Abstract
Epigenetic processes have become increasingly relevant in understanding disease-modifying mechanisms. 5-Methylcytosine methylations of DNA (5mC) and RNA (m5C) have functional transcriptional and RNA translational consequences and are tightly regulated by writer, reader and eraser effector proteins. To investigate the involvement of 5mC/5hmC and m5C effector proteins contributing to the development of dementia neuropathology, RNA sequencing data of 31 effector proteins across four brain regions was examined in 56 aged non-affected and 51 Alzheimer’s disease (AD) individuals obtained from the Aging, Dementia and Traumatic Brain Injury Study. Gene expression profiles were compared between AD and controls, between neuropathological Braak and CERAD scores and in individuals with a history of traumatic brain injury (TBI). We found an increase in the DNA methylation writers DNMT1, DNMT3A and DNMT3B messenger RNA (mRNA) and a decrease in the reader UHRF1 mRNA in AD samples across three brain regions whilst the DNA erasers GADD45B and AICDA showed changes in mRNA abundance within neuropathological load groupings. RNA methylation writers NSUN6 and NSUN7 showed significant expression differences with AD and, along with the reader ALYREF, differences in expression for neuropathologic ranking. A history of TBI was associated with a significant increase in the DNA readers ZBTB4 and MeCP2 (p < 0.05) and a decrease in NSUN6 (p < 0.001) mRNA. These findings implicate regulation of protein pathways disrupted in AD and TBI via multiple pre- and post-transcriptional mechanisms including potentially acting upon transfer RNAs, enhancer RNAs as well as nuclear-cytoplasmic shuttling and cytoplasmic translational control. The targeting of such processes provides new therapeutic avenues for neurodegenerative brain conditions.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dementia, a progressive decline in neurocognitive ability, is the principal cause of disability in the elderly population [1]. In 2019, the number of people diagnosed with dementia worldwide reached 50 million, incurring a significant socio-economic burden on society [2, 3]. Forms of dementia are caused by progressive and irreversible degeneration of neurons and include Alzheimer’s disease (AD), dementia of Lewy bodies and frontotemporal dementia. Diagnosis of AD by brain tissue histology is made through the identification of intraneuronal aggregation of tau protein known as neurofibrillary tangles (NFTs) and the accumulation of insoluble beta-amyloid peptide (Aβ) termed senile and neuritic plaques [4]. These pathological features progress through stages of severity which correlate to burden of pathology and anatomical localization [5, 6].
Most neurodegenerative dementias have a multifactorial aetiology and are thought to develop due to multiple environmental and genomic factors. One proposed life event risk factor is a history of traumatic brain injury (TBI) which results from head trauma that leads to either focal or diffuse injury [7, 8]. The effect of TBI can be acute or chronic with the latter having a greater link to cognitive decline [9,10,11]. Neurodegenerative lesions are commonly found in individuals exposed to TBI and are referred to as TBI-related neurodegeneration (TReND) [10, 12, 13]. However, although changes in clinical phenotype, brain activity and histopathological features have been reported in TBI and TReND, less is known about the molecular mechanisms which contribute to long-term neuropathological changes.
Epigenetic processes which regulate transcriptional and translational activity are proposed as mechanisms which mediate environmental influences on brain physiology [14, 15]. Such epigenetic modifications are tissue specific and change during cellular maturation, i.e. during differentiation and ageing, and therefore have been implicated in disease processes including dementia pathology [16,17,18]. A commonly studied epigenetic process, 5-methylcytosine (5mC) DNA methylation, involves the addition of a methyl group to the fifth carbon of the cytosine base at CpG (mCG) and CpA (mCA) sites and is associated with transcription repression [19]. However, 5mC can be converted into alternative oxidised methylation states known as 5-hydroxymethylcytosine (5hmC), 5-formylcytosine (5fC) and 5-carboxylcytosine (5caC) through ten-eleven translocation (TET) methylcytosine dioxygenase and thymine DNA glycosylase (TDG)–mediated active demethylation [20, 21]. Such oxidised states have been shown to be stable and to potentially have functional transcriptional consequences [22,23,24].
Methylation of RNA species at the same cytosine base, known as 5-methylcytosine (m5C), also undergoes oxidisation to form 5-hydroxymethylcytosine (hm5C), 5-formylcytidine (f5C) and 5-carboxycytidine (ca5C) [25,26,27,28]. m5C methylation of messenger RNA (mRNA) is indicated to increase RNA stability and abundance and regulate nuclear exportation, negatively correlating with translation efficiency [29, 30]. In transfer RNA (tRNA), ribosomal RNA (rRNA) and mt-tRNA, m5C modification provides structural stability and improves the accuracy of translation [28, 29]. Similarly, hm5C modification enhances mRNA translation whereas f5C of mitochondrial tRNA is important for translation of AUA to methionine [31,32,33]. As such, the modification of DNA and RNA may have important consequences on cellular localisation of RNAs and regional patterns of gene expression.
DNA and RNA methylation are governed by three families of effector proteins known as writers, readers and erasers. The writer family of proteins, methyltransferases, add the methyl groups to nucleotides whereas reader proteins bind to methylated DNA/RNA and assist in the formation of protein complexes which influence transcriptional or translational processes. The eraser group of proteins are enzymes known as demethylases which generate the oxidised states and takes off methyl groups. The expression of these effector proteins across brain regions and within cellular environments will have important consequences for transcription and translation activity.
To investigate the involvement of 5mC and m5C methylation processes contributing to the development of AD neuropathology, we analysed RNA sequencing data from the Aging, Dementia and Traumatic Brain Injury Study to characterise RNA expression profiles of 31 DNA and RNA methylation effector protein genes across four brain regions in 56 unaffected individuals and 51 individuals with AD. In a second phase, we examined gene expression within these DNA/RNA methylation genes by grouping the samples by Braak stage and CERAD specific neuropathological staging. To better understand the relationship between TBI and dementia and changes in effector protein abundance, we also explored gene expression profiles in groups of individuals with the following: no TBI and no AD, all TBI (with and without AD), TBI and no AD, TBI and AD and, AD without TBI.
