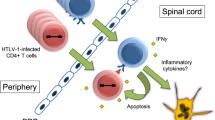
Abstract
Human T lymphotropic virus–associated myelopathy/tropical spastic paraparesis (HTLV/TSP), also known as HTLV-associated myelopathy/tropical spastic paraparesis (HAM/TSP), and multiple sclerosis (MS) are chronic debilitating diseases of the central nervous system; although the etiology of which is different, similarities have been observed between these two demyelinating diseases, especially in clinical manifestation and immunopathogenesis. Exorbitant response of the immune system to the virus and neurons in CNS is the causative agent of HAM/TSP and MS, respectively. Helper T lymphocyte-17 cells (Th17s), a component of the immune system, which have a proven role in immunity and autoimmunity, mediate protection against bacterial/fungal infections. The role of these cells has been reviewed in several CNS diseases. A pivotal role for Th17s is presented in demyelination, even more axial than Th1s, during MS. The effect of Th17s is not well determined in HTLV-1-associated infections; however, the evidence that we have supplied in this review illustrates the attendance, also the role of Th17 cells during HAM/TSP. Furthermore, for better conception concerning the trace of these cells in HAM/TSP, a comparative characterization with MS, the resembling disease, has been applied here.


Similar content being viewed by others
Data Availability
Not applicable.
References
Ernzen KJ, Panfil AR (2022) Regulation of HTLV-1 transformation. Biosci Rep 42(3)
Nozuma S, Jacobson S (2019) Neuroimmunology of human T-lymphotropic virus type 1-associated myelopathy/tropical spastic paraparesis. Front Microbiol 10:885
Azodi S, Nair G, Enose-Akahata Y, Charlip E, Vellucci A, Cortese I et al (2017) Imaging spinal cord atrophy in progressive myelopathies: HTLV-I-associated neurological disease (HAM/TSP) and multiple sclerosis (MS). Ann Neurol 82(5):719–728
Oger J (2007) HTLV-1 infection and the viral etiology of multiple sclerosis. J Neurol Sci 262(1-2):100–104
Bangham CR, Araujo A, Yamano Y, Taylor GP (2015) HTLV-1-associated myelopathy/tropical spastic paraparesis. Nat Rev Dis Primers 1:15012
Bangham CRM (2018) Human T cell leukemia virus type 1: persistence and pathogenesis. Annu Rev Immunol 36:43–71
Futsch N, Prates G, Mahieux R, Casseb J, Dutartre H (2018) Cytokine networks dysregulation during HTLV-1 infection and associated diseases. Viruses 10(12)
Sospedra M, Martin R (2016) Immunology of multiple sclerosis. Semin Neurol 36(2):115–127
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25:821–852
Bedoya SK, Lam B, Lau K, Larkin J 3rd. (2013) Th17 cells in immunity and autoimmunity. Clin Dev Immunol 2013:986789
Yasuda K, Takeuchi Y, Hirota K (2019) The pathogenicity of Th17 cells in autoimmune diseases. Semin Immunopathol 41(3):283–297
Nakagawa M, Izumo S, Ijichi S, Kubota H, Arimura K, Kawabata M et al (1995) HTLV-I-associated myelopathy: analysis of 213 patients based on clinical features and laboratory findings. J Neurovirol 1(1):50–61
Hofstetter H, Gold R, Hartung HP (2009) Th17 cells in MS and experimental autoimmune encephalomyelitis. Int MS J 16(1):12–18
Waisman A, Hauptmann J, Regen T (2015) The role of IL-17 in CNS diseases. Acta Neuropathol 129(5):625–637
Beurel E, Lowell JA (2018) Th17 cells in depression. Brain Behav Immun 69:28–34
