Abstract
Bone marrow mesenchymal stem cell (BMSC) is previously reported to present a certain effect on treating spinal cord injury (SCI), while the underlying mechanism is largely uncovered. Therefore, the current study aimed to investigate the involvement of exosome-delivered circRNA profile in the BMSC’s effect on pyroptosis for SCI treatment. H2O2 treated rat primary neurons were cultured with normal medium, BMSC, BMSC plus GW4869, and BMSC-derived exosome, respectively, then inflammasome-related pyroptosis markers, and circRNA profiles were detected. Subsequently, circ_003564-knockdown BMSC exosome was transfected into H2O2 treated rat primary neurons and NGF-stimulated PC-12 cells. Furthermore, in vivo validation was conducted. BMSC and BMSC-derived exosome both decreased inflammasome-related pyroptosis markers including cleaved caspase-1, GSDMD, NLRP3, IL-1β, and IL-18 in H2O2-treated neurons, while exosome-free BMSC (BMSC plus GW4869) did not obviously reduce these factors. Microarray assay revealed that BMSC (vs. exosome-free BMSC) and BMSC-derived exosome (vs. normal medium) greatly regulated circRNA profiles, which were enriched in neuroinflammation pathways (such as neurotrophin, apoptosis, and TNF). Among three functional candidate circRNAs (circ_015525, circ_008876, and circ_003564), circ_003564 was most effective to regulate inflammasome-related pyroptosis. Interestingly, circ_003564-knockdown BMSC exosome showed higher expression of inflammasome-related pyroptosis markers compared to negative-control-knockdown BMSC exosome in H2O2 treated primary neurons/NGF-stimulated PC-12 cells. In vivo, BMSC exosome improved the function recovery and decreased tissue injury and inflammasome-related pyroptosis in SCI rats, whose effect was attenuated by circ_003564 knockdown transfection. BMSC exosome attenuates inflammasome-related pyroptosis via delivering circ_003564, contributing to its treatment efficacy for SCI.











Similar content being viewed by others
Availability of Data and Materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Perrouin-Verbe B, Lefevre C, Kieny P, Gross R, Reiss B, Le Fort M (2021) Spinal cord injury: a multisystem physiological impairment/dysfunction. Rev Neurol (Paris) 177(5):594–605. https://doi.org/10.1016/j.neurol.2021.02.385
Huang H, Young W, Skaper S, Chen L, Moviglia G, Saberi H, Al-Zoubi Z, Sharma HS et al (2020) Clinical neurorestorative therapeutic guidelines for spinal cord injury (IANR/CANR version 2019). J Orthop Translat 20:14–24. https://doi.org/10.1016/j.jot.2019.10.006
Anjum A, Yazid MD, Fauzi Daud M, Idris J, Ng AMH, Selvi Naicker A, Ismail OHR, Athi Kumar RK et al (2020) Spinal cord injury: pathophysiology, multimolecular interactions, and underlying recovery mechanisms. Int J Mol Sci 21(20). https://doi.org/10.3390/ijms21207533
Hachem LD, Fehlings MG (2021) Pathophysiology of spinal cord injury. Neurosurg Clin N Am 32(3):305–313. https://doi.org/10.1016/j.nec.2021.03.002
Liu X, Zhang Y, Wang Y, Qian T (2021) Inflammatory response to spinal cord injury and its treatment. World Neurosurg 155:19–31. https://doi.org/10.1016/j.wneu.2021.07.148
Al Mamun A, Wu Y, Monalisa I, Jia C, Zhou K, Munir F, Xiao J (2021) Role of pyroptosis in spinal cord injury and its therapeutic implications. J Adv Res 28:97–109. https://doi.org/10.1016/j.jare.2020.08.004
Kreydin E, Welk B, Chung D, Clemens Q, Yang C, Danforth T, Gousse A, Kielb S et al (2018) Surveillance and management of urologic complications after spinal cord injury. World J Urol 36(10):1545–1553. https://doi.org/10.1007/s00345-018-2345-0
Sueda T, Takahashi S (2018) Spinal cord injury as a complication of thoracic endovascular aneurysm repair. Surg Today 48(5):473–477. https://doi.org/10.1007/s00595-017-1588-5