Methods
Study samples
Data were obtained from the Aging, Dementia and Traumatic Brain Injury Study from the Allen Institute for Brain Science (http://aging.brain-map.org/) [34, 35]. The cohort of case and age–matched unaffected control individuals was provided by the Adult Changes in Thought (ACT) Study [36]. Information was gathered from 107 individuals consisting of 51 individuals with clinical diagnosis of AD and 56 individuals without AD (referred to as control individuals). Twenty-six of the control individuals had a history of TBI, and 30 had no history of TBI, whereas 27 individuals with AD had a history of TBI and 24 had no history of TBI (Table 1). Brain tissue was procured by macrodissection from fresh frozen brain with a mean post-mortem interval (PMI) of 4.6 ± 1.5 h for AD individuals and 4.7 ± 2 h for control individuals [34]. RNA expression values were obtained for the hippocampus (HIP), the superior temporal gyrus (STG) of the temporal cortex, the inferior parietal cortex (IPC) and the white matter (WM) from the parietal cortex (Supplementary Table 1).
Diagnosis criteria for AD established by the National Institute of Ageing and the CERAD [6] is based on the ‘ABC’ score which consists of the Aβ senile plaque rating, a Braak score and a CERAD staging. The Braak score measures the presence and distribution of NFT through specific brain regions and is classified in stages 0 to VI accordingly. The CERAD rating is a semiquantitative assessment of the presence of Aβ as neuritic plaques (Supplementary Table 2).
Analysis of RNA expression profiles
mRNA expression profiles were examined for the following genes: DNA methylation writers, DNMT1 (NM_001130823), DNMT3A (NM_022552.5) and DNMT3B (NM_006892.4); readers, MeCP2 (NM_004992.4), ZBTB4 (NM_001128833.2), ZBTB33 (NM_001184742.2), ZBTB38 (NM_001376113.1) and UHRF1 (NM_001048201.3); and erasers, TET1 (NM_030625.3), TET2 (NM_001127208.3), TET3 (NM_001287491.2), TDG (NM_003211.6), MBD4 (NM_001276270.2), AICDA (NM_020661.4), GADD45A (NM_001924.4), GADD45B (NM_015675.4) and GADD45G (NM_006705.4). RNA methylation effector genes examined were as follows: writers, NOP2/NSUN1 (NM_001258308.2), NSUN2 (NM_017755.6), NSUN3 (NM_022072.5), NSUN4 (NM_199044.4), NSUN5 (NM_148956.4), NSUN6 (NM_182543.5), NSUN7 (NM_024677.6) and TRDMT1/DNMT2 (NM_004412.7); readers, ALYREF (NM_005782.4) and YBX1 (NM_004559.5); and erasers, ALKBH1 (NM_006020.3), TET1 (NM_030625.3), TET2 (NM_001127208.3) and TET3 (NM_001287491.2). Expression data were obtained from normalised fragments per kilobase of transcript per million (FPKM) values derived from RNA sequencing and presented as a z-score value of expression.
Individuals were categorised by clinical diagnosis AD versus control and grouped by age of death, APOE4 allele carrier status and Braak and CERAD staging in HIP and STG (Supplementary Table 3) and IPC or WM (Supplementary Table 4). For Braak staging, three groups were generated to represent levels of pathology. Group 1 corresponded to individuals representing low levels of pathology with a Braak stage between 0 and II. Group 2 had a Braak stage between III and IV and had moderate pathology. In the third group, samples had Braak stages between V and VI, indicating the highest level of pathology. To analyse by CERAD ranking, samples were divided into two groups with a CERAD score of 0–1 indicating low amyloid load and a CERAD score of 2–3 representing high amyloid load.
Supplementary Fig. 1 provides a flow chart of our analysis pipeline. In a first phase, differences between age, sex, APOE4 allele status and Braak and CERAD staging in the groups AD and control were assessed. We then tested for significant correlations between diagnosis status, Braak, CERAD and APOE4 across each of the four brain regions. In a second phase, we compared RNA abundance profiles across the 32 effector proteins in AD and control groups for each brain region. Subsequently, differences in gene expression were examined in individuals classified by Braak and CERAD rankings. In the third arm of the study, we assessed differential expression in individuals who self-reported TBI. For this analysis, the cohort was divided into five groups: a TBI-control group of aged individuals without TBI and without AD (referred to as ‘TBI-control’); a group of individuals which includes all TBI individuals, i.e. with and without dementia (All TBI); a third group of individuals with TBI but no AD (TBI + no AD); a fourth group which includes individuals with TBI and AD (TBI + AD); and a final group with no TBI but have AD (no TBI + AD). No differences were observed between gender, age and brain tissue post-mortem interval across the TBI groupings (Supplementary Tables 5 and 6). Similarly, in individuals who reported head injuries, we found no difference between age of first TBI incident, number of TBI incidents with loss of consciousness or duration or across the TBI groupings (Supplementary Table 7).
Statistical analysis
Normality of gene expression z-scores was tested by applying the Shapiro–Wilk test as well as visual analysis of Q-Q plots. Descriptive statistics was performed presenting the mean with standard deviation (SD) or median with 95% confidence interval (CI). In the first stage of analysis, differences in age between diagnosis status were examined using the Mann–Whitney test. Chi-square tests were performed to assess the relationships between diagnosis status and sex, APOE4 and Braak and CERAD scores. Differences in gene expression values of DNA and RNA effector proteins between AD and controls, and between the CERAD groupings, were evaluated using t tests and Mann–Whitney tests. To identify significant differences in the expression values between Braak groups, a one-way ANOVA or Kruskal–Wallis test was performed with the Tukey post hoc test or Dunn’s test analysis, respectively. In the final stage of analysis, corrections for multiple comparisons across TBI groupings were performed using Dunnett’s test for multiple comparisons. All tests were two tailed, and a level of significance of 0.05 was accepted. Data analysis and graphs were generated using IBM SPSS Statistics for Windows (version 27.0) and GraphPad Prism (version 9.1.1) for Windows.
Results
Demographics and neuropathologic load in control and Alzheimer’s disease groups
We first examined differences in group demographics and pathology scores between control individuals and individuals with AD (Table 1). No significant differences were observed for age, sex, APOE4 allele status or TBI history between the groups. We found a significant difference between AD and control individuals in Braak staging (χ2 = 14.7, p = 0.0006) and CERAD scores (χ2 = 7.7, p = 0.005). As expected, AD samples had a higher prevalence of Braak V–VI and of CERAD 2–3 staging, whilst most control individuals presented with a Braak stage of 0–IV and a CERAD score of 0–1. This pattern was also consistently observed across the four brain regions with the highest AD case and control group difference for Braak (χ2 = 17.2, p = 0.0002) and CERAD (χ2 = 12.4, p = 0.0004) ranks in the hippocampus. As expected, a significant positive correlation between Braak and CERAD scores was apparent across all brain regions (r = 0.6, p < 0.0001) (Supplementary Fig. 2).