Maciak K, Pietrasik S, Dziedzic A, Redlicka J, Saluk-Bijak J, Bijak M et al (2021) Th17-related cytokines as potential discriminatory markers between neuromyelitis optica (Devic’s disease) and multiple sclerosis-a review. Int J Mol Sci 22(16)
Luckheeram RV, Zhou R, Verma AD, Xia B (2012) CD4(+)T cells: differentiation and functions. Clin Dev Immunol 2012:925135
Mosmann TR, Cherwinski H, Bond MW, Giedlin MA, Coffman RL (1986) Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol 136(7):2348–2357
Chang YC, Hee SW, Chuang LM (2021) T helper 17 cells: a new actor on the stage of type 2 diabetes and aging? J Diabetes Investig 12(6):909–913
Hueber W, Sands BE, Lewitzky S, Vandemeulebroecke M, Reinisch W, Higgins PD et al (2012) Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn’s disease: unexpected results of a randomised, double-blind placebo-controlled trial. Gut 61(12):1693–1700
Majumder S, McGeachy MJ (2021) IL-17 in the pathogenesis of disease: good intentions gone awry. Annu Rev Immunol 39:537–556
Stadhouders R, Lubberts E, Hendriks RW (2018) A cellular and molecular view of T helper 17 cell plasticity in autoimmunity. J Autoimmun 87:1–15
Harbour SN, Maynard CL, Zindl CL, Schoeb TR, Weaver CT (2015) Th17 cells give rise to Th1 cells that are required for the pathogenesis of colitis. Proc Natl Acad Sci USA 112(22):7061–7066
Wu X, Tian J, Wang S (2018) Insight into non-pathogenic Th17 cells in autoimmune diseases. Front Immunol 9:1112
Peters A, Lee Y, Kuchroo VK (2011) The many faces of Th17 cells. Curr Opin Immunol. 23(6):702–706
Volchenkov R, Nygaard V, Sener Z, Skalhegg BS (2017) Th17 Polarization under hypoxia results in increased IL-10 production in a pathogen-independent manner. Front Immunol 8:698
Li MO, Flavell RA (2008) TGF-beta: a master of all T cell trades. Cell 134(3):392–404
Zhang S (2018) The role of transforming growth factor beta in T helper 17 differentiation. Immunology 155(1):24–35
Korn T, Bettelli E, Oukka M, Kuchroo VK (2009) IL-17 and Th17 cells. Annu Rev Immunol 27:485–517
Kimura A, Kishimoto T (2010) IL-6: regulator of Treg/Th17 balance. Eur J Immunol 40(7):1830–1835
Zhou L, Ivanov II, Spolski R, Min R, Shenderov K, Egawa T et al (2007) IL-6 programs T(H)-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol 8(9):967–974
Wu B, Wan Y (2020) Molecular control of pathogenic Th17 cells in autoimmune diseases. Int Immunopharmacol 80:106187
Ahern PP, Schiering C, Buonocore S, McGeachy MJ, Cua DJ, Maloy KJ et al (2010) Interleukin-23 drives intestinal inflammation through direct activity on T cells. Immunity 33(2):279–288
Tesmer LA, Lundy SK, Sarkar S, Fox DA (2008) Th17 cells in human disease. Immunol Rev 223:87–113
McGeachy MJ, Bak-Jensen KS, Chen Y, Tato CM, Blumenschein W, McClanahan T et al (2007) TGF-beta and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain T(H)-17 cell-mediated pathology. Nat Immunol 8(12):1390–1397
Lee Y, Awasthi A, Yosef N, Quintana FJ, Xiao S, Peters A et al (2012) Induction and molecular signature of pathogenic TH17 cells. Nat Immunol 13(10):991–999
Lee PW, Yang Y, Racke MK, Lovett-Racke AE (2015) Analysis of TGF-beta1 and TGF-beta3 as regulators of encephalitogenic Th17 cells: implications for multiple sclerosis. Brain Behav Immun 46:44–49
Ghoreschi K, Laurence A, Yang XP, Tato CM, McGeachy MJ, Konkel JE et al (2010) Generation of pathogenic T(H)17 cells in the absence of TGF-beta signalling. Nature 467(7318):967–971
