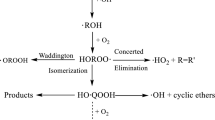
Abstract
A theoretical investigation has been carried out on the mechanism, kinetics, and thermochemistry of gas-phase reaction of 2,2,2-trifluoroethyl butyrate (TFEB, CH3CH2CH2C(O)OCH2CF3) with OH radicals using a modern DFT functional. The involvement of pre- and post-reactive complexes was explored and the reaction profiles were modeled. Energetic calculations were performed using the M06-2X/6-31 + G(d,p) method. The intrinsic reaction coordinate (IRC) calculation has been performed to confirm the smooth transition from the reactant to product through the respective transition state. It has been found that the dominant path of the H-atom abstraction takes place from the –CH2- position, which is attached with the methyl group at the one end of TFEB. Theoretically calculated rate constant at 298 K using canonical transition state theory (CTST) is found to be in reasonable agreement with the experimental data. Using group-balanced isodesmic procedure, the standard enthalpy of formation for TFEB is reported for the first time. The branching ratios of the different reaction channels are also determined. The atmospheric lifetime of TFEB is determined to be 6.8 days.

The reaction kinetics of H-atom abstraction reaction of 2,2,2-trifluoroethyl butyrate (TFEB) with OH radicals was investigated at M06-2X/6-31+G (d,p) level of theory. The branching ratios of the different reaction channels are also determined. The atmospheric lifetime of TFEB is estimated to be 6.8 days.



Similar content being viewed by others
References
Tsai W T 2005 J. Hazard. Mater. 119 69
Sekiya A and Misaki S 2000 J. Fluorine Chem. 101 215
Powell R L 2002 J. Fluorine Chem. 114 237
Bravo I, Dıaz-de-Mera Y, Aranda A, Moreno E, Nutt D R and Marston G 2011 Phys. Chem. Chem. Phys. 13 17185
Blanco M B, Bejan I, Barnes I, Wiesen P and Teruel M A 2010 Environ. Sci. Technol. 44 (4) 2354
Oyaro N, Sellevag S R and Neilsen C J 2004 Environ. Sci. Technol. 38 5567
Jordan A and Frank H 1999 Environ. Sci. Technol. 33 (2) 522
Ninomiya Y, Kawasaki M, Guschin A, Molina L T, Molina M J and Wallington T J 2000 Environ. Sci. Technol. 34 (14) 2973
Blanco M B, Barnes I and Teruel M A 2010 J. Phys. Org. Chem. 23 950
Blanco M B, Bejan I, Barnes I, Wiesen P and Teruel M A 2008 Chem. Phys. Lett. 453 18
Blanco M B and Teruel M A 2007 Atmos. Environ. 41 (34) 7330
Blanco M B, Rivela C and Teruel M A 2013 Chem. Phys. Lett. 578 33
Chakrabartty A K, Mishra B K, Bhattacharjee D and Deka R C 2013 Mol. Phys. 111 860
Mishra B K, Chakrabartty A K and Deka R C 2013 J. Mol. Model. 19 2189
Singh H J, Tiwari L and Rao P K 2014 Mol. Phys. 112 1892
Mishra B K, Chakrabartty A K and Deka R C 2014 Struct. Chem. 25 463
Mishra B K and Deka R C 2014 Struct. Chem. 25 1475
Mishra B K, Singh H J and Tiwari L 2014 J. Mol. Model. 20 2475
Mishra B K and Deka R C 2014 J. Phys. Chem. A 118 (38) 8779
Zhao Y and Truhlar D G 2008 Theor. Chem. Acc. 120 215
Lily M, Mishra B K and Chandra A K 2014 J. Fluorine Chem. 161 51
Dinadayalane T C, Paytakov G and Leszczynski J 2013 J. Mol. Model. 19 2855
Deka R C and Mishra B K 2014 J. Mol. Graph. Model. 53 23
Sandhiya L, Kolandaivel P and Senthilkumar K 2012 Struct. Chem. 23 1475
Gour N K, Deka R C, Singh H J and Mishra B K 2014 J. Fluorine Chem. 160 64
Chakrabartty A K, Mishra B K, Bhattacharjee D and Deka R C 2013 J. Fluorine Chem. 154 60
Mishra B K, Lily M, Chakrabartty A K, Deka R C and Chandra A K 2014 J. Fluorine Chem. 159 57
Gonzalez C and Schlegel H B 1989 J. Chem. Phys. 90 2154
Frisch M J et al. 2009 In Gaussian 09 Revision- B.01 Gaussian, Inc.: Wallingford C T)
Hammond G S 1955 J. Am. Chem. Soc. 77 334
Laidler K J 2004 In Chemical Kinetics, 3rd edn (New Delhi: Pearson Education)
Johnston H S and Heicklen J 1962 J. Phys. Chem. 66 532
Chuang Y Y and Truhlar D G 2000 J. Chem. Phys. 112 1221
Kwok E S C and Atkinson R 1995 Atmos. Environ. 29 1685
Spicer C W, Chapman E G, Finlayson-Pitts B J, Plastridge R A, Hubbe J M, Fast J D and Berkowitz C M 1998 Nature 394 353
Bravo I, Aranda A, Hurley M D, Marston G, Nutt D R, Shine K P, Smith K and Wallington T J 2010 J. Geophys. Res. 115 D24317
Spivakovsky C M, Logan J A, Montzka S A, Balkanski Y J, Foreman-Fowler M, Jones D B A, Horowitz L W, Fusco A C, Brenninkmeijer C A M, Prather M J, Wofsy S C and McElroy M B 2000 J. Geophys. Res. 105 (D7) 8931
Chase M W Jr, NIST-JANAF Thermochemical Tables, 4th edition. J. Phys. Chem. Ref. Data 9 1 (1998)
Witter R A and Neta P 1973 J. Org. Chem. 38 484
Gao H, Wang Y, Wan S Q, Liu J Y and Sun C C 2009 J. Mol. Struct. Theochem. 913 107
El-Nahas A M, Navarro M V, Simmie J M, Bozzelli J W, Curran H J, Dooley S and Metcalfe W 2007 J. Phys. Chem. A 111 3727
Csontos J, Rolik Z, Das S and Kallay M 2010 J. Phys. Chem. A 114 13093
Chen S S, Rodgers A S, Chao J, Wilhoit R C and Zwolinski B J 1975 J. Phys. Chem. Ref. Data 4 441
Lide D R 2009 In In CRC Handbook of Chemistry and Physics 89th ed. (New York: CRC Press)
Acknowledgements
BKM is thankful to University Grant Commission (UGC), New Delhi, for providing Dr. D. S. Kothari Post Doctoral Fellowship. NKG and HJS are thankful to Council of Scientific and Industrial Research (CSIR), New Delhi, for providing financial assistance.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information
Harmonic vibrational frequencies of the species, relative energies, calculated partition functions, and IRC plots for transition states at the M06-2X/6-31+G(d,p) level of theory are available at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
GOUR, N.K., MISHRA, B.K. & SINGH, H.J. Theoretical study on mechanism, kinetics, and thermochemistry of the gas phase reaction of 2,2,2-trifluoroethyl butyrate with OH radicals at 298 K. J Chem Sci 127, 1015–1023 (2015). https://doi.org/10.1007/s12039-015-0860-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-015-0860-8