Effector protein expression profiles in control and Alzheimer’s disease individuals
To determine patterns of expression across the brain regions in the normal brain, we first examined the expression profiles of effector proteins in control individuals (Fig. 1). In healthy aged individuals, DNA effector proteins showed varied expression across the four brain regions and generally showed higher expression in the WM and lowest expression in the HIP (Fig. 1A, Supplementary Fig. 3A). The STG and parietal cortex (IPC) were found to have similar gene expression patterns and indicated that the writer DNMT3B, readers MeCP2 and ZBTB4 and erasers GADD45B and GADD45G had the highest abundance in these regions. In the WM of the parietal lobe, the writers DNMT1 and DNMT3A, the reader UHRF1 and the erasers TET1 and TET2, which are involved in both DNA and RNA modification, were found to be the most highly expressed transcripts. These observations suggest that individual effector proteins may have varied roles in DNA methylation processes depending on the brain region and that function may be dependent on the cell and tissue context.
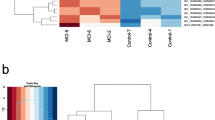
Expression profiles of DNA and RNA effector proteins within the hippocampus, superior temporal gyrus, parietal cortex and white matter tissue. A Relative expression of DNA effector proteins in non-affected individuals and individuals with AD. B, C Significantly increased expression of DNA effector proteins, DNMT1, DNMT3A and DNMT3B, and decreased expression of UHRF1 were evident in individuals with AD as compared to unaffected individuals in the superior temporal gyrus and white matter regions. D Relative expression of RNA effector proteins in non-affected individuals and individuals with AD. E, F Significantly decreased expression of the RNA writer effector protein NSUN6 in AD individuals in the white matter and superior gyrus and significantly increased abundance of NSUN7 in AD individuals in the hippocampus were observed. In A, B, D and E, low expression values, < 0, are presented as green and high expression value, > 0, is shown as red. *p < 0.05
In the aged individuals with Alzheimer’s disease, DNA effector proteins commonly showed similar tissue-specific patterns in expression to the non-affected samples but showed overall higher abundance of effector transcript expression in the STG, IPC and WM as compared to control individuals (Fig. 1A and Supplementary Fig. 3A). The eraser proteins indicated the most varied expression profiles across the brain regions and typically showed increased expression in AD cases across all regions. Quantitative comparisons of gene expression from DNA effectors between AD and control individuals (Con) revealed significant differences in expression of the writers DNMT1 (p = 0.01) and DNMT3A (p < 0.05) in the STG and DNMT3B (p < 0.05) in the WM (Fig. 1C, Supplementary Table 8). In all significant differences, higher expression was observed in AD cases compared to controls. Conversely, the DNA reader UHRF1 showed significantly higher abundance in control individuals compared to AD cases (p < 0.05) in the WM (Fig. 1B and 1C). No significant differences in 5mC effector protein abundance were observed in the IPC or HIP.
Like the DNA effector proteins, RNA effector proteins in the healthy brain exhibited overall highest relative expression in the WM tissue (Fig. 1D, Supplementary Fig. 3B). However, in contrast to DNA effector proteins, gene expression of effector proteins in the hippocampus, although varied, was moderately high particularly for the writer proteins with the exception of NSUN4. YBX1 and TET1 and TET2 were the most abundant reader and eraser proteins, respectively, in the HIP and WM, whereas in the STG and IPC, the reader ALYREF and the erasers ALKBH1 and TET3 were more highly expressed again providing evidence for tissue-specific RNA methylation effector protein mechanisms.
RNA effector proteins in the AD group again showed a similar overall expression profile to healthy controls across the brain regions (Fig. 1D). However, in the AD group, the writer NSUN6 was found to have significantly lower abundance in two regions: in the STG (p = 0.02) and in the WM (p = 0.03), whereas NSUN7 showed higher abundance in the HIP (p = 0.02) (Fig. 1E and 1F, Supplementary Table 8). No differences in the expression of the writers between AD and control individuals were found in the IPC or for any reader or eraser protein transcripts in any of the four brain regions.
Differences in DNA/RNA effector protein expression grouped by Braak and CERAD neuropathological scales
Table 2 presents the relative expression values and associated p values for differences in 5mC DNA and m5C RNA methylation effector transcript expression grouped by Braak stages. We observed significantly higher abundance in the two DNA eraser protein transcripts GADD45B and AICDA which were associated with differences in Braak staging (Fig. 2A). Here, GADD45B gene expression was significantly lower in the mid-neuropathology scores compared to low-early and high-late neuropathological Braak staging in the hippocampus (H = 6.9, p = 0.03; post hoc between stages 0–II and III–IV, p = 0.01) and the superior temporal gyrus (H = 7.0, p = 0.03; post hoc between stages 0–II and III–IV, p = 0.008). Similarly, expression of the eraser AICDA in the WM was significantly higher in low-early Braak stages compared to both mid and late stages (H = 10.1, p < 0.01; post hoc between stages 0–II and V–VI, p = 0.002; post hoc between stages III–IV and V–VI, p = 0.03). Conversely, and consistent with differences between healthy aged and AD tissues, the reader effector transcript UHRF1 showed significant lower expression in low and mid Braak staging groups compared to Braak V–VI late stages in the STG (H = 7.9, p = 0.02; post hoc between stages 0–II and V–VI, p = 0.02; post hoc between stages III–IV and V–VI, p = 0.01).
DNA and RNA methylation effector proteins show significant differences in abundance across Braak stages and CREAD rankings. A The reader UHRF1 and eraser GADD45B showed lower expression in the mid Braak stages in the superior temporal gyrus. Significantly lower expression of mid-Braak stages was also evident for GADD45B in the hippocampus whereas AICDA was significantly reduced in tissue with the highest Braak pathology in the white matter. B The writers NSUN6 and NSUN7 and the reader ALYREF showed significant differences in relative expression across Braak stages in the hippocampus and inferior parietal lobe. C Consistent with these Braak staging, NSUN7 was significantly increased in individuals with the highest neuropathological CERAD score in the hippocampus. *p < 0.05; **p < 0.01
Differences in levels of expression of the RNA methylation effector transcripts NSUN6, NSUN7 and ALYREF were also observed across the Braak staging groups (Table 2, Fig. 2B). In the hippocampus, NSUN6 showed lower expression in the early and late neuropathological Braak stages I–II and V–VI (F = 4.7, p = 0.01; post hoc between stages 0–II and III–IV, p = 0.03; post hoc between stages III–IV and V–VI, p = 0.005). In contrast, NSUN7 showed higher expression in mid and later Braak stages (F = 3.8, p = 0.03; post hoc between stages 0–II and III–IV, p = 0.01; post hoc between stages 0–II and V–VI, p = 0.02). This finding is consistent with a higher relative expression of NSUN7 in CERAD stages 2–3 compared to CERAD stages 0–1 (Fig.2C) (p = 0.03) (CERAD 0–1, x̃ = 0.50; CERAD 2–3, x̃ = 0.88). In contrast, ALYREF showed significantly low abundance in late Braak stages V–VI in both the hippocampus (H = 6.2, p = 0.04; post hoc between stages 0–II and V–VI, p = 0.01) and the parietal cortex (H = 8.4, p = 0.02; post hoc between stages 0–II and V–VI, p = 0.006; post hoc between stages III–IV and V–VI, p = 0.03) (Fig. 2B, Table 2).
