Abstract
Background
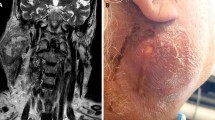
Fine needle aspiration cytology (FNAC) is an integral part of the preoperative work-up of parotid tumours.
Aim
To determine the rate of concordance between FNAC and histology following parotidectomy.
Methods
A review of records of patients who had parotidectomy which was preceded FNAC was done. Data collected included patients’ demography, presenting symptoms and clinical signs; cytology and post-operative histology results.
Results
Seventy-seven records were found and 14 were excluded. Forty-five (71%: 45/63) of the tumours were benign, 21% (13/63) malignant and 8% (5/63) inflammatory lesions. Forty-one (91.1%: 41/45) of the benign tumours had concordance between FNAC and final histology. Seven (63.6%: 7/11) of FNAC diagnosed malignancies were confirmed on histology.
Conclusion
Around 71% of parotid masses were benign. Painful masses are more likely to be malignant and FNAC is more reliable for the diagnosis of pleomorphic adenoma than rare benign and malignant tumours of the parotid gland.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neoplasms of the salivary gland are rare and constitute around 3–6% of all head and neck tumours, and majority of them occur in the parotid gland [1]. Over 80% of neoplasms of the parotid gland are benign [2]. Pleomorphic adenoma and Warthin’s tumour are the most common benign tumours of the parotid gland [2, 3]. The common malignant tumours of the parotid gland are mucoepidermoid carcinoma, adenoid cystic carcinoma and acinic cell carcinoma [1, 2]. Rare malignant tumours of the parotid gland include adenocarcinoma, squamous cell carcinoma, lymphoma [4] and carcinoma ex-pleomorphic adenoma [5].
There is a similarity between the clinical presentation of benign and malignant lesions of the parotid gland [1, 6]. Facial nerve involvement, a painful mass, cervical lymphadenopathy in a patient who has a parotid tumour is/are highly suggestive of malignancy. Facial nerve palsy and enlargement of cervical lymph nodes may however occur in patients who had benign diseases of the parotid gland such as pleomorphic adenoma, odontoma and granulomatous parotitis [7,8,9]. Fine needle aspiration cytology (FNAC) is key in the preoperative work-up of parotid masses [6, 10, 11]. The result of FNAC is used to guide management including the need for additional investigations [12, 13].
The sensitivity and specificity of FNAC in benign and malignant tumours of the parotid gland is 71-100% and 33 − 100%, respectively [10, 12, 14, 15]. The positive and negative predictive value, and accuracy of FNAC for parotid masses ranges from as low as 33% to over 80% [14, 15]. Although FNAC has a high negative predictive value, it is less helpful for pre-operative subclassification of primary or metastatic malignancies of the parotid gland [14]. The diagnostic yield of FNAC is significantly low in Warthin’s tumour, low-grade mucoepidermoid carcinoma and rare malignant tumours of the parotid gland [16,17,18,19,20,21,22]. An incorrect FNAC result may lead to a delay in surgical intervention or performance of an unnecessary parotidectomy such as for tuberculous parotitis [23].
The difficulties associated with the interpretation and reporting of results together with the heterogeneity of salivary gland tumours have necessitated the development of systems to standardize reporting results following FNAC of salivary gland tumours [24]. The Milan System for reporting salivary gland cytopathology is the most commonly used system globally [14, 16, 24]. The Milan System categorizes FNAC result into non-diagnostic (I), non-neoplastic (II), atypia of unknown undetermined significance (III), benign neoplasm (IVa), neoplasm of uncertain malignant potential (IVb), suspicious of malignancy (V) and malignant (VI).
In our setting FNAC is done free-hand and the Milan System has not yet been adopted. The researchers have dealt with patients who had radical parotidectomy based on pre-operative FNAC result when surgery was not actually necessary. The aim of the study was to determine the rate of concordance between pre-operative FNAC and histology following parotidectomy.
Materials and methods
Records of patients 18 years and older who had a preoperative FNAC followed by parotidectomy for parotid gland mass over a 5-year were reviewed. Patients who had redo-parotidectomies were excluded. Categorical data such as gender, diagnosis and type of surgical procedure were summarised using counts and percentage. The average or median was used to aggregate continuous data such as age. Sensitivity, specificity, accuracy, positive predictive value and negative predictive were calculated. The chi-square test was used for comparison of categorical data. Statistical significance was set at a p-value below 0.05.
