Abstract
Accurate classification of laryngeal cancer is a critical step for diagnosis and appropriate treatment. Radiomics is a rapidly advancing field in medical image processing that uses various algorithms to extract many quantitative features from radiological images. The high dimensional features extracted tend to cause overfitting and increase the complexity of the classification model. Thereby, feature selection plays an integral part in selecting relevant features for the classification problem. In this study, we explore the predictive capabilities of radiomics on Computed Tomography (CT) images with the incidence of laryngeal cancer to predict the histopathological grade and T stage of the tumour. Working with a pilot dataset of 20 images, an experienced radiologist carefully annotated the supraglottic lesions in the three-dimensional plane. Over 280 radiomic features that quantify the shape, intensity and texture were extracted from each image. Machine learning classifiers were built and tested to predict the stage and grade of the malignant tumour based on the calculated radiomic features. To investigate if radiomic features extracted from CT images can be used for the classification of laryngeal tumours. Out of 280 features extracted from every image in the dataset, it was found that 24 features are potential classifiers of laryngeal tumour stage and 12 radiomic features are good classifiers of histopathological grade of the laryngeal tumor. The novelty of this paper lies in the ability to create these classifiers before the surgical biopsy procedure, giving the clinician valuable, timely information.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carcinoma of the larynx is one of the most common forms of head and neck cancer and the 20th most common form of cancer overall [1]. Laryngeal cancer is caused by the spread of malignant cells in the tissues of the voice box or the larynx. The larynx has three main subsites: the supraglottis, the glottis and the sub-glottis. Common sites of the laryngeal cancer origin are the supraglottis and the glottis. Subglottic laryngeal cancers are very rare. Fig 1 shows the incidence of cancer across subsites of the larynx. A significant portion of the population is affected by laryngeal cancer, with over 184,000 new cases detected every year [2].
Computed Tomography (CT) is a radiographic imaging technique that uses a con-trast agent to acquire detailed medical images of the tissue and lesion in the larynx. CT scans are proven to be accurate, time-efficient, and quick in acquisition and computing times and are, therefore, an effective tool for diagnosing laryngeal cancer [3]. CT scans are routinely used for cancer diagnosis in India [1]. Once acquired, the radiologist analyses the CT images for the extent of the tumor and inspects for its spread into adjacent subsites and organs.
Radiomics is a rapidly advancing field that deals with the extraction of quantitative information in radiological images [4]. Radiomic features capture characteristics such as heterogeneity, shape, and texture of lesions or tissues present in the medical images. This information is often combined with other attributes for decision support analysis [5]. Radiologists can visually analyse only a finite number of features via training and experience. However, a lot of contextual information is contained in the pixels of the medial images that can be extracted and analysed [6]. This radiomic data from the medical images can be extracted and mined. Tremendous efforts are being put into discovering previously unknown markers and patterns of disease evolution, progression, and treatment response [7].
Once the Radiographic Imaging is complete (CT/MRI), a tissue sample of the lesion is surgically removed for a histopathology examination. This reveals tumor characteristics such as grade and stage. Tumor grade describes the abnormality of the tumor cells under a microscope [6. High-grade cells are well differentiated abnormal cells that have rapid growth. Lower-grade tumors are poorly differentiated and multi-ply faster than normal cells but not as much as high-grade tumor cells. The grades of laryngeal tumor have been illustrated in Fig. 2. The TNM staging system followed by the AJCC uses numbers to describe the cancer [8]. The T stands for Tumor, and the number following it represents the size of the tumor. N stands for Node, and the number describes the spread of cancer to the nodes. M indicates if the tumor metastasized [8]. The T stages and their relevance have been illustrated in Fig. 3 below.
Literature Survey
This section comprises a brief literature survey of how radiomics has been explored in laryngeal cancer.
As application of radiomics to laryngeal cancer is an emerging area of research interest, there is limited literature available. AD Kumar et al. [9] reviewed all studies (n = 15) that applied radiomics to laryngeal cancer. In the review, two studies assisted the prediction of cancer stage using radiomics. Estimation of patient survival time and prediction of prognostic factors such as treatment response were incorporated. The details of accuracy were not mentioned.
