Abstract
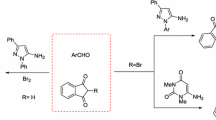
A facile and versatile method for the synthesis of alicyclic fused pyrimidones from aminoacrylates and lactams in the presence of phosphorous oxychloride is described. The results suggest that this method is widely applicable except for cyclobutyl fused systems. This method gives better yield than the method of condensing aminopyrrolidines with the beta keto esters.

Synthesis and characterization of twenty five 2-aryl-1,2-fused pyrimidone derivatives are reported. The results suggested that the methodology is widely applicable except for cyclobutyl fused systems. This method gives better yield and more versatile than the method of condensing aminopyrrolidines with the beta keto esters.






Similar content being viewed by others
References
Roberto A, Roberto B, Giorgio B and Anna M C 2007 Cyclopenta[d]pyrimidines and dihydropyrrolo[2,3-d]pyrimidines as potent and selective corticotropin-releasing factor 1 receptor antagonists Chem. Med. Chem. 2 528
Chen C, Wilcoxen K M, Huang C Q, Xie Y -F, McCarthy J R, Webb T R, Zhu Y -F, Saunders J, Liu X -J, Chen T -K, Bozigian H and Grigoriadis D E 2004 Design of 2,5- Dimethyl-3-(6-dimethyl-4-methylpyridin-3-yl)-7-dipropylaminopyrazolo[1,5-a]pyrimidine (NBI 30775/R121919) and Structure −Activity Relationships of a Series of Potent and Orally Active Corticotropin-Releasing Factor Receptor Antagonists J. Med. Chem. 47 4787
Michael C L, Yili, David L S, Maureen K S, Amy N F, Wei L, Richard L W, Kofi A O, Christopher D E, Mark W B, Steven V O, John A W and Thomas 2005 Synthesis and evaluation of tricyclic pyrrolopyrimidinones as dipeptide mimetics: Inhibition of interleukin-1 β-converting enzyme P D Bio. Med. Chem. Lett. 15 4322
Bashir A D, Praveen P, Sunil K, Mohammad A W, Akshay K S, Parduman R S, Sanjay P, Meena S and Baldev S 2013 Fe–Al/clay as an efficient heterogeneous catalyst for solvent-free synthesis of 3, 4-dihydropyrimidones J. Chem. Sci. 125 545
Steve C, Chris Y, Stuart F, Gareth B, John B, Osamu I, Elise G, Anthony R, Takashi K, Akira I, Shingo N, Ryo S and Sho K 2013 Pyridinone and pyrimidinone derivatives as factor xia inhibitors Int. Appl. WO2013093484 A1
Benjamin S and Henry S 1956 A new synthesis of 2 : 3 : 5 : 6-substituted 4-pyrimidones J. Chem. Soc. 4708
Oripov E O, Zakhidov K A and Shakhidoyatov Kh M 1991 Synthesis of bicyclic analogues of deoxyvasicinone Chem. Nat. Comp. 27 341
Abdillahi I and Kirsch G 2011 Synthesis of a Series of Novel Thieno- and Benzothieno[3,2-d]pyrimidin-4(3 H)-ones Synthesis 1314
Rokach J, Hamel P, Hunter N R, Reader G and Rooney C S 1979 Cyclic amidine inhibitors of indolamine N-methyltransferase J. Med. Chem. 22 237
Blaise E E 1901 New reactions of the organo metal derivatives C.R. Hebd. Seances Acad. Sci. 132 478
Lee J H, Choi B S, Chang J H, Lee J B, Yoon J Y, Lee J and Shin H 2007 The Decarboxylative Blaise Reaction J. Org. Chem. 72 1026
Toussaint O, Capdevielle P and Maumy M 1986 The Copper(I)-catalyzed decarboxylation of malonic acids; A new mild and quantitative method Synthesis 1029
Darensbourg D J, Holtcamp M W, Khandelwal B and Reibenspies J H 1994 Intramolecular and intermolecular hydrogen-bonding in triphenylphosphine derivatives of Copper(I) carboxylates, (Ph3P)2CuO2C(CH2)nCOOH. Role of copper(I) in the decarboxylation of malonic-acid and its derivatives Inorg. Chem. 33 531
Hassner A and Stumer C 2002 In Organic synthesis based on name reactions 2nd Ed. (Oxford: Elsevier)
Mundy B P, Ellerd M G and Favaloro F G 2005 In Name reactions and reagents in organic synthesis (Canada: Wiley Interscience)
Rao H S P, Rafi S and Padmavathy K 2008 The Blaise Reaction Tetrahedron 64 8037
Andre E, Andre L B and Christian R 1966 Synthesis of pyrrolopyrimidinones from 2-amino-1-pyrroline 1966 C. R. Sean. Acad. Sci., Serie. C: Sci. Chim. 262 365
Jozsef V, Pal K, Miklos J and Gyorgy S 1988 Effect of changes in phase composition on retention values. I. Reproducibility of RMO values in open and closed chromatographic systems Acta Pharm. Hung. 58 8
Aude F, Thierry G, Alistair L, Mourad S, Corinne V and Assignee Y P 2008 Sanofi-Aventis, Fr.; Mitsubishi Tanabe Pharma Corporation, substituted heteroaryl pyridopyrimidone derivatives WO 2008078196
Grigoryan N P, Tarzyan L A, Markosyan A I, Paronikyan R G and Sukasyan R S 2011 Synthesis and psychotropic activity of 5-cyclohexyl-5-methyl-2-sulfanyl-3,4,5,6-tetrahydrobenzo[h]quinazolin-4-one Pharm. Chem. J. 45 79
Acknowledgements
Authors thank Department of Discovery Analytical Services of Biocon Bristol-Myers Squibb Research Center for their valuable support and, Jianqing Li, Richard Rampulla and Bei Wang of Bristol Myers Squibb for logistic help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information (SI)
Characerization data and Figures S1–S25 for all the compounds are provided in the Supplementary Information which is available at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
RAMESH, S., SARAVANAKUMAR, P., DURAISAMY, R. et al. One-pot synthesis of 2-aryl-1,2-fused pyrimidones. J Chem Sci 129, 329–333 (2017). https://doi.org/10.1007/s12039-017-1244-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-017-1244-z