Results
A total of 76 records of patients who had parotidectomy were accessed of which 13 were excluded. Four (30.8%: 4/13) of the records were excluded as the patients were below the age of 18 years, 2/13 (15.4%) had re-do parotidectomy, 4/13 (30.8%) did not have histology results and in 3/13 (23.19%) FNAC results could not be traced. The final sample was made up of 63 patients that had parotidectomy which was preceded by FNAC. Thirty-three (52%: 33/63) of the patients were males and their mean age was 43.3 ± 12.9 years (range: 23–74 years). Forty-five (71.4%: 45/63) of the patients were below the age of 51 years (Fig. 1).
Eight (12.7%:8/63) patients presented with a painful mass and 1.6% (1/63) had evidence of paralysis of the facial nerve. Forty-eight 76.2% (48/63) of the masses had a solid architecture (Table 1). The most commonly reported category as per the Milan system was Category IVa (benign neoplasm) making up 47.6% (30/63) of the records and was followed by Category II at 31.7% (20/63) (Table 2). The diagnosis of isolated pleomorphic adenoma was captured in 27/63 (42.9%) of the records. Malignant tumours which were either confirmed or suspected on FNAC included adenoid cystic carcinoma, mucoepidermoid carcinoma and poorly differentiated carcinoma (Table 3).
Forty-two (66.7%: 42/63) of the masses were histologically confirmed to be neoplastic whereas 7.9% (5/63) were inflammatory. Twenty-six (61.9%: 26/42) of the neoplastic lesions were pleomorphic adenoma. Lymphoepithelial cysts were confirmed in 75.0% (12/16) of the cases which were suspected following preoperative FNAC. All seven malignant tumours of the parotid gland which were suspected on pre-operative FNAC were confirmed on histology and the overall false negative rate was 36.4% (4/11). Three (27.3%: 3/11) of the malignant tumours were mucoepidermoid carcinomas (Table 4).
Forty-one (91.1%: 41/45) the non-malignant lesions were correctly diagnosed as benign following preoperative FNAC. Seven (63.6%: 7/11) malignant tumours were correctly diagnosed pre-operatively following FNAC. One (5%: 1/20) malignant tumour was diagnosed in the 19–35 years age group compared to 20% (5/25) and 38.9% (7/18) each for the 36–50 years and the above 50 years age groups, respectively. The difference in the rate of occurrence of malignancies in the patients in the age groups was statistically significant (p-value = 0.032). None of the patients who had benign neoplasms complained of pain as compared to 38.5% (5/13) in patients who had malignant tumours, and the difference was statistically significant (p-value of 0.000) (Table 5). The overall positive predictive value was 100%( all seven of the cases assessed as malignant on FNAC were confirmed on histology; there were no false positive cases) and negative predictive value was 91%( 41/45 cases that were identified as benign on FNAC were confirmed as benign following parotidectomy; however, 4/11 cases that were missed on FNAC were confirmed malignant on histology).
Discussion
The aim of the study was to ascertain the usefulness of fine-needle aspiration cytology (FNAC) in clinical decision-making for parotid gland masses. The study compared preoperative FNAC results with the post-operative histological findings. Among the key findings is that FNAC is highly reliable for diagnosing benign parotid gland tumours with specificity above 90% as compared to low specificity of 30% for malignant lesions.
Pleomorphic adenoma, Warthin’s tumour and lymphoepithelial cyst were as expected among the most commonly diagnosed causes of parotid masses [2, 25]. The most common malignant tumour of the parotid gland was mucoepidermoid carcinoma which is in keeping with what is reported in the literature [2, 25]. The higher rate of discordance between pre-operative FNAC and histology involved acinic cell carcinoma and other rare benign and malignant tumours of the parotid gland, which is not surprising especially in cases of low-grade tumours [21].
The study only consisted of adult patients whose mean age at presentation was 43.3 years. Although neoplasms of the parotid gland can occur in children, they are exceedingly rare [23] There was a significant relationship between age and final histology results and the incidence of malignant disease increased with age and this is in keeping with current literature [2]. All patients who were studied presented with a parotid mass. Only in one patient who had a malignant tumour had evidence of facial nerve paralysis. Facial nerve palsy is however sometimes seen in patients who have benign tumours such as pleomorphic adenoma [26]. Unlike the involvement of the facial nerve, a painful tumour is likely to be associated with an underlying malignancy as it was shown in the current study; which differs with the findings by Mallon and colleagues [14].
The most common neoplasm of the parotid in the current study was pleomorphic adenoma, which is like what is reported in the literature [2, 14, 27]. The next most common cause of parotid masses was lymphoepithelial cyst. High prevalence of lymphoepithelial cyst is likely to be due high incidence of HIV in the study population [28, 29]. The finding however underestimates the prevalence of lymphoepithelial lesions of the parotid gland as parotidectomy is not among the recommended treatment options for a lymphoepithelial cyst [30].