In the final stage of the study, expression of 5mC DNA and m5C RNA methylation effector proteins was investigated within the five TBI groupings: TBI-control (no TBI + no AD), all TBI, TBI + no AD, TBI + AD and no TBI + AD (Supplementary Fig. 1). Two DNA readers, ZBTB4 and MeCP2, showed significant changes in RNA abundance, and both were observed to be less abundant in the ‘TBI-control’ group compared to the ‘all TBI’ group (p < 0.05) (Fig. 3A and 3B). MeCP2 was also observed to be less abundant in the TBI-control group when compared to the ‘no TBI + AD’ group (p < 0.05) (Fig. 3B, Supplementary Table 9). Finally, similar to the AD and Braak scoring analysis, the RNA methylation writer NSUN6 exhibited a significant difference across these TBI groupings. In the STG, we observed a significantly lower expression of NSUN6 in the group ‘TBI + no AD’ (p ≤ 0.05) and in groups ‘all TBI’ (p ≤ 0.0001) and ‘TBI + AD’ (p ≤ 0.001), when compared to the TBI-control group (Fig. 3C, Supplementary Table 9). These findings suggest that decreased expression of NSUN6 in TBI is not driven by a dementia phenotype.
Cytosine methylation effector proteins showing significant differential expression between traumatic brain injury (TBI) groupings. All significant differences in expression between TBO groupings were found in the superior temporal gyrus. A The DNA effector reader protein ZBTB4 showed significant decreased expression in TBI-controls compared with the all-TBI group. B The DNA reader MECP2 showed significantly higher expression in the all-TBI group and no TBI + AD group compared with the control-TBI individuals. C The RNA effector writer protein NSUN6 showed significantly higher expression in the TBI-control group as compared to all individuals with TBI, individuals with TBI and no AD, and TBI and AD. *p ≤ 0.05; ***p ≤ 0.001; ****p ≤ 0.0001
Discussion
Epigenetic processes have become increasingly relevant in understanding disease-modifying mechanisms [37,38,39]. In the current study, we observed changes in the expression of the DNA writer proteins DNMT1, DNMT3A and DNMT3B and of the reader UHRF1 in AD individuals whilst the DNA methyltransferase erasers GADD45B and AICDA showed changes in mRNA abundance within neuropathological load groupings. RNA methylation writers NSUN6 and NSUN7 presented contrasting profiles, with NSUN6 significantly decreased in AD individuals and individuals with TBI in the hippocampus and temporal gyrus, whereas NSUN7 was increased within the hippocampus in individuals with AD or high neuropathology. ALYREF, a RNA reader, showed decreased abundance with higher pathological stages, and a history of TBI was associated with significant increases in DNA readers ZBTB4 and MeCP2, but a decrease in NSUN6 abundance.
DNMT writer proteins interact with chromatin and histone modification enzymes to stabilise the methylation machinery complex and direct the addition of methyl groups onto DNA [40]. DNA reader proteins bind to methylated DNA CpH sites and repress or, in the case of MeCP2, potentially activate transcription [41]. The functional consequences of mutations within DNMT1, DNMT3B and DNMT3A, which cause monogenic human diseases and rare cancers, e.g. DNMT1 complex disorders (HSAN1E) [42, 43], DNMT3B (ICF syndrome) [44, 45] and DNMT3A (Tatton Brown Rahman syndrome) [46, 47], are proposed to be dependent on mutation type and the extent they cause a reduction in gene function or expression, i.e. the degree of hypomorphism. As a result, DNMT complex activity can be either weakly, moderately or strongly disrupted and can cause localised changes or more global widespread changes in DNA methylation [48,49,50,51]. Therefore, our findings of differential expression of DNMT1, DNMT3, DNMT3B and UHRF1 in AD and ZBTB4 and MeCP2 in TBI could lead to alterations in complex binding which is consistent with causing changes to DNA methylation sites. Indeed, evidence already exists that disruption of MeCP2 in neuronal tissue in mice increases expression of long neuronal genes through binding to mCA and mCG within, and neighbouring, long neuronal genes [52, 53]. However, whether changes to the writer and reader complexes commonly affect widespread or specific patterns of unoxidised and oxidised mCG and mCA cytosine bases which influence increased transcriptional activity remain to be investigated.
RNA methylation effector proteins are known to target various types of RNA species. For example, m5C occurs on mRNA, tRNA, rRNA and mt-RNA and alternative non-coding RNA species such as enhancer RNA (eRNA), vault RNA (vtRNA) and circular RNA (circRNA) [54,55,56]. Consequently, disruption to methylated bases could have multiple effects on cellular processes. We found consistent and contrasting differences in increases and decreases in expression in two m5C RNA writer proteins, NSUN7 and NSUN6, within the hippocampus and with disease/pathology groups. The function of NSUN7 is still relatively unknown although it has been suggested to regulate the stability of enhancer RNAs of genes targeted by the transcriptional co-activator PGC-1α [57]. Interestingly, PGC-1α, a known mitochondria and energy metabolism regulator, has been linked to neuronal survival and synaptic maintenance and its dysregulation is suggested to be involved in pathogenesis of neurodegenerative diseases [58,59,60].
In contrast, NSUN6 is known to methylate tRNAThr and tRNACys tRNA molecules in human cells [61] and was recently discovered, together with NSUN2, to determine most mRNA transcription-wide m5C sites [62]. Furthermore, NSUN6 is associated with what has been termed ‘type II’ m5C mRNA sites which contain a downstream UCCA motif and are predicted to be located in the loops of putative hairpin structures [62,63,64]. Like NSUN6, type II m5C mRNAs are commonly found in the cytoplasm of cells and in NSUN6 knock-out cells, and type II modification is associated with a modest overall increase in translation efficiency although translational efficiency is dependent on genic location [62]. Of interest, within several regions of the human brain, a circular RNA transcript is predicted to be encoded within NSUN6 coding regions [65, 66]. As circular RNAs can regulate mRNA through binding to RNA binding proteins such as RNA methylation effector proteins and have been shown to be abnormally expressed in Alzheimer’s disease brain [67], this highlights the potential for a more complex transcriptional regulatory system involving NSUN6 underlying AD pathology.