Guo et al. [10] used radiomic features computed from CT images of the larynx and investigated the accuracy of predicting thyroid cartilage invasion. They had a dataset of 265 CT scans. Using an oversampling method, they balanced their dataset and used Logistic Regression to obtain an area under the ROC curve of 0.905. They claimed that their model predicted cartilage invasion significantly better than the radiologist assessment. Radiomics can be used as a non-invasive alternative to the preoperative prediction of thyroid cartilage abnormality.
Agarwal et al. [11] worked with radiomic features of a dataset of 60 individuals analyzed after chemotherapy treatment and a follow-up period of 24 months. They concluded that Medium texture entropy, a radiomic feature, can be used as an accurate predictor for inferior local control. It could also predict survival where laryngectomy would not be necessary and also in laryngopharyngeal cancers where the stages were advanced; this feature could be used to complement clinical and radiological findings to determine prognostics. A filtration-histogram technique was used where the filtration step extracted and enhanced features of different sizes and intensity variations corresponding to a particular spatial scale filter. The ability of texture analysis to predict LFS or local control was determined using Kaplan-Meier analysis and the multivariate cox model.
Wu et al. [12], in their comprehensive multicentric study, used 211 laryngeal images for the problem of stage prediction into T3/T4. Using radiomics, peritumoral models were constructed and observed the influence of the peritumors (compared intra-tumors vs. intra-peritumors) for stage prediction. The conclusion of the study was that the dataset that used radiomic feature assessment(intra-peritumors) performed better 0.660 versus 0.579 (P-value: 0.431).
Materials and Methods
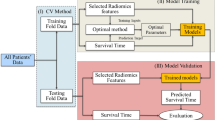
An initial round of suitable data identification and collection followed by which this data was converted to the required format and preprocessed. The radiological images and the obtained expert segmentation masks were used to obtain 280 radiomic features per image. The features were then examined for the possibility of prediction of our variables of grade and stage of the laryngeal tumor. The steps are explained in detail below. The methodology followed in the work is illustrated in Fig. 4.
Image Acquisition
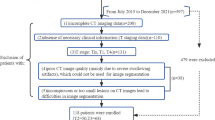
The first step was identification of patients diagnosed with laryngeal cancer. The CT images of the respective patients were retrieved in the native Digital Imaging and Communications in Medicine (DICOM) [13] format from the Picture Archival and Communication System (PACS) [14]. The histopathology reports, the radiologists’ reports and the final staging information from the discharge summary from the clinician was collected. Twenty contrast CT images that contained lesions in the supraglottis subsite were con-sidered for this study.
Image Processing and Segmentation
The contrast-enhanced CT images were converted from DICOM to NIfTI (Neu-roimaging Informatics Technology Initiative) format [15]. A window of 60,360 was chosen in the ITK-Snap tool [16] to ensure the contrast of the tumor was enhanced and visible to the radiologist for annotation. An expert radiologist manually annotated all the images slice by slice. The area of presence of tumor tissue was manually annotated for all the slices in all the images considered in our study. This comprised of the segmentation of tumor region step.
Radiomics Feature Extraction
The next step was to extract the radiomic features present in the segmented area of the images. PyRadiomics [17] was used to extract over 280 features from the segmented images. Histogram features [18], Form factor Features, GLCM features [19], Harlick features, GLSZM features [20], Texture features, Intensity features, Shape features [21, 22] and various filters were applied and the radiomic features were stored in a csv file, made ready to apply the classification algorithms.
Prediction Engine Model
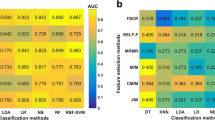
The radiomic features stored in the file were then used to predict the stage and grade of the tumor. Stages T1 and T2 were grouped as Low Stage Tumors. Stages T3 and T4 were grouped as High Stage Tumors. For the prediction of grade, the two classification classes were Grade II (moderately differentiated squamous cells) and Grade III (poorly differentiated squamous cells). The data was divided into a 80:20 split for training and validation. A support vector machine (SVM) model [23] was built by using features selected using the maximum relevance minimum redundancy (mRMR) algorithm [24]. The mRMR method ranked each feature according to its relevance to the status and redundancy with other features. An SVM score was calculated for each patient to reflect the grade and stage probability from the SVM model [25].