Li YF, Zhang SX, Ma XW, Xue YL, Gao C, Li XY (2017) Levels of peripheral Th17 cells and serum Th17-related cytokines in patients with multiple sclerosis: a meta-analysis. Mult Scler Relat Disord 18:20–25
Miyatake Y, Ikeda H, Ishizu A, Baba T, Ichihashi T, Suzuki A et al (2006) Role of neuronal interferon-gamma in the development of myelopathy in rats infected with human T-cell leukemia virus type 1. Am J Pathol 169(1):189–199
Refaat A, Zhou Y, Suzuki S, Takasaki I, Koizumi K, Yamaoka S et al (2011) Distinct roles of transforming growth factor-beta-activated kinase 1 (TAK1)-c-Rel and interferon regulatory factor 4 (IRF4) pathways in human T cell lymphotropic virus 1-transformed T helper 17 cells producing interleukin-9. J Biol Chem 286(24):21092–21099
Nishimoto N, Yoshizaki K, Eiraku N, Machigashira K, Tagoh H, Ogata A et al (1990) Elevated levels of interleukin-6 in serum and cerebrospinal fluid of HTLV-I-associated myelopathy/tropical spastic paraparesis. J Neurol Sci 97(2-3):183–193
Brites C, Abrahao M, Bozza P, Netto EM, Lyra A, Bahia F (2018) Infection by HTLV-1 is associated with high levels of proinflammatory cytokines in HIV-HCV-coinfected patients. J Acquir Immune Defic Syndr 77(2):230–234
Aranami T, Yamamura T (2008) Th17 Cells and autoimmune encephalomyelitis (EAE/MS). Allergol Int 57(2):115–120
Hiltensperger M, Korn T (2018) The interleukin (IL)-23/T helper (Th)17 axis in experimental autoimmune encephalomyelitis and multiple sclerosis. Cold Spring Harb Perspect Med 8(1)
Mah AY, Cooper MA (2016) Metabolic regulation of natural killer Cell IFN-gamma production. Crit Rev Immunol 36(2):131–147
Alspach E, Lussier DM, Schreiber RD (2019) Interferon gamma and its important roles in promoting and inhibiting spontaneous and therapeutic cancer immunity. Cold Spring Harb Perspect Biol 11(3)
Tang C, Chen S, Qian H, Huang W (2012) Interleukin-23: as a drug target for autoimmune inflammatory diseases. Immunology 135(2):112–124
Arellano G, Ottum PA, Reyes LI, Burgos PI, Naves R (2015) Stage-specific role of interferon-gamma in experimental autoimmune encephalomyelitis and multiple sclerosis. Front Immunol 6:492
Dungan LS, McGuinness NC, Boon L, Lynch MA, Mills KH (2014) Innate IFN-gamma promotes development of experimental autoimmune encephalomyelitis: a role for NK cells and M1 macrophages. Eur J Immunol 44(10):2903–2917
Moore KW, de Waal Malefyt R, Coffman RL, O’Garra A (2001) Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol 19:683–765
Zu DM, Zhang L (2022) Assessment of mechanisms of infectious pneumonia based on expression of fibrinogen, procalcitonin, high-sensitivity C-reactive protein expression, T helper 17 cells, regulatory T cells interleukin-10, and interleukin-17. Transl Pediatr 11(1):73–84
Beebe AM, Cua DJ, de Waal MR (2002) The role of interleukin-10 in autoimmune disease: systemic lupus erythematosus (SLE) and multiple sclerosis (MS). Cytokine Growth Factor Rev 13(4-5):403–412
Vogel DY, Kooij G, Heijnen PD, Breur M, Peferoen LA, van der Valk P et al (2015) GM-CSF promotes migration of human monocytes across the blood brain barrier. Eur J Immunol 45(6):1808–1819
Uzawa A, Mori M, Kuwabara S (2014) Cytokines and chemokines in neuromyelitis optica: pathogenetic and therapeutic implications. Brain Pathol 24(1):67–73
Himes SR, Coles LS, Katsikeros R, Lang RK, Shannon MF (1993) HTLV-1 tax activation of the GM-CSF and G-CSF promoters requires the interaction of NF-kB with other transcription factor families. Oncogene 8(12):3189–3197