The m5C reader ALYREF, also known as THOC4, also showed increased expression with increasing Braak pathology. ALYREF has been described as the main regulator of m5C-modified mRNA export out of the nucleus, and mutations within transcription and export (TREX) complex proteins involved in the export process cause syndromic forms of intellectual disabilities [68,69,70]. Moreover, the recognition and hence nuclear-cytoplasmic shuttling of specific transcripts by ALYREF is reduced with the knockdown of NSUN2 [30, 71]. Together, these observations suggest that both NSUN6 and ALYREF require NSUN2 for their function in the distribution of m5C mRNA sites as well as of m5C mRNA transport into the cytoplasm and that tissue-specific changes in m5C writer abundance may have cell type–dependent consequences on protein translation and protein complexes within cytoplasmic sites.
Many studies have reported differential 5mC DNA methylation at mCG bases in AD or with Braak stage neuropathology. However, these reports mostly come from candidate gene studies or methylation array studies which examine pre-selected, known CpG sites often preferentially located within promoter regions [72,73,74]. To date, very few studies have performed hypothesis-free bisulphite DNA sequencing and hence have comprehensively examined, at a base resolution, mCG/mCH or 5hmC sites in individuals with dementia or TBI [75, 76]. As yet, no studies have assessed single-base transcriptome-wide mRNA m5C methylation in cohorts with neurodegenerative diseases. Therefore, the consequences of change to the DNA and RNA methylome and relationship to pathological processes are still unclear. However, the non-protein α-amino acid homocysteine is an important intermediate in the one-carbon pathway which is essential for the production of methyl groups available for DNA/RNA methylation [77]. High homocysteine is also an established risk factor for both AD and TBI [39, 78,79,80] and has been suggested to increase β-amyloid and tau pathology, protein aggregation as well as mitochondrial dysfunction involving oxidative stress pathways [81,82,83,84]. Further studies are needed to elucidate the functional relationship between changes in homocysteine, methylation effector-protein processes and pathology-inducing mechanisms.
One limitation of this study is that changes in expression were identified from heterogenous cellular tissue sections and were therefore not cell-type population specific or subcellular region specific. Cell nuclei extracts from healthy human brain tissue have been reported to show cell population–specific differences in 5mC DNA profiles with differences between oligodendrocyte and neuron population profiles estimated to be as high as ~ 35% [85]. Alterations in cell type composition in brain tissue with increased pathological staging are also well documented, e.g. atrophy of neuronal and glial cell populations concurrent with increases in reactive astrocytic and microglial population abundance. Such shifts in cell type abundance are one explanation for age-associated, or Braak stage–associated, changes in 5mC DNA [86, 87] or m5C RNA methylation profiles, and which could be a significant factor influencing effector protein transcript expression. Alternatively, mRNA methylation effector proteins are known to undergo autoregulation, i.e. are modified themselves [88], which potentially influences their spatiotemporal transcriptional abundance and hence regulatory feedback loops.
A second related limitation of this study is that we cannot say whether changes in effector transcript expression result in changes in protein abundance and hence whether post-transcriptional regulatory mechanisms, proteomic changes or a combination of both mechanisms may be influencing pathological processes. Indeed, knockdown of mouse Nsun2, which, like NSUN6, is part of the tRNA regulome, results in decreased tRNA m5C levels, deficits in tRNA glycine codon–specific defects and a loss of Gly-rich synaptic proteins [89]. Whether such consequential molecular changes are co-moderated by Nsun2 effector transcript mechanisms remains to be determined. However, cell population–specific high-throughput transcriptomic and proteomic studies of dementia and TBI pathological brain tissue will be necessary to elucidate methylome-specific mechanisms and consequences.
Our findings provide novel evidence of epitranscriptional control involved in AD and TBI, and with pharmacological targeting of DNA and RNA methyltransferases and methylation pathways currently underway for forms of cancers, new therapeutic avenues for dementia may advance.
Data availability
The dataset analysed during the current study are publicly available from the Allen Institute for Brain Science, University of Washington Medicine, and Kaiser Permanente Washington Health Research Institute (2016) (Aging, Dementia, and TBI Study [NG00059], available from aging.brain-map.org. RRID:SCR_014554 | Primary publication: Miller J. A., et al. (2017). Neuropathological and transcriptomic characteristics of the aged brain. eLife, 2017;6:e31126. https://doi.org/10.7554/eLife.31126).