Yu P, Zhang X, Liu N, Tang L, Peng C, Chen X (2021) Pyroptosis: mechanisms and diseases. Signal Transduct Target Ther 6(1):128. https://doi.org/10.1038/s41392-021-00507-5
Hu X, Chen H, Xu H, Wu Y, Wu C, Jia C, Li Y, Sheng S et al (2020) Role of pyroptosis in traumatic brain and spinal cord injuries. Int J Biol Sci 16(12):2042–2050. https://doi.org/10.7150/ijbs.45467
Xu S, Wang J, Zhong J, Shao M, Jiang J, Song J, Zhu W, Zhang F et al (2021) CD73 alleviates GSDMD-mediated microglia pyroptosis in spinal cord injury through PI3K/AKT/Foxo1 signaling. Clin Transl Med 11(1):e269. https://doi.org/10.1002/ctm2.269
Liau LL, Looi QH, Chia WC, Subramaniam T, Ng MH, Law JX (2020) Treatment of spinal cord injury with mesenchymal stem cells. Cell Biosci 10:112. https://doi.org/10.1186/s13578-020-00475-3
Lin L, Lin H, Bai S, Zheng L, Zhang X (2018) Bone marrow mesenchymal stem cells (BMSCs) improved functional recovery of spinal cord injury partly by promoting axonal regeneration. Neurochem Int 115:80–84. https://doi.org/10.1016/j.neuint.2018.02.007
Lv C, Zhang T, Li K, Gao K (2020) Bone marrow mesenchymal stem cells improve spinal function of spinal cord injury in rats via TGF-beta/Smads signaling pathway. Exp Ther Med 19(6):3657–3663. https://doi.org/10.3892/etm.2020.8640
Chen Y, Lian XH, Liao LY, Liu YT, Liu SL, Gao Q (2019) Transplantation of bone marrow mesenchymal stem cells alleviates spinal cord injury via inhibiting Notch signaling. Eur Rev Med Pharmacol Sci 23(3 Suppl):31–38. https://doi.org/10.26355/eurrev_201908_18625
Liu X, Xu W, Zhang Z, Liu H, Lv L, Han D, Liu L, Yao A et al (2020) Vascular endothelial growth factor-transfected bone marrow mesenchymal stem cells improve the recovery of motor and sensory functions of rats with spinal cord injury. Spine 45(7):E364–E372. https://doi.org/10.1097/BRS.0000000000003333
Zhou Y, Wen LL, Li YF, Wu KM, Duan RR, Yao YB, Jing LJ, Gong Z et al (2022) Exosomes derived from bone marrow mesenchymal stem cells protect the injured spinal cord by inhibiting pericyte pyroptosis. Neural Regen Res 17(1):194–202. https://doi.org/10.4103/1673-5374.314323
Sheng Y, Zhou X, Wang J, Shen H, Wu S, Guo W, Yang Y (2021) MSC derived EV loaded with miRNA-22 inhibits the inflammatory response and nerve function recovery after spinal cord injury in rats. J Cell Mol Med 25(21):10268–10278. https://doi.org/10.1111/jcmm.16965
Li C, Qin T, Zhao J, He R, Wen H, Duan C, Lu H, Cao Y et al (2021) Bone marrow mesenchymal stem cell-derived exosome-educated macrophages promote functional healing after spinal cord injury. Front Cell Neurosci 15:725573. https://doi.org/10.3389/fncel.2021.725573
Xin W, Qiang S, Jianing D, Jiaming L, Fangqi L, Bin C, Yuanyuan C, Guowang Z et al (2021) Human bone marrow mesenchymal stem cell-derived exosomes attenuate blood-spinal cord barrier disruption via the TIMP2/MMP pathway after acute spinal cord injury. Mol Neurobiol 58(12):6490–6504. https://doi.org/10.1007/s12035-021-02565-w
Dilsiz N (2022) Hallmarks of exosomes. Future Sci OA 8(1):FSO764. https://doi.org/10.2144/fsoa-2021-0102
Chen M, Lai X, Wang X, Ying J, Zhang L, Zhou B, Liu X, Zhang J et al (2021) Long non-coding RNAs and circular RNAs: insights into microglia and astrocyte mediated neurological diseases. Front Mol Neurosci 14:745066. https://doi.org/10.3389/fnmol.2021.745066
Bie F, Wang K, Xu T, Yuan J, Ding H, Lv B, Liu Y, Lan M (2021) The potential roles of circular RNAs as modulators in traumatic spinal cord injury. Biomed Pharmacother 141:111826. https://doi.org/10.1016/j.biopha.2021.111826
Fanale D, Taverna S, Russo A, Bazan V (2018) Circular RNA in exosomes. Adv Exp Med Biol 1087:109–117. https://doi.org/10.1007/978-981-13-1426-1_9
Zhang M, Liu D, Li S, Chang L, Zhang Y, Liu R, Sun F, Duan W et al (2015) Bone marrow mesenchymal stem cell transplantation retards the natural senescence of rat hearts. Stem Cells Transl Med 4(5):494–502. https://doi.org/10.5966/sctm.2014-0206
