhosis and hepatic encephalopathy. Cochrane Database Syst Rev 2017;1(7):CD002798
Hammer H, Santa Ana C, Schiller L, et al. Studies of osmotic diarrhea induced in normal subjects by ingestion of polyethylene glycol and lactulose. J Clin Invest 1989;84:1056–1062
Rahimi RS, Singal AG, Cuthbert JA, et al. Lactulose vs polyethylene glycol 3350-electrolyte solution for treatment of overt hepatic encephalopathy: the HELP randomized clinical trial. JAMA Intern Med 2014;174:1727–1733
Naderian M, Akbari H, Saeedi M, et al. Polyethylene glycol and lactulose versus lactulose alone in the treatment of HE in patients with cirrhosis: a non-inferiority randomized controlled trial. Middle East J Dig Dis 2017;9:12–19
Gammal S, Basile A, Geller D, et al. Reversal of the behavioral and electrophysiological abnormalities of an animal model of hepatic encephalopathy by benzodiazepine receptor ligands. Hepatology 1990;11:371–378
Jiang Q, Jiang G, Welty T, et al. Naloxone in the management of hepatic encephalopathy. J Clin Pharm Ther 2010;35:333–341
Coelho Santos RP, de Brito Toscano EC, Rachid MA. Anti-inflammatory strategies for hepatic encephalopathy: preclinical studies. Arq Neuropsiquiatr 2023;81(7):656–669
Pereira K, Carrion AF, Salsamendi J, et al. Endovascular management of refractory hepatic encephalopathy complication of transjugular intrahepatic portosystemic shunt (TIPS): Comprehensive Review and Clinical Practice Algorithm. Cardiovasc Intervent Radiol 2016;39(2):170–182
Nardelli S, Bellafante D, Ridola L, et al. Prevention of post-tips hepatic encephalopathy: the search of the ideal candidate. Metab Brain Dis 2023;38(5):1729–1736
Pereira K, Carrion AF, Martin P, et al. Current diagnosis and management of post-transjugular intrahepatic portosystemic shunt refractory hepatic encephalopathy. Liver Int 2015;35(12):2487–2494
Saab S, Kim NG, Lee EW. Practical tips on TIPS: when and when not to request it. Am J Gastroenterol 2020;115(6):797–800
Maleux G, Verslype C, Heye S, et al. Endovascular shunt reduction in the management of transjugular portosystemic shunt-induced hepatic encephalopathy: preliminary experience with reduction stents and stent-grafts. AJR Am J Roentgenol 2007;188(3):659–664
Cookson DT, Zaman Z, Gordon-Smith J, et al. Management of transjugular intrahepatic portosystemic shunt (TIPS)-associated refractory hepatic encephalopathy by shunt reduction using the parallel technique: outcomes of a retrospective case series. Cardiovasc Intervent Radiol 2011;34(1):92–99
Madoff DC, Wallace MJ, Ahrar K, et al. TIPS-related hepatic encephalopathy: management options with novel endovascular techniques. Radiographics 2004;24(1):21–36 (discussion 36–37)
Wu W, He C, Han G. Embolization of spontaneous splenorenal shunt for after-TIPS hepatic encephalopathy in a patient with cirrhosis and variceal bleeding. Hepatology 2015;61(5):1761–1762
Shioyama Y, Matsueda K, Horihata K, et al. Post-TIPS hepatic encephalopathy treated by occlusion balloon-assisted retrograde embolization of a coexisting spontaneous splenorenal shunt. Cardiovasc Intervent Radiol 1996;19(1):53–55
Higuera-de-la-Tijera F, Servín-Caamaño AI, Salas-Gordillo F, et al. Primary prophylaxis to prevent the development of hepatic encephalopathy in cirrhotic patients with acute variceal bleeding. Can J Gastroenterol Hepatol 2018;10(2018):1–10
Hassanein TI, Tofteng F, Brown RS Jr, et al. Randomized controlled study of extracorporeal albumin dialysis for hepatic encephalopathy in advanced cirrhosis. Hepatology 2007;46(6):1853–1862
Banares R, Nevens F, Larsen FS, et al. Extracorporeal albumin dialysis with the molecular adsorbent recirculating system in acute-on-chronic liver failure: the RELIEF trial. Hepatology 2013;57:1153–1162
March KL, Peters MJ, Finch CK, et al. Pharmacist transition-of-care services improve patient satisfaction and decrease hospital readmissions. J Pharm Pract 2022;35(1):86–93