Zhang L, Yuan S, Cheng G, Guo B (2011) Type I IFN promotes IL-10 production from T cells to suppress Th17 cells and Th17-associated autoimmune inflammation. PLoS One 6(12):e28432
Stumhofer JS, Silver JS, Laurence A, Porrett PM, Harris TH, Turka LA et al (2007) Interleukins 27 and 6 induce STAT3-mediated T cell production of interleukin 10. Nat Immunol 8(12):1363–1371
Yamashita I, Katamine S, Moriuchi R, Nakamura Y, Miyamoto T, Eguchi K et al (1994) Transactivation of the human interleukin-6 gene by human T-lymphotropic virus type 1 Tax protein. Blood 84(5):1573–1578
Chabaud M, Garnero P, Dayer JM, Guerne PA, Fossiez F, Miossec P (2000) Contribution of interleukin 17 to synovium matrix destruction in rheumatoid arthritis. Cytokine 12(7):1092–1099
Dodon MD, Li Z, Hamaia S, Gazzolo L (2004) Tax protein of human T-cell leukaemia virus type 1 induces interleukin 17 gene expression in T cells. J Gen Virol 85(Pt 7):1921–1932
Sarkis S, Belrose G, Peloponese JM Jr, Olindo S, Cesaire R, Mesnard JM et al (2013) Increased osteopontin expression in HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) patient cells is associated with IL-17 expression. J Clin Virol 58(1):295–298
Subramanian K, Dierckx T, Khouri R, Menezes SM, Kagdi H, Taylor GP et al (2019) Decreased RORC expression and downstream signaling in HTLV-1-associated adult T-cell lymphoma/leukemia uncovers an antiproliferative IL17 link: a potential target for immunotherapy? Int J Cancer 144(7):1664–1675
Leal FE, Ndhlovu LC, Hasenkrug AM, Bruno FR, Carvalho KI, Wynn-Williams H et al (2013) Expansion in CD39(+) CD4(+) immunoregulatory t cells and rarity of Th17 cells in HTLV-1 infected patients is associated with neurological complications. PLoS Negl Trop Dis 7(2):e2028
Carvalho NB, de Lourdes BM, Souza AS, Netto EM, Arruda S, Santos SB et al (2018) Impaired TNF, IL-1beta, and IL-17 production and increased susceptibility to Mycobacterium tuberculosis infection in HTLV-1 infected individuals. Tuberculosis (Edinb) 108:35–40
Neco H, Teixeira VGS, Trindade ACL, Magalhaes PMR, Lorena VMB, Vasconcelos LR et al (2017) IL17A polymorphism is not associated with human T-lymphotropic virus 1-associated myelopathy/tropical spastic paraparesis. Viral Immunol 30(4):298–301
Yamano Y, Araya N, Sato T, Utsunomiya A, Azakami K, Hasegawa D et al (2009) Abnormally high levels of virus-infected IFN-gamma+ CCR4+ CD4+ CD25+ T cells in a retrovirus-associated neuroinflammatory disorder. PLoS One 4(8):e6517
Seich Al Basatena NK, Macnamara A, Vine AM, Thio CL, Astemborski J, Usuku K et al (2011) KIR2DL2 enhances protective and detrimental HLA class I-mediated immunity in chronic viral infection. PLoS Pathog. 7(10):e1002270
Jeffery KJ, Usuku K, Hall SE, Matsumoto W, Taylor GP, Procter J et al (1999) HLA alleles determine human T-lymphotropic virus-I (HTLV-I) proviral load and the risk of HTLV-I-associated myelopathy. Proc Natl Acad Sci USA 96(7):3848–3853
Espinoza JL, Takami A, Nakata K, Onizuka M, Kawase T, Akiyama H et al (2011) A genetic variant in the IL-17 promoter is functionally associated with acute graft-versus-host disease after unrelated bone marrow transplantation. PLoS One 6(10):e26229
Nordang GB, Viken MK, Hollis-Moffatt JE, Merriman TR, Forre OT, Helgetveit K et al (2009) Association analysis of the interleukin 17A gene in Caucasian rheumatoid arthritis patients from Norway and New Zealand. Rheumatology (Oxford) 48(4):367–370