References
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders (DSM-5®). American Psychiatric Publishing, Washington, United States
Nichols E, Steinmetz JD, Vollset SE et al (2022) Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health 7:e105–e125. https://doi.org/10.1016/S2468-2667(21)00249-8
(2018) World Alzheimer Reports | Alzheimer’s Disease International. https://www.alz.co.uk/research/world-report. Accessed 24 Mar 2020
DeTure MA, Dickson DW (2019) The neuropathological diagnosis of Alzheimer’s disease. Mol Neurodegener 14https://doi.org/10.1186/s13024-019-0333-5
Braak E, Strotkamp B, Braak H (1991) Parvalbumin-immunoreactive structures in the hippocampus of the human adult. Cell Tissue Res 264:33–48. https://doi.org/10.1007/BF00305720
Hyman BT, Phelps CH, Beach TG et al (2012) National Institute on Aging–Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimers Dement J Alzheimers Assoc 8:1–13. https://doi.org/10.1016/j.jalz.2011.10.007
Blennow K, Hardy J, Zetterberg H (2012) The neuropathology and neurobiology of traumatic brain injury. Neuron 76:886–899. https://doi.org/10.1016/j.neuron.2012.11.021
Dams-O’Connor K, Gibbons LE, Landau A et al (2016) Health problems precede traumatic brain injury in older adults. J Am Geriatr Soc 64:844–848. https://doi.org/10.1111/jgs.14014
Kenney K, Iacono D, Edlow BL et al (2018) Dementia after moderate-severe traumatic brain injury: coexistence of multiple proteinopathies. J Neuropathol Exp Neurol 77:50–63. https://doi.org/10.1093/jnen/nlx101
Smith DH, Dollé J-P, Ameen-Ali KE et al (2021) COllaborative Neuropathology NEtwork Characterizing ouTcomes of TBI (CONNECT-TBI). Acta Neuropathol Commun 9:32. https://doi.org/10.1186/s40478-021-01122-9
Torjesen I (2018) Traumatic brain injury raises dementia risk, large study suggests. BMJ k1597. https://doi.org/10.1136/bmj.k1597
Smith DH, Johnson VE, Stewart W (2013) Chronic neuropathologies of single and repetitive TBI: substrates of dementia? Nat Rev Neurol 9:211–221. https://doi.org/10.1038/nrneurol.2013.29
Wilson L, Stewart W, Dams-O’Connor K et al (2017) The chronic and evolving neurological consequences of traumatic brain injury. Lancet Neurol 16:813–825. https://doi.org/10.1016/S1474-4422(17)30279-X
Jaenisch R, Bird A (2003) Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nat Genet 33:245–254. https://doi.org/10.1038/ng1089
Mirabella AC, Foster BM, Bartke T (2016) Chromatin deregulation in disease. Chromosoma 125:75–93. https://doi.org/10.1007/s00412-015-0530-0
Lee J-H, Park S-J, Nakai K (2017) Differential landscape of non-CpG methylation in embryonic stem cells and neurons caused by DNMT3s. Sci Rep 7:11295. https://doi.org/10.1038/s41598-017-11800-1
Lister R, Mukamel EA, Nery JR et al (2013) Global epigenomic reconfiguration during mammalian brain development. Science 341:1237905. https://doi.org/10.1126/science.1237905
Sanchez-Mut JV, Heyn H, Vidal E et al (2016) Human DNA methylomes of neurodegenerative diseases show common epigenomic patterns. Transl Psychiatry 6:e718. https://doi.org/10.1038/tp.2015.214
Bird AP, Wolffe AP (1999) Methylation-induced repression–belts, braces, and chromatin. Cell 99:451–454. https://doi.org/10.1016/s0092-8674(00)81532-9
He Y-F, Li B-Z, Li Z et al (2011) Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Sci 333:1303–1307. https://doi.org/10.1126/science.1210944
Ito S, Shen L, Dai Q et al (2011) Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Sci 333:1300–1303. https://doi.org/10.1126/science.1210597
Bachman M, Uribe-Lewis S, Yang X et al (2015) 5-Formylcytosine can be a stable DNA modification in mammals. Nat Chem Biol 11:555–557. https://doi.org/10.1038/nchembio.1848
Inoue A, Shen L, Dai Q et al (2011) Generation and replication-dependent dilution of 5fC and 5caC during mouse preimplantation development. Cell Res 21:1670–1676. https://doi.org/10.1038/cr.2011.189
Shen L, Wu H, Diep D et al (2013) Genome-wide analysis reveals TET- and TDG-dependent 5-methylcytosine oxidation dynamics. Cell 153:692–706. https://doi.org/10.1016/j.cell.2013.04.002
Amos H, Korn M (1958) 5-Methyl cytosine in the RNA of Escherichia coli. Biochim Biophys Acta 29:444–445. https://doi.org/10.1016/0006-3002(58)90214-2
Basanta-Sanchez M, Wang R, Liu Z et al (2017) TET1-mediated oxidation of 5-formylcytosine (5fC) to 5-carboxycytosine (5caC) in RNA. Chembiochem Eur J Chem Biol 18:72–76. https://doi.org/10.1002/cbic.201600328
Kawarada L, Suzuki T, Ohira T et al (2017) ALKBH1 is an RNA dioxygenase responsible for cytoplasmic and mitochondrial tRNA modifications. Nucleic Acids Res 45:7401–7415. https://doi.org/10.1093/nar/gkx354
Van Haute L, Lee S-Y, McCann BJ et al (2019) NSUN2 introduces 5-methylcytosines in mammalian mitochondrial tRNAs. Nucleic Acids Res 47:8720–8733. https://doi.org/10.1093/nar/gkz559
Schumann U, Zhang H-N, Sibbritt T, et al (2020) Multiple links between 5-methylcytosine content of mRNA and translation. BMC Biol 18https://doi.org/10.1186/s12915-020-00769-5
Yang X, Yang Y, Sun B-F et al (2017) 5-Methylcytosine promotes mRNA export - NSUN2 as the methyltransferase and ALYREF as an m5C reader. Cell Res 27:606–625. https://doi.org/10.1038/cr.2017.55
Delatte B, Wang F, Ngoc LV et al (2016) RNA biochemistry. Transcriptome-wide distribution and function of RNA hydroxymethylcytosine. Sci 351:282–285. https://doi.org/10.1126/science.aac5253
Haag S, Sloan KE, Ranjan N et al (2016) NSUN3 and ABH1 modify the wobble position of mt-tRNAMet to expand codon recognition in mitochondrial translation. EMBO J 35:2104–2119. https://doi.org/10.15252/embj.201694885
Takemoto C, Spremulli LL, Benkowski LA et al (2009) Unconventional decoding of the AUA codon as methionine by mitochondrial tRNA Met with the anticodon f5CAU as revealed with a mitochondrial in vitro translation system. Nucleic Acids Res 37:1616–1627. https://doi.org/10.1093/nar/gkp001
Aging, Dementia and TBI Study (2017) Technical white paper: overview. Available from Allen Brain Atlas – Aging, Dementia and TBI. https://aging.brain-map.org/
Miller JA, Guillozet-Bongaarts A, Gibbons LE et al (2017) Neuropathological and transcriptomic characteristics of the aged brain. eLife 6:31126