Kehl LJ, Fairbanks CA, Laughlin TM, Wilcox GL (1997) Neurogenesis in postnatal rat spinal cord: a study in primary culture. Science 276(5312):586–589. https://doi.org/10.1126/science.276.5312.586
Luo Z, Wu F, Xue E, Huang L, Yan P, Pan X, Zhou Y (2019) Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival by inducing HIF-1alpha in injured neuronal cells derived exosomes culture system. Cell Death Dis 10(2):134. https://doi.org/10.1038/s41419-019-1410-y
Che Y, Shi X, Shi Y, Jiang X, Ai Q, Shi Y, Gong F, Jiang W (2019) Exosomes derived from miR-143-overexpressing MSCs inhibit cell migration and invasion in human prostate cancer by downregulating TFF3. Mol Ther Nucleic Acids 18:232–244. https://doi.org/10.1016/j.omtn.2019.08.010
Wang N, Yang Y, Pang M, Du C, Chen Y, Li S, Tian Z, Feng F et al (2020) MicroRNA-135a-5p promotes the functional recovery of spinal cord injury by targeting SP1 and ROCK. Mol Ther Nucleic Acids 22:1063–1077. https://doi.org/10.1016/j.omtn.2020.08.035
Luo W, Wang Y, Lin F, Liu Y, Gu R, Liu W, Xiao C (2020) Selenium-doped carbon quantum dots efficiently ameliorate secondary spinal cord injury via scavenging reactive oxygen species. Int J Nanomed 15:10113–10125. https://doi.org/10.2147/IJN.S282985
Luo Y, Xu T, Liu W, Rong Y, Wang J, Fan J, Yin G, Cai W (2021) Exosomes derived from GIT1-overexpressing bone marrow mesenchymal stem cells promote traumatic spinal cord injury recovery in a rat model. Int J Neurosci 131(2):170–182. https://doi.org/10.1080/00207454.2020.1734598
Rong H, Zhao Z, Feng J, Lei Y, Wu H, Sun R, Zhang Z, Hou B et al (2017) The effects of dexmedetomidine pretreatment on the pro- and anti-inflammation systems after spinal cord injury in rats. Brain Behav Immun 64:195–207. https://doi.org/10.1016/j.bbi.2017.03.006
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21. https://doi.org/10.1089/neu.1995.12.1
Li TE, Wang S, Shen XT, Zhang Z, Chen M, Wang H, Zhu Y, Xu D et al (2020) PKM2 drives hepatocellular carcinoma progression by inducing immunosuppressive microenvironment. Front Immunol 11:589997. https://doi.org/10.3389/fimmu.2020.589997
Li GQ, Fang YX, Liu Y, Meng FR, Wu X, Zhang CW, Zhang Y, Liu YQ et al (2021) MicroRNA-21 from bone marrow mesenchymal stem cell-derived extracellular vesicles targets TET1 to suppress KLF4 and alleviate rheumatoid arthritis. Ther Adv Chronic Dis 12:20406223211007369. https://doi.org/10.1177/20406223211007369
Wang Q, Zhao SF, Xiao X, Liu YN, Wang XL, Du YX (2022) Combined treatment of bone marrow mesenchymal stem cells and fasudil promotes neurovascular remodeling and neurological function recovery in ischemic stroke. Appl Biochem Biotechnol 194(2):801–812. https://doi.org/10.1007/s12010-021-03679-6
Khodabandeh Z, Mehrabani D, Dehghani F, Gashmardi N, Erfanizadeh M, Zare S, Bozorg-Ghalati F (2021) Spinal cord injury repair using mesenchymal stem cells derived from bone marrow in mice: a stereological study. Acta Histochem 123(5):151720. https://doi.org/10.1016/j.acthis.2021.151720
Oraee-Yazdani S, Akhlaghpasand M, Golmohammadi M, Hafizi M, Zomorrod MS, Kabir NM, Oraee-Yazdani M, Ashrafi F et al (2021) Combining cell therapy with human autologous Schwann cell and bone marrow-derived mesenchymal stem cell in patients with subacute complete spinal cord injury: safety considerations and possible outcomes. Stem Cell Res Ther 12(1):445. https://doi.org/10.1186/s13287-021-02515-2
Nie H, Jiang Z (2021) Bone mesenchymal stem cell-derived extracellular vesicles deliver microRNA-23b to alleviate spinal cord injury by targeting toll-like receptor TLR4 and inhibiting NF-kappaB pathway activation. Bioengineered 12(1):8157–8172. https://doi.org/10.1080/21655979.2021.1977562