Liu J, Xu Q, Yuan Q, Wang Z, Xing C, Yuan Y (2015) Association of IL-17A and IL-17F polymorphisms with gastric cancer risk in Asians: a meta-analysis. Hum Immunol 76(1):6–12
Domingos JA, Soares LS, Bandeira LM, Bonin CM, Vicente AC, Zanella L et al (2017) Cytokine profile and proviral load among Japanese immigrants and non-Japanese infected with HTLV-1 in a non-endemic area of Brazil. PLoS One 12(4):e0174869
Santos SB, Oliveira P, Luna T, Souza A, Nascimento M, Siqueira I et al (2012) Immunological and viral features in patients with overactive bladder associated with human T-cell lymphotropic virus type 1 infection. J Med Virol 84(11):1809–1817
Garlet GP, Giozza SP, Silveira EM, Claudino M, Santos SB, Avila-Campos MJ et al (2010) Association of human T lymphotropic virus 1 amplification of periodontitis severity with altered cytokine expression in response to a standard periodontopathogen infection. Clin Infect Dis 50(3):e11–e18
Kagi D, Ledermann B, Burki K, Zinkernagel RM, Hengartner H (1996) Molecular mechanisms of lymphocyte-mediated cytotoxicity and their role in immunological protection and pathogenesis in vivo. Annu Rev Immunol 14:207–232
Goon PK, Hanon E, Igakura T, Tanaka Y, Weber JN, Taylor GP et al (2002) High frequencies of Th1-type CD4(+) T cells specific to HTLV-1 Env and Tax proteins in patients with HTLV-1-associated myelopathy/tropical spastic paraparesis. Blood 99(9):3335–3341
Iwasaki Y (1993) Human T cell leukemia virus type I infection and chronic myelopathy. Brain Pathol 3(1):1–10
Hanon E, Goon P, Taylor GP, Hasegawa H, Tanaka Y, Weber JN et al (2001) High production of interferon gamma but not interleukin-2 by human T-lymphotropic virus type I-infected peripheral blood mononuclear cells. Blood 98(3):721–726
Journo C, Mahieux R (2011) HTLV-1 and innate immunity. Viruses 3(8):1374–1394
Schoenborn JR, Wilson CB (2007) Regulation of interferon-gamma during innate and adaptive immune responses. Adv Immunol 96:41–101
Hieshima K, Nagakubo D, Nakayama T, Shirakawa AK, Jin Z, Yoshie O (2008) Tax-inducible production of CC chemokine ligand 22 by human T cell leukemia virus type 1 (HTLV-1)-infected T cells promotes preferential transmission of HTLV-1 to CCR4-expressing CD4+ T cells. J Immunol 180(2):931–939
Yoshie O (2021) CCR4 as a therapeutic target for cancer immunotherapy. Cancers (Basel) 13(21)
Leung K, Nabel GJ (1988) HTLV-1 transactivator induces interleukin-2 receptor expression through an NF-kappa B-like factor. Nature 333(6175):776–778
Zhao H, Bo C, Kang Y, Li H (2017) What else can CD39 tell us? Front Immunol 8:727
Alvarez-Sanchez N, Cruz-Chamorro I, Diaz-Sanchez M, Lardone PJ, Guerrero JM, Carrillo-Vico A (2019) Peripheral CD39-expressing T regulatory cells are increased and associated with relapsing-remitting multiple sclerosis in relapsing patients. Sci Rep 9(1):2302
Yamano Y, Takenouchi N, Li HC, Tomaru U, Yao K, Grant CW et al (2005) Virus-induced dysfunction of CD4+CD25+ T cells in patients with HTLV-I-associated neuroimmunological disease. J Clin Invest 115(5):1361–1368
Toulza F, Nosaka K, Tanaka Y, Schioppa T, Balkwill F, Taylor GP et al (2010) Human T-lymphotropic virus type 1-induced CC chemokine ligand 22 maintains a high frequency of functional FoxP3+ regulatory T cells. J Immunol 185(1):183–189
Shimizu Y, Takamori A, Utsunomiya A, Kurimura M, Yamano Y, Hishizawa M et al (2009) Impaired Tax-specific T-cell responses with insufficient control of HTLV-1 in a subgroup of individuals at asymptomatic and smoldering stages. Cancer Sci 100(3):481–489