As expected, 21% of the neoplastic masses of the parotid gland were malignant tumours [27]. The most common malignant tumour was mucoepidermoid carcinoma which is in keeping with prior findings [2, 3]. Majority of malignancies were suspected on preoperative FNAC(7/11; 63.6%). None of acinic cell carcinoma, carcinoma ex-pleomorphic adenoma and lymphoma was diagnosed on FNAC. It is however known that FNAC may miss low grade mucoepidermoid carcinoma and the rare tumours of the salivary gland [18, 21, 22].
Granulomatous diseases accounted for 3,2% of the causes of parotid masses. Among the granulomatous diseases of the parotid gland is tuberculosis (TB) which was unfortunately only diagnosed following parotidectomy in the current study. Tuberculosis of the parotid gland is an uncommon condition even in countries where the disease is endemic [31]. Tuberculous parotitis may present as a hard mass mimicking malignancy [23]. A low index of suspicion for TB, along with an inconclusive FNAC result led to an unnecessary parotidectomy instead of medical therapy which is the recommended treatment [31]. It is however possible to diagnose TB of the parotid gland on FNAC as was demonstrated by Gupta and colleagues [32].
Other rare benign conditions such as lipomas were found on 1.6% patient in the study which is in keeping with the reported prevalence rate of 0.6–4.4% [33]. Lymphoma of the salivary glands is rare and account for 2–5% of all salivary gland tumours. Around 70–80% of lymphoma of the parotid gland occur in the parotid gland [34]. In our study, one case of non-Hodgkin’s lymphoma needed parotidectomy as FNAC result was inconclusive. Other authors have also reported a low probability of diagnosing basal cell adenoma [35], oncocytoma [8], low grade mucoepidermoid carcinoma [36], carcinoma ex pleomorphic adenoma [5], lymphoma [34], adenocarcinoma, acinic cell carcinoma [1, 18]. However, contrary to the reports FNAC was able to diagnose lipoma of the parotid gland [20].
Majority of malignant tumours in the current study were diagnosed following FNAC. The relatively high overall false negative rate of 36.4% might have been because salivary gland tumours in our setting are not managed by a dedicated multidisciplinary team [37]. Furthermore, FNAC in our setting is done free hand and not under ultrasound guidance. Our false negative rate of diagnosis of malignant tumours was however lower that the figures of 43.8% for mucoepidermoid carcinoma reported by a team led by Reerds [37].
Limitations of the Study
The number of cases included in the study is relatively small. Salivary gland tumours are rare, and the size of samples included in majority of previous studies rarely have more than 100 patients. The study relied mainly on records over a period of five years and the challenge encountered was that some details were incorrectly entered, making it very difficult to trace both FNAC and histology results of each patient. This meant that with every incomplete or incorrect record, that patient could not be included in the study. Parotid gland masses are also managed by other departments such as general surgery, whose records were not reviewed. This further skew the sample it is not a true representation of all patients presenting with parotid gland masses to this academic centre. Only patients from one academic hospital were studied which introduces some bias into the study.
Conclusion
Majority of parotid swellings occur in patients below the age of 51 years. Around 71% of parotid masses are due to benign neoplasms. A painful parotid tumour is more likely to be malignant. Although fine needle aspiration cytology is more reliable for the diagnosis of pleomorphic adenoma, it is less useful in the diagnostic work-up of malignant tumours of the parotid gland.
Data Availability
Data will be made available when requested.