Aging, Dementia and TBI Study (2017) Technical white paper: ACT cohort. Available from Allen Brain Atlas – Aging, Dementia and TBI. https://aging.brain-map.org/
The ENCODE Project Consortium (2012) An integrated encyclopedia of DNA elements in the human genome | Nature. https://www.nature.com/articles/nature11247. Accessed 9 Nov 2022
Dai Z, Ramesh V, Locasale JW (2020) The evolving metabolic landscape of chromatin biology and epigenetics. Nat Rev Genet 21:737–753. https://doi.org/10.1038/s41576-020-0270-8
Flitton M, Rielly N, Warman R et al (2019) Interaction of nutrition and genetics via DNMT3L-mediated DNA methylation determines cognitive decline. Neurobiol Aging 78:64–73. https://doi.org/10.1016/j.neurobiolaging.2019.02.001
Bayraktar & R. Kreutz G Michael (2018) Neuronal DNA methyltransferases: epigenetic mediators between synaptic activity and gene expression? - Gonca Bayraktar, Michael R. Kreutz, 2018. https://journals.sagepub.com/doi/https://doi.org/10.1177/1073858417707457. Accessed 4 Oct 2021
Chahrour M, Jung SY, Shaw C et al (2008) MeCP2, a key contributor to neurological disease, activates and represses transcription. Sci 320:1224–1229. https://doi.org/10.1126/science.1153252
Baets J, Duan X, Wu Y et al (2015) Defects of mutant DNMT1 are linked to a spectrum of neurological disorders. Brain J Neurol 138:845–861. https://doi.org/10.1093/brain/awv010
Klein CJ, Botuyan M-V, Wu Y et al (2011) Mutations in DNMT1 cause hereditary sensory neuropathy with dementia and hearing loss. Nat Genet 43:595–600. https://doi.org/10.1038/ng.830
Hansen RS, Wijmenga C, Luo P et al (1999) The DNMT3B DNA methyltransferase gene is mutated in the ICF immunodeficiency syndrome. Proc Natl Acad Sci U S A 96:14412–14417
Xu GL, Bestor TH, Bourc’his D, et al (1999) Chromosome instability and immunodeficiency syndrome caused by mutations in a DNA methyltransferase gene. Nature 402:187–191. https://doi.org/10.1038/46052
Ley TJ, Ding L, Walter MJ et al (2010) DNMT3A mutations in acute myeloid leukemia. N Engl J Med 363:2424–2433. https://doi.org/10.1056/NEJMoa1005143
Tatton-Brown K, Seal S, Ruark E et al (2014) Mutations in the DNA methyltransferase gene DNMT3A cause an overgrowth syndrome with intellectual disability. Nat Genet 46:385–388. https://doi.org/10.1038/ng.2917
Lyko F (2018) The DNA methyltransferase family: a versatile toolkit for epigenetic regulation. Nat Rev Genet 19:81–92. https://doi.org/10.1038/nrg.2017.80
Russler-Germain DA, Spencer DH, Young MA et al (2014) The R882H DNMT3A mutation associated with AML dominantly inhibits wild-type DNMT3A by blocking its ability to form active tetramers. Cancer Cell 25:442–454. https://doi.org/10.1016/j.ccr.2014.02.010
Spencer DH, Russler-Germain DA, Ketkar S et al (2017) CpG island hypermethylation mediated by DNMT3A is a consequence of AML progression. Cell 168:801-816.e13. https://doi.org/10.1016/j.cell.2017.01.021
Sun Z, Wu Y, Ordog T et al (2014) Aberrant signature methylome by DNMT1 hot spot mutation in hereditary sensory and autonomic neuropathy 1E. Epigenetics 9:1184–1193. https://doi.org/10.4161/epi.29676
Gabel HW, Kinde B, Stroud H et al (2015) Disruption of DNA-methylation-dependent long gene repression in Rett syndrome. Nature 522:89–93. https://doi.org/10.1038/nature14319
Kinde B, Wu DY, Greenberg ME, Gabel HW (2016) DNA methylation in the gene body influences MeCP2-mediated gene repression. Proc Natl Acad Sci U S A 113:15114–15119. https://doi.org/10.1073/pnas.1618737114
He Y, Zhang Q, Zheng Q et al (2020) Distinct 5-methylcytosine profiles of circular RNA in human hepatocellular carcinoma. Am J Transl Res 12:5719–5729
Hussain S, Sajini AA, Blanco S et al (2013) NSun2-mediated cytosine-5 methylation of vault noncoding RNA determines its processing into regulatory small RNAs. Cell Rep 4:255–261. https://doi.org/10.1016/j.celrep.2013.06.029
Rozenski J, Crain PF, McCloskey JA (1999) The RNA modification database: 1999 update. Nucleic Acids Res 27:196–197. https://doi.org/10.1093/nar/27.1.196
Aguilo F, Li S, Balasubramaniyan N et al (2016) Deposition of 5-methylcytosine on enhancer RNAs enables the coactivator function of PGC-1α. Cell Rep 14:479–492. https://doi.org/10.1016/j.celrep.2015.12.043
Cheng A, Wan R, Yang J-L et al (2012) Involvement of PGC-1α in the formation and maintenance of neuronal dendritic spines. Nat Commun 3:1250. https://doi.org/10.1038/ncomms2238
McMeekin LJ, Fox SN, Boas SM, Cowell RM (2021) Dysregulation of PGC-1α-dependent transcriptional programs in neurological and developmental disorders: therapeutic challenges and opportunities. Cells 10:352. https://doi.org/10.3390/cells10020352
Panes JD, Godoy PA, Silva-Grecchi T et al (2020) Changes in PGC-1α/SIRT1 signaling impact on mitochondrial homeostasis in amyloid-beta peptide toxicity model. Front Pharmacol 11:709. https://doi.org/10.3389/fphar.2020.00709
Haag S, Warda AS, Kretschmer J et al (2015) NSUN6 is a human RNA methyltransferase that catalyzes formation of m5C72 in specific tRNAs. RNA 21:1532–1543. https://doi.org/10.1261/rna.051524.115
Liu J, Huang T, Zhang Y, et al (2021) Sequence- and structure-selective mRNA m5C methylation by NSUN6 in animals. Natl Sci Rev 8https://doi.org/10.1093/nsr/nwaa273
Fang L, Wang W, Li G et al (2020) CIGAR-seq, a CRISPR/Cas-based method for unbiased screening of novel mRNA modification regulators. Mol Syst Biol 16:e10025. https://doi.org/10.15252/msb.202010025
Selmi T, Hussain S, Dietmann S et al (2020) Sequence- and structure-specific cytosine-5 mRNA methylation by NSUN6. Nucleic Acids Res 49:1006–1022. https://doi.org/10.1093/nar/gkaa1193
Rybak-Wolf A, Stottmeister C, Glažar P et al (2015) Circular RNAs in the mammalian brain are highly abundant, conserved, and dynamically expressed. Mol Cell 58:870–885. https://doi.org/10.1016/j.molcel.2015.03.027
Salzman J, Chen RE, Olsen MN et al (2013) Cell-type specific features of circular RNA expression. PLoS Genet 9:e1003777. https://doi.org/10.1371/journal.pgen.1003777
Dube U, Del-Aguila JL, Li Z et al (2019) An atlas of cortical circular RNA expression in Alzheimer disease brains demonstrates clinical and pathological associations. Nat Neurosci 22:1903–1912. https://doi.org/10.1038/s41593-019-0501-5