Thibaudin M, Chaix M, Boidot R, Vegran F, Derangere V, Limagne E et al (2016) Human ectonucleotidase-expressing CD25(high) Th17 cells accumulate in breast cancer tumors and exert immunosuppressive functions. Oncoimmunology 5(1):e1055444
Lang C, Wang J, Chen L (2017) CD25-expressing Th17 cells mediate CD8(+) T cell suppression in CTLA-4 dependent mechanisms in pancreatic ductal adenocarcinoma. Exp Cell Res 360(2):384–389
Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B et al (2003) Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421(6924):744–748
Brucklacher-Waldert V, Stuerner K, Kolster M, Wolthausen J, Tolosa E (2009) Phenotypical and functional characterization of T helper 17 cells in multiple sclerosis. Brain 132(Pt 12):3329–3341
Moser T, Akgun K, Proschmann U, Sellner J, Ziemssen T (2020) The role of TH17 cells in multiple sclerosis: therapeutic implications. Autoimmun Rev 19(10):102647
Melnikov M, Rogovskii V, Boyksmall o CA, Pashenkov M (2020) Dopaminergic therapeutics in multiple sclerosis: focus on Th17-cell functions. J Neuroimmune Pharmacol 15(1):37–47
Kebir H, Ifergan I, Alvarez JI, Bernard M, Poirier J, Arbour N et al (2009) Preferential recruitment of interferon-gamma-expressing TH17 cells in multiple sclerosis. Ann Neurol 66(3):390–402
van Langelaar J, van der Vuurst de Vries RM, Janssen M, Wierenga-Wolf AF, Spilt IM, Siepman TA et al (2018) T helper 17.1 cells associate with multiple sclerosis disease activity: perspectives for early intervention. Brain 141(5):1334–1349
Ntolkeras G, Barba C, Mavropoulos A, Vasileiadis GK, Dardiotis E, Sakkas LI et al (2019) On the immunoregulatory role of statins in multiple sclerosis: the effects on Th17 cells. Immunol Res 67(4-5):310–324
Sie C, Korn T, Mitsdoerffer M (2014) Th17 cells in central nervous system autoimmunity. Exp Neurol 262(Pt A):18–27
Duhen R, Glatigny S, Arbelaez CA, Blair TC, Oukka M, Bettelli E (2013) Cutting edge: the pathogenicity of IFN-gamma-producing Th17 cells is independent of T-bet. J Immunol 190(9):4478–4482
Hirota K, Duarte JH, Veldhoen M, Hornsby E, Li Y, Cua DJ et al (2011) Fate mapping of IL-17-producing T cells in inflammatory responses. Nat Immunol 12(3):255–263
Belpaire A, van Geel N, Speeckaert R (2022) From IL-17 to IFN-gamma in inflammatory skin disorders: is transdifferentiation a potential treatment target? Front Immunol 13:932265
Tsukasaki K, Tobinai K (2014) Human T-cell lymphotropic virus type I-associated adult T-cell leukemia-lymphoma: new directions in clinical research. Clin Cancer Res 20(20):5217–5225
Yamauchi J, Coler-Reilly A, Sato T, Araya N, Yagishita N, Ando H et al (2015) Mogamulizumab, an anti-CCR4 antibody, targets human T-lymphotropic virus type 1-infected CD8+ and CD4+ T cells to treat associated myelopathy. J Infect Dis 211(2):238–248
Toulza F, Heaps A, Tanaka Y, Taylor GP, Bangham CR (2008) High frequency of CD4+FoxP3+ cells in HTLV-1 infection: inverse correlation with HTLV-1-specific CTL response. Blood 111(10):5047–5053
Lee GR (2018) The balance of Th17 versus Treg cells in autoimmunity. Int J Mol Sci 19(3)
Seif F, Khoshmirsafa M, Aazami H, Mohsenzadegan M, Sedighi G, Bahar M (2017) The role of JAK-STAT signaling pathway and its regulators in the fate of T helper cells. Cell Commun Signal 15(1):23