Results
A total of 20 patients with supraglottic cancer were included in this study. Each of the contrast CT scan was annotated with an expert radiologist slice by slice and 280 radiomic features were extracted from every image. The stage of carcinoma and histo-pathological grade of the tumors were the variables. Using the machine learning prediction engine model, feature selection was performed on the data. These radiomic features are not visible but the values can computed from the CT images. The threshold was defined as 0.49 based on the Youden Index. Patients with scores higher than the threshold were classified as high stage, while patients with scores lower than the threshold were classified as having low stage supraglottic tumor. A similar method was used to predict the grade. The prediction accuracy was 74.5% for the training group and 69.5% for the validation group. The ROC curves and scatter plots for the SVM score are presented in Fig. 5.
Table 1 lists the select radiomic features classify the stage of the tumor into high stage or low stage. Figure 6 shows the boxplots of a few of the features in Table 1.
Likewise, for the classification of the image into tumor grades, our feature prediction engine selected the radiomic features represented in Table 2. These features were found to be useful in predicting the grade of the tumor where Grade II represented the moderately differentiated squamous cells) and Grade III is poorly differentiated squamous cells. Figure 7 shows the boxplots of a few of the features in Table 2.
Conclusion
In this study, 280 features were extracted from 20 CT images with supraglottic laryngeal carcinoma. Feature selection methods determined the specific radiomic features to predict the stage into high and how and grade of the tumor cells into poorly differentiated and well-differentiated. This preliminary analysis is an encouraging result of the ability to create classifiers. The application of this work would be to provide the clinician with valuable information before conducting an invasive surgical biopsy procedure.
The future work of this study will include an increase in the size of the dataset and explore combinations of features for the prediction of prognostic markers of laryngeal cancer. The mining of radiomics data has a promising future. The conclusions drawn are robust when the study sample sizes are big, heterogeneous and multicentric. A unified research effort to standardize the annotation, segmentation and radiomics data computation practices would lead to the translation of results across centres, increasing the overall impact and usefulness.
References
Bobdey S, Jain A, Balasubramanium G (2015) Epidemiological review of laryngeal cancer: an Indian perspective. Indian J Med Paediatr Oncol 36(3):154–160. https://doi.org/10.4103/0971-5851.166721
Koroulakis A and Manuj A (2021) Laryngeal Cancer. Statpearls Publishing, https://www.ncbi.nlm.nih.gov/books/NBK526076/. Accessed 11 Apr 2022
Rao D, Singh KP (2022) Automated segmentation of the larynx on computed tomography images: a review. Biomed Eng Lett. https://doi.org/10.1007/s13534-022-00221-3
Lambin P, Leijenaar R, Deist T et al (2017) Radiomics: the bridge between medical imaging andpersonalized medicine. Nat Rev Clin Oncol 14:749–762. https://doi.org/10.1038/nrclinonc.2017.141
van Timmeren JE and Alkadhi CDTLS (2020). Radiomics in medical imaging—“how-to” guide and critical reflection Insights Imaging, 11(1): 1.