Heath CG, Viphakone N, Wilson SA (2016) The role of TREX in gene expression and disease. Biochem J 473:2911–2935. https://doi.org/10.1042/BCJ20160010
Kumar R, Corbett MA, van Bon BWM et al (2015) THOC2 mutations implicate mRNA-export pathway in X-linked intellectual disability. Am J Hum Genet 97:302–310. https://doi.org/10.1016/j.ajhg.2015.05.021
Pühringer T, Hohmann U, Fin L et al (2020) Structure of the human core transcription-export complex reveals a hub for multivalent interactions. eLife 9:61503. https://doi.org/10.7554/eLife.61503
Liu Y, Zhao Y, Wu R et al (2021) mRNA m5C controls adipogenesis by promoting CDKN1A mRNA export and translation. RNA Biol 18:711–721. https://doi.org/10.1080/15476286.2021.1980694
Lardenoije R, Roubroeks JAY, Pishva E et al (2019) Alzheimer’s disease-associated (hydroxy)methylomic changes in the brain and blood. Clin Epigenetics 11:164. https://doi.org/10.1186/s13148-019-0755-5
Slieker RC, Bos SD, Goeman JJ et al (2013) Identification and systematic annotation of tissue-specific differentially methylated regions using the Illumina 450k array. Epigenetics Chromatin 6:26. https://doi.org/10.1186/1756-8935-6-26
De Jager PL, Srivastava G, Lunnon K et al (2014) Alzheimer’s disease: early alterations in brain DNA methylation at ANK1, BIN1, RHBDF2 and other loci. Nat Neurosci 17:1156–1163. https://doi.org/10.1038/nn.3786
Chen L, Shen Q, Xu S et al (2022) 5-Hydroxymethylcytosine signatures in circulating cell-free DNA as diagnostic biomarkers for late-onset Alzheimer’s disease. J Alzheimers Dis JAD 85:573–585. https://doi.org/10.3233/JAD-215217
Konki M, Malonzo M, Karlsson IK et al (2019) Peripheral blood DNA methylation differences in twin pairs discordant for Alzheimer’s disease. Clin Epigenetics 11:130. https://doi.org/10.1186/s13148-019-0729-7
Brosnan JT, Brosnan ME (2006) The sulfur-containing amino acids: an overview. J Nutr 136:1636S-1640S. https://doi.org/10.1093/jn/136.6.1636S
Dash PK, Hergenroeder GW, Jeter CB et al (2016) Traumatic brain injury alters methionine metabolism: implications for pathophysiology. Front Syst Neurosci 10:36. https://doi.org/10.3389/fnsys.2016.00036
Morris MS (2003) Homocysteine and Alzheimer’s disease. Lancet Neurol 2:425–428. https://doi.org/10.1016/s1474-4422(03)00438-1
Rahmani A, Hatefi M, Dastjerdi MM et al (2016) Correlation between serum homocysteine levels and outcome of patients with severe traumatic brain injury. World Neurosurg 87:507–515. https://doi.org/10.1016/j.wneu.2015.09.016
Di Meco A, Li J-G, Barrero C et al (2019) Elevated levels of brain homocysteine directly modulate the pathological phenotype of a mouse model of tauopathy. Mol Psychiatry 24:1696–1706. https://doi.org/10.1038/s41380-018-0062-0
Khayati K, Antikainen H, Bonder EM et al (2017) The amino acid metabolite homocysteine activates mTORC1 to inhibit autophagy and form abnormal proteins in human neurons and mice. FASEB J Off Publ Fed Am Soc Exp Biol 31:598–609. https://doi.org/10.1096/fj.201600915R
Velazquez R, Ferreira E, Winslow W et al (2020) Maternal choline supplementation ameliorates Alzheimer’s disease pathology by reducing brain homocysteine levels across multiple generations. Mol Psychiatry 25:2620–2629. https://doi.org/10.1038/s41380-018-0322-z
Wang L, Niu H, Zhang J (2020) Homocysteine induces mitochondrial dysfunction and oxidative stress in myocardial ischemia/reperfusion injury through stimulating ROS production and the ERK1/2 signaling pathway. Exp Ther Med 20:938–944. https://doi.org/10.3892/etm.2020.8735
Mendizabal I, Berto S, Usui N et al (2019) Cell type-specific epigenetic links to schizophrenia risk in the brain. Genome Biol 20:135. https://doi.org/10.1186/s13059-019-1747-7
Gasparoni G, Bultmann S, Lutsik P et al (2018) DNA methylation analysis on purified neurons and glia dissects age and Alzheimer’s disease-specific changes in the human cortex. Epigenetics Chromatin 11:41. https://doi.org/10.1186/s13072-018-0211-3
Teschendorff AE, West J, Beck S (2013) Age-associated epigenetic drift: implications, and a case of epigenetic thrift? Hum Mol Genet 22:R7–R15. https://doi.org/10.1093/hmg/ddt375
Martinez De La Cruz B, Markus R, Malla S et al (2021) Modifying the m6A brain methylome by ALKBH5-mediated demethylation: a new contender for synaptic tagging. Mol Psychiatry 26:7141–7153. https://doi.org/10.1038/s41380-021-01282-z
Blaze J, Navickas A, Phillips H, et al (2021) Neuronal Nsun2 deficiency produces tRNA epitranscriptomic alterations and proteomic shifts impacting synaptic signaling and behavior. Nat Commun 12https://doi.org/10.1038/s41467-021-24969-x
Acknowledgements
The study used data taken from the Allen Institute for Brain Science, University of Washington Medicine, and Kaiser Permanente Washington Health Research Institute (2016) (Aging, Dementia, and TBI Study; Adult Changes in Thought study [NG00059], available from https://aging.brain-map.org. RRID:SCR_014554).
Funding
The project was funded by Nottingham University and the Neuroscience Support Group Charity, UK. A. PerezGrovas-Saltijeral was supported by a CONACYT PhD scholarship.
Author information
Authors and Affiliations
Contributions
H. M. Knight and A. PerezGrovas-Saltijeral designed the study. A. PerezGrovas-Saltijeral performed the data analysis. H. M. Knight, A. PerezGrovas-Saltijeral and A. P. Rajkumar wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The study used data from the Allen Institute for Brain Science, University of Washington Medicine, and Kaiser Permanente Washington Health Research Institute (2016) (Aging, Dementia, and TBI Study; Adult Changes in Thought study [NG00059]). Ethics for this study was reviewed and approved by Institutional Review Boards at Kaiser Permanente Washington and the University of Washington [35]. Consent to participate is not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
PerezGrovas-Saltijeral, A., Rajkumar, A.P. & Knight, H.M. Differential expression of m5C RNA methyltransferase genes NSUN6 and NSUN7 in Alzheimer’s disease and traumatic brain injury. Mol Neurobiol 60, 2223–2235 (2023). https://doi.org/10.1007/s12035-022-03195-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-03195-6