Oh U, Grant C, Griffith C, Fugo K, Takenouchi N, Jacobson S (2006) Reduced Foxp3 protein expression is associated with inflammatory disease during human t lymphotropic virus type 1 Infection. J Infect Dis 193(11):1557–1566
Danikowski KM, Jayaraman S, Prabhakar BS (2017) Regulatory T cells in multiple sclerosis and myasthenia gravis. J Neuroinflammation 14(1):117
Lazibat I, Rubinic Majdak M, Zupanic S (2018) Multiple sclerosis: new aspects of immunopathogenesis. Acta Clin Croat 57(2):352–361
Garg N, Smith TW (2015) An update on immunopathogenesis, diagnosis, and treatment of multiple sclerosis. Brain Behav 5(9):e00362
Holmoy T, Hestvik AL (2008) Multiple sclerosis: immunopathogenesis and controversies in defining the cause. Curr Opin Infect Dis 21(3):271–278
Orland JR, Engstrom J, Fridey J, Sacher RA, Smith JW, Nass C et al (2003) Prevalence and clinical features of HTLV neurologic disease in the HTLV Outcomes Study. Neurology 61(11):1588–1594
Aye MM, Matsuoka E, Moritoyo T, Umehara F, Suehara M, Hokezu Y et al (2000) Histopathological analysis of four autopsy cases of HTLV-I-associated myelopathy/tropical spastic paraparesis: inflammatory changes occur simultaneously in the entire central nervous system. Acta Neuropathol 100(3):245–252
Iwasaki Y (1990) Pathology of chronic myelopathy associated with HTLV-I infection (HAM/TSP). J Neurol Sci 96(1):103–123
Yoshioka A, Hirose G, Ueda Y, Nishimura Y, Sakai K (1993) Neuropathological studies of the spinal cord in early stage HTLV-I-associated myelopathy (HAM). J Neurol Neurosurg Psychiatry 56(9):1004–1007
Cooper SA, van der Loeff MS, Taylor GP (2009) The neurology of HTLV-1 infection. Pract Neurol 9(1):16–26
Comabella M, Khoury SJ (2012) Immunopathogenesis of multiple sclerosis. Clin Immunol 142(1):2–8
Ijichi S, Izumo S, Eiraku N, Machigashira K, Kubota R, Nagai M et al (1993) An autoaggressive process against bystander tissues in HTLV-I-infected individuals: a possible pathomechanism of HAM/TSP. Med Hypotheses 41(6):542–547
Umehara F, Izumo S, Nakagawa M, Ronquillo AT, Takahashi K, Matsumuro K et al (1993) Immunocytochemical analysis of the cellular infiltrate in the spinal cord lesions in HTLV-I-associated myelopathy. J Neuropathol Exp Neurol 52(4):424–430
Grindstaff P, Gruener G (2005) The peripheral nervous system complications of HTLV-1 myelopathy (HAM/TSP) syndromes. Semin Neurol 25(3):315–327
Gillet NA, Malani N, Melamed A, Gormley N, Carter R, Bentley D et al (2011) The host genomic environment of the provirus determines the abundance of HTLV-1-infected T-cell clones. Blood 117(11):3113–3122
Abe M, Umehara F, Kubota R, Moritoyo T, Izumo S, Osame M (1999) Activation of macrophages/microglia with the calcium-binding proteins MRP14 and MRP8 is related to the lesional activities in the spinal cord of HTLV-I associated myelopathy. J Neurol 246(5):358–364
Jacobson S (2002) Immunopathogenesis of human T cell lymphotropic virus type I-associated neurologic disease. J Infect Dis 186(Suppl 2):S187–S192
Aghajanian S, Teymoori-Rad M, Molaverdi G, Mozhgani SH (2020) Immunopathogenesis and cellular interactions in human T-cell leukemia virus type 1 associated myelopathy/tropical spastic paraparesis. Front Microbiol 11:614940
Korn T, Kallies A (2017) T cell responses in the central nervous system. Nat Rev Immunol 17(3):179–194
Balasa R, Barcutean L, Balasa A, Motataianu A, Roman-Filip C, Manu D (2020) The action of TH17 cells on blood brain barrier in multiple sclerosis and experimental autoimmune encephalomyelitis. Hum Immunol 81(5):237–243