References
Speight PM, Barrett AW (2020) Salivary gland tumours: diagnostic challenges and an update on the latest WHO classification. Diagn HIistopathol 26(4):147–158. https://doi.org/10.1016/j.mpdhp.2020.01.001
Sentani K, Ogawa I, Ozana K et al (2019) Characteristic of 5015 salivary gland neoplasms registered in the Hiroshima Tumor tissue Registry over a period of 39 years. J Clin Med 8:506. https://doi.org/10.3390/jcm8050566
Chauhan N, Shah JA (2019) Parotid gland tumours: our experience. Indian J Otolaryngol Head Neck Surg 71(3):378–382. https://doi.org/10.1007/s12070-018-1413-5
Wang J, Li R, Wang Q et al (2020) The clinicopathological features of parotid lymphoma. Int J Clin Exp Pathol 13(8):2050–2057
Khanna D, Chaubal T, Bapat R, Philip ST, Arora S et al (2019) Carcinoma ex pleomorphic adenoma: a case report and review of literature. Afri Health Sci 19(4):3253–3263. https://doi.org/10.4314/ahs.v19i4.50
Thielker J, Grosheva M, Ihrler S et al (2018) Guntinas-Lichius O. Contemporary Management of Benign and malignant parotid tumours. Front Surg. https://doi.org/10.3389/fsurg.2018.00039
Nwawka OK, Nadgir R, Fujita A, Sakai O (2014) Granulomatous Disease in the Head and Neck: developing a Differential diagnosis. RG 34(5). https://doi.org/10.1148/rg.345130068
Hamada S, Fujiwara K, Hatakeyama H, Homma A Oncocytoma of the parotid gland with facial nerve paralysis. Case Reports in Otolaryngology 2018. https://doi.org/10.1155/2018/7687951
Stewart KE, Bannon R, Bannister M (2021) ; 103: 47–52. doi.1308/rcsann.2020.0194
Gudmundsson JK, Ajan A, Abtahi J Accuracy of fine-needle aspiration cytology for diagnosis of parotid gland masses: a clinicopathological study of 114 patients.J. of Appl. Sciences. 2016 July; 24(6):561–567. doi: https://doi.org/10.1590/1678-775720160214
Yildiz S, Seneldir L, Tepe KC, Zer Toros S (2021) Fine-needle aspiration cytology of salivary gland Tumours before the Milan System: ten years of experience at a Tertiary Care Center in Turkey. Medeni Med J 36:233–240. https://doi.org/10.5222/MMJ.2021.90912
To VSH, Chan JYW, Tsang RKY, Wei WI (2012) Review of salivary gland neoplasms. Int Sch Res Netw. https://doi.org/10.5402/2012/872982
Barbarite E, Puram SV, Derakhshan A et al (2020) A call for Universal Acceptance of the Milan System for reporting salivary gland cytopathology. Laryngoscope 130(1):80–85. https://doi.org/10.1002/lary.27905
Mallon DH, Kostalas M, MacPherson FJ et al (2013) The diagnostic value of fine needle aspiration in parotid lumps. Ann R Coll Surg Engl 95:258–262. https://doi.org/10.1308/003588413X13511609958370
Shalley S, Chand N, Aggarwal A, Garg LN, Yadav V, Yadav A (2018) Diagnostic accuracy of fine needle aspiration cytology in lesions of oral cavity and salivary glands: a clinico-pathological study. The Open Dentistry Journal 12:782–790. https://doi.org/10.2174/1745017901814010782,2018,12,782-790
Jurczyk M, Peevey JF, Vande Haar MA, Lin X (2015) Pitfalls of fine-needle aspiration cytology of parotid membranous basal cell adenoma – A review of Pitfalls in FNA Cytology of salivary gland neoplasms with basaloid cell features. Diag Cytopath 43(5):432–437. https://doi.org/10.1002/dc.23234
Liu CC, Jethwa AR, Khariwala SS, Johnson J, Shin JJ (2016) Sensitivity, specificity, and Posttest Probability of Parotid Fine-Needle aspiration: a systematic review and Meta-analysis. Otolaryngol Head Neck Surg 154(1):9–23. https://doi.org/10.1177/0194599815607841
Marzouki HZ, Altabsh MA, Albakrei MO, Al-Khatib TA, Merdad MA, Farsi NJ (2017) Accuracy of preoperative fine needle aspiration in diagnosis of malignant parotid tumours. Saudi Med J 38(10). https://doi.org/10.15537/smj.2017.10.20988
Eytan DF, Yin LX, Maleki Z, Koch WM, Tufuno RP Utility of preoperative fine-needle aspiration in parotid lesions.The Larynx 2018Feb; 128:298–402
Tilaveridis I, Kalaitsidou I, Pastelli N, Antoniades K (2018) Lipoma of Parotid Gland: report of two cases. J Maxilofac Oral Srg 17(4):453–457. https://doi.org/10.1007/s12663-018-1080-9
Iftikhar H, Awan MS, Usman M, Khoja A, Khan W (2020) Discordance between fine-needle aspiration cytology and histopathology in patients with mucoepidermoid carcinoma of parotid gland. Ann R Coll Surg Engl 102:340–342. https://doi.org/10.1308/rcsann.2020.0022