Stotts MJ, Grischkan JA, Khungar V. Improving cirrhosis care: the potential for telemedicine and mobile health technologies. WJG 2019;25(29):3849–3856
Ganapathy D, Acharya C, Lachar J, et al. The patient buddy app can potentially prevent hepatic encephalopathy-related readmissions. Liver Int 2017;37(12):1843–1851
Garrido M, Turco M, Formentin C, et al. An educational tool for the prophylaxis of hepatic encephalopathy. BMJ Open Gastroenterol 2017;4(1): e000161
Aamann L, Tandon P, Bémeur C. Role of exercise in the management of hepatic encephalopathy: experience from animal and human studies. J Clin Exp Hepatol 2019;9(1):131–136
Tapper EB, Parikh ND, Green PK, et al. Reduced incidence of hepatic encephalopathy and higher odds of resolution associated with eradication of HCV infection. Clin Gastroenterol Hepatol 2020;18(5):1197-1206.e7
Gairing SJ, Schleicher EM, Galle PR, et al. Prediction and prevention of the first episode of overt hepatic encephalopathy in patients with cirrhosis. Hepatol Commun 2023;7(4): e0096
Wen J, Liu Q, Song J, et al. Lactulose is highly potential in prophylaxis of hepatic encephalopathy in patients with cirrhosis and upper gastrointestinal bleeding: results of a controlled randomized trial. Digestion 2013;87(2):132–138
Sharma P, Agrawal A, Sharma BC, et al. Prophylaxis of hepatic encephalopathy in acute variceal bleed: a randomized controlled trial of lactulose versus no lactulose: lactulose in acute variceal bleed. J Gastroenterol Hepatol 2011;26(6):996–1003
Aires FT, Ramos PT, Bernardo WM. Efficacy of lactulose in the prophylaxis of hepatic encephalopathy in cirrhotic patients presenting gastrointestinal bleeding. Rev Assoc Med Bras 2016;62(3):243–247
Rattanasupar A, Chang A, Akarapatima K, et al. Role of lactulose for prophylaxis against hepatic encephalopathy in cirrhotic patients with upper gastrointestinal bleeding: a randomized trial. Indian J Gastroenterol 2021;40(6):621–629
Maharshi S, Sharma BC, Srivastava S, et al. Randomised controlled trial of lactulose versus rifaximin for prophylaxis of hepatic encephalopathy in patients with acute variceal bleed. Gut 2015;64(8):1341–1342
Prakash RK, Kanna S, Mullen KD. Evolving concepts: the negative effect of minimal hepatic encephalopathy and role for prophylaxis in patients with cirrhosis. Clin Ther 2013;35(9):1458–1473
Hartmann IJC, Groeneweg M, Quero JC, et al. The prognostic significance of subclinical hepatic encephalopathy. Am J Gastroenterol 2000;95(8):2029–2034
Mullen KD, Ferenci P, Bass NM, et al. An algorithm for the management of hepatic encephalopathy. Semin Liver Dis 2007;27(S2):32–48
Bajaj JS, Schubert CM, Heuman DM, et al. Persistence of cognitive impairment after resolution of overt hepatic encephalopathy. Gastroenterology 2010;138(7):2332–2340
Sanyal A, Younossi ZM, Bass NM, et al. Randomised clinical trial: rifaximin improves health-related quality of life in cirrhotic patients with hepatic encephalopathy-a double blind placebo-controlled study. Aliment Pharmacol Ther 2011;34(8):853–861
Funding
None.
Author information
Authors and Affiliations
Contributions
Sudhir Maharshi, Barjesh Chander Sharma: drafting of article and critical revision for important intellectual content, final approval of the version to be published. Barjesh Chander Sharma: agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the article are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
Sudhir Maharshi and Barjesh Chander Sharma have not disclosed any competing interests.
Compliance with ethical standards
This article does not contain any studies with human or animal subjects.
Ethical approval
Not required (review article).
Informed consent
Not required (review article).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maharshi, S., Sharma, B.C. Prophylaxis of hepatic encephalopathy: current and future drug targets. Hepatol Int (2024). https://doi.org/10.1007/s12072-024-10647-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12072-024-10647-9