Afonso PV, Ozden S, Cumont MC, Seilhean D, Cartier L, Rezaie P et al (2008) Alteration of blood-brain barrier integrity by retroviral infection. PLoS Pathog 4(11):e1000205
Tsukada N, Tanaka Y, Yanagisawa N (1989) Autoantibodies to brain endothelial cells in the sera of patients with human T-lymphotropic virus type I associated myelopathy and other demyelinating disorders. J Neurol Sci 90(1):33–42
Ikegami M, Umehara F, Ikegami N, Maekawa R, Osame M (2002) Selective matrix metalloproteinase inhibitor, N-biphenyl sulfonyl phenylalanine hydroxamic acid, inhibits the migration of CD4+ T lymphocytes in patients with HTLV-I-associated myelopathy. J Neuroimmunol 127(1-2):134–138
Afonso PV, Ozden S, Prevost MC, Schmitt C, Seilhean D, Weksler B et al (2007) Human blood-brain barrier disruption by retroviral-infected lymphocytes: role of myosin light chain kinase in endothelial tight-junction disorganization. J Immunol 179(4):2576–2583
Curis C, Percher F, Jeannin P, Montange T, Chevalier SA, Seilhean D et al (2016) Human T-lymphotropic virus type 1-induced overexpression of activated leukocyte cell adhesion molecule (ALCAM) facilitates trafficking of infected lymphocytes through the blood-brain barrier. J Virol 90(16):7303–7312
Huppert J, Closhen D, Croxford A, White R, Kulig P, Pietrowski E et al (2010) Cellular mechanisms of IL-17-induced blood-brain barrier disruption. FASEB J 24(4):1023–1034
Wojkowska DW, Szpakowski P, Ksiazek-Winiarek D, Leszczynski M, Glabinski A (2014) Interactions between neutrophils, Th17 cells, and chemokines during the initiation of experimental model of multiple sclerosis. Mediators Inflamm 2014:590409
O’Connor RA, Prendergast CT, Sabatos CA, Lau CW, Leech MD, Wraith DC et al (2008) Cutting edge: Th1 cells facilitate the entry of Th17 cells to the central nervous system during experimental autoimmune encephalomyelitis. J Immunol 181(6):3750–3754
Imaizumi Y, Sugita S, Yamamoto K, Imanishi D, Kohno T, Tomonaga M et al (2002) Human T cell leukemia virus type-I Tax activates human macrophage inflammatory protein-3 alpha/CCL20 gene transcription via the NF-kappa B pathway. Int Immunol 14(2):147–155
Baba M, Imai T, Nishimura M, Kakizaki M, Takagi S, Hieshima K et al (1997) Identification of CCR6, the specific receptor for a novel lymphocyte-directed CC chemokine LARC. J Biol Chem 272(23):14893–14898
Reboldi A, Coisne C, Baumjohann D, Benvenuto F, Bottinelli D, Lira S et al (2009) C-C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat Immunol 10(5):514–523
Author information
Authors and Affiliations
Contributions
MS performed data collection, prepared the figures (by Adobe Illustrator and Microsoft PowerPoint), and was the major contributor to writing the manuscript. S-HM conceptualized the review article, critically revised the manuscript, had a minor contribution to writing, and provided oversight and verification of the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
N/A.
Consent to Participate
N/A.
Consent for Publication
All the authors give their consent for publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shafiei, M., Mozhgani, SH. Th17/IL-17 Axis in HTLV-1-Associated Myelopathy Tropical Spastic Paraparesis and Multiple Sclerosis: Novel Insights into the Immunity During HAMTSP. Mol Neurobiol 60, 3839–3854 (2023). https://doi.org/10.1007/s12035-023-03303-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03303-0