PreImran S, Allen A, Shokouh-Amiri M, Garzon S, Saran N (2020) Parotid oncocytoma: CVT and pathologic correlation of a rare benign parotid tumor. Radiol Case Rep 15:31–34. https://doi.org/10.1016/j.radcr.2019.10.003
Iro H, Zenk J (2014) Salivary gland diseases in children. Curr Top Otorhinolaryngol – Head Neck Surg 13. https://doi.org/10.3205/cto000109
Rossi ED, Faquin WC, Baloch Z, Barkan GA, Pusztaszeri M, Vielh P et al (2017) The Milan System for reporting salivary gland cytopathology: analysis and suggestions of initial survey. Cancer Cytopathol 125(10):757–766. https://doi.org/10.1002/cncy.21898
Shetty A, Geethamani V (2016) Role of fine-needle aspiration cytology in the diagnosis of major salivary gland tumours: a study with histological and clinical correlation. JOMFP Oral Maxillofac Pathol 20(2):224–229. https://doi.org/10.4103/0973-029X.185899
Nader M, Bell D, Sturgis EM, Ginsberg LE, Gidley PW (2014) Facial nerve paralysis due to a Pleomorphic Adenoma with the imaging characteristics of a facial nerve Schwannoma 2014. J Neurol Surg Rep 75:e84–e88. https://doi.org/10.1055/s-0034-1368149
Lin CC, Tsui MH, Huang CC, Hua CH (2008) Parotid tumours: a 10-year experience. Amer J of Otol 29:94–100. https://doi.org/10.1016/j.amjoto.2007.03.002
Ebrahim S, Singh B, Ramklass SS (2014) HIV-associated salivary gland enlargement: a clinical review. South Afr Dent Journal 69(9):400–403
Wu B, Ngo R (2014) Cystic lymphoid hyperplasia of the parotid gland as the initial manifestation of HIV infection. Singap Med J 55(1):e12–e16. https://doi.org/10.11622/smedj.2014010
Mourad WF, Patel S, Young R, Khorsandi AS, Concert C, Shourbaji RA, Ciarrocca K et al (2016) Management algorithm for HIV-associated lymphoepithelial cysts. Eur Arch Otorhinolaryngol 273:3355–3362. https://doi.org/10.1007/s00405-016-39326-4
Jain A, Meher R, Wadhwa V, Raj A, Tandon S (2017) Parotid tuberculosis: a diagnostic dilemma. Acta Otolaryngol Case Rep 2(1):73–76. https://doi.org/10.1080/23772484.2017.130
Gupta V, Patankar K, Shinde A, Bhosale C, Tamhane A (2012) Tuberculosis of the parotid gland. Case Rep Radiol. https://doi.org/10.1155/2012/278793
Tong KN, Seltzer S, Castle JT (2020) Lipoma of the parotid gland. Head Neck Pathol 14:220–223. https://doi.org/10.1007/s12105-019-01023-3
Jamal B (2017) Treatment of parotid Non-Hodgkin Lymphoma: a Meta-analysis. J Glob Oncol. https://doi.org/10.1200/JGP.17.00071
Lu J, Zhang W, Wang Z, Jia H, Ma Y, Wu H et al (2015) Basal cell adenoma of the parotid gland: clinical and pathological findings in 29 cases. Int J Clin Exp Pathol 8(3):2899–2908
Iyer J, Hariharan A, Cao UMN, Mai CTT, Wang A, Khayambashi P et al (2021) An overview on the histogenesis and morphogenesis of salivary gland neoplasms and evolving diagnostic approaches. Cancers 13(15):3910. https://doi.org/10.3390/cancers13153910
Reerds STH, Van Engen-Van Grunsven ACH, van den Hoogen FJA, Takes RP, Marres HAM, Honings J (2021) Accuracy of parotid gland FNA cytology and reliability of the Milan System for reporting salivary gland cytopathology in clinical practice. Cancer Cytopathol. https://doi.org/10.1002/cncy.22435
Acknowledgements
We sincerely appreciate the valuable contributions and criticisms of the Postgraduate Protocol Assessment Committee of the Neurosciences Departments in the School of Clinical Medicine during the preparation phase of the proposals.
Funding
The study was self-funded.
Open access funding provided by University of the Witwatersrand.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval Statement
The study received prior ethics approval from the Human Research Ethics Committee (Medical) of University of the Witwatersrand (M180333) before it was started.
Patient Consent Statement
Patient’s consent was waived as it was retrospective record review.
Declarations
The study was self-funded and was done as part of mandatory requirement for Master of Medicine (Otorhinolaryngology) for the first author.
Conflict of Interest
All authors have no conflict of interest to declare.
Conflict of Interest
All authors have no conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mdletshe, F.B., Luvhengo, T.E. & Masege, D. The Usefulness of Fine Needle Aspiration Cytology in the Management of Parotid Gland Masses at a Tertiary Academic Hospital. Indian J Otolaryngol Head Neck Surg 75, 3199–3204 (2023). https://doi.org/10.1007/s12070-023-03685-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-023-03685-6