Abstract
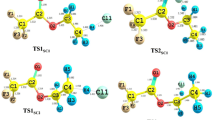
The kinetics of the reaction between pinonaldehyde (C 10 H 16 O 2) and Cl atom were studied using high level ab initio G3(MP2) and DFT based MPWB1K/6-31 + G(d) and MPW1K/6-31 + G(d) levels of theories coupled with Conventional Transition State Theory in the temperature range between 200 and 400 K. The negative temperature dependent rate expression for the title reaction obtained with Wigner’s and Eckart’s symmetrical tunneling corrections are k(T) =(5.1 ± 0.56) × 10 −19 T 2.35exp[(2098 ± 2)/T] cm 3 molecule −1 s −1, and k(T) =(0.92 ± 0.18) × 10 −19 T 2.60exp[(2204 ± 4)/T] cm 3 molecule −1 s −1, respectively, at G3(MP2)//MPWB1K method. The H abstraction reaction from the –CHO group was found to be the most dominant reaction channel among all the possible reaction pathways and its corresponding rate coefficient at 300 K is k(Eckart’s unsymmetrical) = 3.86 ×10−10 cm 3 molecule −1 s −1. Whereas the channel with immediate lower activation energy is the H-abstraction from –CH- group (Tertiary H-abstraction site, C g). The rate coefficient for this channel is k Cg(Eckart’s unsymmetrical) = 1.83 ×10−15 cm 3 molecule −1 s −1 which is smaller than the dominant channel by five orders of magnitude. The atmospherically relevant parameters such as lifetimes were computed in this investigation of its reaction with Cl atom.

The kinetics of the reaction between pinonaldehyde (C10H16O2) and Cl atom were studied computationally and computed the rate coefficients in the temperature range between 200 and 400 K. The H abstraction reaction from the –CHO group is found to be the most dominant reaction channel among all the possible reaction pathways.




Similar content being viewed by others
References
Guenther A, Hewitt C N, Erickson D, Fall R, Geron C, Graedel T, Harley P, Klinger L, Lerdau M and McKay W A 1995 J. Geophys. Res. 100 8873
Guenther A, Geron C, Pierce T, Lamb B, Harley P and Fall R 1996 Atmos. Environ. 34 2205
Fall R and Hewitt C N (Ed.) 1999 In Reactive Hydrocarbons in the Atmosphere (San Diego: Academic Press) p. 41
Fuentes J D, Lerdau M, Atkinson R, Baldocchi D, Bottenheim J W, Ciccioli P, Lamb B, Geron C, Gu L, Guenther A, Sharkey T D and Stockwell W 2000 B. Am. Met. Soc. 81 1537
Geron C, Rasmussen R, Arnts R R and Guenther A 2000 Atmos. Environ. 34 1761
Atkinson R, Baulch D L, Cox R A, Crowley J N, Hampson R F, Hynes R G, Jenkin M E, Rossi M J and Troe J 2006 Atmos. Chem. Phys. 6 3625
Singh H B, Thakur A N, Chen Y E and Kanakidou M 1996 Geophys. Res. Lett. 23 1523
Spicer C W, Chapman E G, Finlayson-Pitts B J, Plastridge R A, Hubbe J M, Fast J D and Berkowitz C M 1998 Nature 394 353
Platt U, Allan W and Lowe D 2004 Atmos. Chem. Phys. 4 2393
Wingenter O W, Kubo M K, Blake N J, Smith Jr. T W, Blake D R and Rowland F S 1996 J. Geophys. Res. 101 4331
Graedel T E and Keene W C 1995 Global Biogeochem. Cycl. 9 47
DeHaan D O, Brauers T, Oum K, Stutz J, Nordmeyer T and Finlayson-Pitts B J 1999 Int. Rev. Phys. Chem. 18 343
Keene W C 1995 In Inorganic C1 Cycling in the Marine Boundary Layer: A Review In Naturally Produced Organohalogens A Grimvall A and E W B de Leer (Eds.) (Dordrecht, The Netherlands: Kluwer Academic) p. 363
Keene W C, Jacob D J and Fan S M 1996 Atmos. Environ. 30 R1–R3
DeMore W B, Sander S P, Golden D M, Hampson R F, Kurylo M J, Howard C J, Ravishankara A R, Kolb C E and Molina M J 1997 In Chemical Kinetics a Photochemical Data for Use in Stratospheric Modeling Evaluation Number 12 (California Institute of Technology, Pasadena, CA: J P L Publication) pp. 97-4
Atkinson R, Baulch D L, Cox R A, Hampson Jr. R F, Kerr J A, Rossi M J and Troe J 1997 J. Phys. Chem. Ref. Data 26 521
Atkinson R J 1997 J. Phys. Chem. Ref. Data 26 215
Davis M E, Talukdar R K, Notte G, Ellison G B and Burkholder J B 2007 Environ. Sci. Technol. 41 3959
Hallquist M, Wängberg I and Ljungström E 1997 Environ. Sci. Technol. 31 3166