Chung CK, Stryker JA, Abt AB, Cunningham DE, Strauss M, Connor GH (1980) Histologic grading in the clinical evaluation of laryngeal carcinoma. Arch Otolaryngol 106(10):623–624. https://doi.org/10.1001/archotol.1980.00790340031008
Mayerhoefer ME, Materka A, Langs G, Häggström I, Szczypiński P, Gibbs P, Cook G (2020) Introduction to radiomics. J Nucl Med 61(4):488–495
Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP (2017) The eighth edition AJCC cancer staging manual continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin 67(2):93–99. PMID: 28094848
Rajgor AD, Patel S, McCulloch D, Obara B, Bacardit J, McQueen A, Aboagye E, Ali T, O’Hara J, Hamilton DW (2021) The application of radiomics in laryngeal cancer. Br J Radiol 94(1128):20210499
Guo R, Guo J, Zhang L, Qu X, Dai S, Peng R, Chong VFH, Xian J (2020) CT-based radiomics features in the prediction of thyroid cartilage invasion from laryngeal and hypopharyngeal squamous cell carcinoma. Cancer Imaging 20(1):81. https://doi.org/10.1186/s40644-020-00359-2 (PMID: 33176885)
Agarwal JP, Sinha S, Goda JS, Joshi K, Mhatre R, Kannan S, Laskar SG, Gupta T, Murthy V, Budrukkar A, Mummudi N, Ganeshan B (2020) Tumor radiomic features complement clinico-radiological factors in predicting long-term local control and laryngectomy free survival in locally advanced laryngo-pharyngeal cancers. Br J Radiol 93(1109):20190857
Wu X, Dong D, Zhang L, Fang M, Zhu Y, He B, Ye Z, Zhang M, Zhang S, Tian J (2021) Exploring the predictive value of additional peritumoral regions based on deep learning and radiomics: a multicenter study. Med Phys 48(5):2374–2385
Parisot C (1995) The DICOM standard. Int J Card Imaging 11(3):171–177. https://doi.org/10.1007/bf01143137
Strickland N (2000) Current topic: PACS picture archiving and communication systems—filmless radiology. Arch Dis Child 83(1):82–86. https://doi.org/10.1136/adc.83.1.82
Li X et al (2016) The first step for neuroimaging data analysis: DICOM to NIfTI conversion. J Neurosci Methods 264:47–56
Yushkevich PA, Gao Y, Gerig G (2016) “ITK-SNAP: an interactive tool for semiautomatic segmentation of multi-modality biomedical images. Engineering in Medicine and Biology Society (EMBC), 2016 IEEE 38th Annual International Conference of the IEEE, 2016
van Dijk LV, Thor M, Steenbakkers R, Apte A, Zhai TT, Borra R et al (2018) Parotid gland fat related magnetic resonance image biomarkers improve prediction of late radiation-induced xerostomia. Radiother Oncol 128(3):459–466
Van Griethuysen JJM et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77:e104–e107
Parekh V, Jacobs MA (2016) Radiomics: a new application from established techniques. Expert Rev Precis Med Drug Dev 1(2):207–226
Zwanenburg A et al (2020) The image biomarker standardization initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295(2):328–338
Xu J et al (2012) A comprehensive descriptor of shape: method and application to content-based retrieval of similar appearing lesions in medical images. J Digit Imaging 25(1):121–128
Peura M, Iivarinen J (1997) Efficiency of simple shape descriptors. Asp Vis Form 5:443–451
Hearst MA, Dumais ST, Osuna E, Platt J, Scholkopf B (1998) Support vector machines. IEEE Intell Syst Appl 13(4):18–28. https://doi.org/10.1109/5254.708428
Radovic M, Ghalwash M, Filipovic N et al (2017) Minimum redundancy maximum relevance feature selection approach for temporal gene expression data. BMC Bioinform 18:9. https://doi.org/10.1186/s12859-016-1423-9
Fluss R, Faraggi D, Reiser B (2005) Estimation of the Youden Index and its associated cutoff point. Biom J 47:458–472. https://doi.org/10.1002/bimj.200410135 (PMID: 16161804)
Acknowledgements
The authors thank all the people who contributed to the study. This research was conducted at Medical Imaging and Research Suite with equipment support from Kasturba Medical College, Manipal and partially funded by Philips Innovation Campus, Bangalore. We thank Mr. Manjunatha Maiya for his encouragement. A special mention to our colleagues for the debates over many cups of coffee.
Funding
Open access funding provided by Manipal Academy of Higher Education, Manipal. This work was supported by Manipal Academy of Higher Education, Dr. T.M.A. Pai Research Scholarship under Research Registration No. 170100107-2017 and Philips Innovation Campus, Bangalore under Exhibit B-027.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
Ethical clearance for collecting retrospective data for this non-interventional study was provided by Kasturba Medical College and Kasturba Hospital Institutional Ethics Committee. The Institutional Ethical clearance number for the study is IEC: 101/2018.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rao, D., Koteshwara, P., Singh, R. et al. Exploring Radiomics for Classification of Supraglottic Tumors: A Pilot Study in a Tertiary Care Center. Indian J Otolaryngol Head Neck Surg 75, 433–439 (2023). https://doi.org/10.1007/s12070-022-03239-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-022-03239-2