Alvarado A, Arey J and Atkinson R 1998 J. Atmos. Chem. 31 281
Glasius M, Galogirou A, Jensen N R, Hjorth J and Nielsen C J 1997 Int. J. Chem. Kinet. 29 527
Nozière B, Spittler M, Ruppert L, Barnes I, Becker K H, Pons M and Wirtz K 1999 Int. J. Chem. Kinet. 31 291
Lynch B J, Fast P L, Harris M and Truhlar D G 2000 J. Phys. Chem. A 104 4811
Zhao Y and Truhlar D G 2004 J. Phys. Chem. A 108 6908
Frisch M J et al. 2010 Gaussian 09, Revision B.01 (Gaussian, Inc.: Wallingford CT
Zhao Y, Schultz N E and Truhlar D G 2006 J. Chem. Theor. Comput. 2 364
Chandra A K 2012 J. Mol. Model. 18 4239
Fernandez-Ramos A, Miller J A, Klippenstein S J and Truhlar D G 2006 Chem. Rev. 106 4518
Curtiss L A, Redfern P C, Raghavachari K, Rassolov V and Pople J A 1999 J. Chem. Phys. 110 4703
Dennington I I R, Keith T, Millam J, Eppinnett K, Hovell W L and Gilliland R 2003 GaussView, Version 3.09 (Semichem, Inc.: Shawnee Mission, KS)
Wright M R 1999 In Fundamental Chemical Kinetics and Exploratory Introduction to the Concepts (Chichester UK: Ellis Horwood)
Ralchenko Y, Jou F C, Kelleher D E, Kramida A E, Musgrove A, Reader J, Wiese W L and Olsen K 2007 NIST Atomic Spectra Database, version 3.1.2 (National Institute of Standards and Technology: Gaithersburg, MD)
Wigner E P Z 1932 Phys. Chem. B 19 203
Eckart C 1930 Phys. Rev. 35 1303
Johnston H S and Heicklen J 1962 J. Phys. Chem. 66 532
Louis F, Gonzalez C A, Huie R E and Kurylo M J 2000 J. Phys. Chem. A 104 8773
Ali M A and Rajakumar B 2010 J. Comput. Chem. 31 500
Ali M A and Rajakumar B 2010 J. Mol. Struct: THEOCHEM 949 73
Kaliginedi V, Ali M A and Rajakumar B 2012 Int. J. Quantum Chem. 112 1066
Ali M A and Rajakumar B 2011 Int. J. Chem. Kinet. 43 418
Ali M A, Upendra B and Rajakumar B 2011 Chem. Phys. Lett. 511 440
Dash M R and Rajakumar B 2012 J. Phys. Chem. A 116 5856
Korchowiec J, Kawahara S I, Matsumura K, Uchimara T and Surgie M 1999 J. Phys. Chem. A 103 3548
Chandra A K and Uchimaru T 2000 J. Phys. Chem. A 104 8535
Alvarez-Idaboy J R, Cruz-Torres A, Galano A and Ruiz-Santoyo M E 2004 J. Phys. Chem. A 108 2740
Zhang M, Lin Z and Song C 2007 J. Chem. Phys. 126 34307
Chuang Y and Truhlar D G 2000 J. Chem. Phys. 112 1221
Gruber-Stadler M, Mühlhäuser M, Sellevåg S R and Nielsen C J 2008 J. Phys. Chem. A 112 9
Beukes J A, D’Anna B, Bakken V and Nielsen C J 2000 Phys. Chem. Chem. Phys. 2 4049
Alvarez-Idaboy J R, Mora-Diez N, Boyd R J and Vivier-Bunge A 2001 J. Am. Chem. Soc. 123 2018
Alvarez-Idaboy J R, Mora-Diez N and Vivier-Bunge A 2000 J. Am. Chem. Soc. 122 3715
Galano A, Alvarez-Idaboy J R, Ruiz-Santoyo M. E. and Vivier-Bunge A 2005 J. Phys. Chem. A 109 169
Vereecken L and Peeters J 2002 Phys. Chem. Chem. Phys. 4 467
Wang Y, Liu J -y, Li Z., Wang L, Wu J -y and Sun C -c 2006 J. Phys. Chem. A 110 5853
Sun H and Law C K 2010 J. Phys. Chem. A 114 12088
Gao H, Liu J -y and Sun C -c 2009 J. Chem. Phys. 130 224301
Srinivasulu G and Rajakumar B 2013 J. Phys. Chem. A 117 4534
Aschmann S M and Atkinson R 1995 Int. J. Chem. Kinet. 27 613
Tyndall G S, Orlando J J, Wallington T J, Dill M and Kaiser E W 1997 Int. J. Chem. Kinet. 29 43
Acknowledgements
G.S. and B.R. are very grateful to the Ministry of Earth Sciences, Government of India for funding. We thank Mr. V. Ravichandaran, High performance computing centre (HPCE), Indian Institution of Technology Madras, Chennai, for providing us computational facility.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information (SI)
The electronic supporting information can be seen at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
SRINIVASULU, G., RAJAKUMAR, B. Rate coefficients for hydrogen abstraction reaction of pinonaldehyde (C10H16O2) with Cl atoms between 200 and 400 K: A DFT study. J Chem Sci 128, 977–989 (2016). https://doi.org/10.1007/s12039-016-1075-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-016-1075-3