Zhou L, Ye H, Liu L, Chen Y (2021) Human bone mesenchymal stem cell-derived exosomes inhibit IL-1beta-induced inflammation in osteoarthritis chondrocytes. Cell J 23(4):485–494. https://doi.org/10.22074/cellj.2021.7127
Zong L, Huang P, Song Q, Kang Y (2021) Bone marrow mesenchymal stem cells-secreted exosomal H19 modulates lipopolysaccharides-stimulated microglial M1/M2 polarization and alleviates inflammation-mediated neurotoxicity. Am J Transl Res 13(3):935–951
Charbord P (2010) Bone marrow mesenchymal stem cells: historical overview and concepts. Hum Gene Ther 21(9):1045–1056. https://doi.org/10.1089/hum.2010.115
Batsali AK, Georgopoulou A, Mavroudi I, Matheakakis A, Pontikoglou CG, Papadaki HA (2020) The role of bone marrow mesenchymal stem cell derived extracellular vesicles (MSC-EVs) in normal and abnormal hematopoiesis and their therapeutic potential. J Clin Med 9(3). https://doi.org/10.3390/jcm9030856
He L, He T, Xing J, Zhou Q, Fan L, Liu C, Chen Y, Wu D et al (2020) Bone marrow mesenchymal stem cell-derived exosomes protect cartilage damage and relieve knee osteoarthritis pain in a rat model of osteoarthritis. Stem Cell Res Ther 11(1):276. https://doi.org/10.1186/s13287-020-01781-w
Fan L, Dong J, He X, Zhang C, Zhang T (2021) Bone marrow mesenchymal stem cells-derived exosomes reduce apoptosis and inflammatory response during spinal cord injury by inhibiting the TLR4/MyD88/NF-kappaB signaling pathway. Hum Exp Toxicol 40(10):1612–1623. https://doi.org/10.1177/09603271211003311
Liu X, Zhang M, Liu H, Zhu R, He H, Zhou Y, Zhang Y, Li C et al (2021) Bone marrow mesenchymal stem cell-derived exosomes attenuate cerebral ischemia-reperfusion injury-induced neuroinflammation and pyroptosis by modulating microglia M1/M2 phenotypes. Exp Neurol 341:113700. https://doi.org/10.1016/j.expneurol.2021.113700
Caba L, Florea L, Gug C, Dimitriu DC, Gorduza EV (2021) Circular RNA-is the circle perfect? Biomolecules 11(12). https://doi.org/10.3390/biom11121755
Chen X, Zhou M, Yant L, Huang C (2022) Circular RNA in disease: basic properties and biomedical relevance. Wiley Interdiscip Rev RNA:e1723. https://doi.org/10.1002/wrna.1723
Wu R, Mao S, Wang Y, Zhou S, Liu Y, Liu M, Gu X, Yu B (2019) Differential circular RNA expression profiles following spinal cord injury in rats: a temporal and experimental analysis. Front Neurosci 13:1303. https://doi.org/10.3389/fnins.2019.01303
Yao Y, Wang J, He T, Li H, Hu J, Zheng M, Ding Y, Chen YY et al (2020) Microarray assay of circular RNAs reveals cicRNA.7079 as a new anti-apoptotic molecule in spinal cord injury in mice. Brain Res Bull 164:157–171. https://doi.org/10.1016/j.brainresbull.2020.08.004
Li X, Kang J, Lv H, Liu R, Chen J, Zhang Y, Zhang Y, Yu G et al (2021) CircPrkcsh, a circular RNA, contributes to the polarization of microglia towards the M1 phenotype induced by spinal cord injury and acts via the JNK/p38 MAPK pathway. FASEB J 35(12):e22014. https://doi.org/10.1096/fj.202100993R
Chen J, Fu B, Bao J, Su R, Zhao H, Liu Z (2021) Novel circular RNA 2960 contributes to secondary damage of spinal cord injury by sponging miRNA-124. J Comp Neurol 529(7):1456–1464. https://doi.org/10.1002/cne.25030
Wang W, He D, Chen J, Zhang Z, Wang S, Jiang Y, Wei J (2021) Circular RNA Plek promotes fibrogenic activation by regulating the miR-135b-5p/TGF-betaR1 axis after spinal cord injury. Aging (Albany NY) 13(9):13211–13224. https://doi.org/10.18632/aging.203002
Funding
This study was supported by National Natural Science Foundation of China (No. 81801209), Shanghai Clinical Medical Center (Grant Number 2017ZZ01023), and Shanghai Municipal Key Clinical Specialty (Grant Number shslczdzk00402, shslczdzk07001).
Author information
Authors and Affiliations
Contributions
CL and DT contributed to the conception and design of the study. YZ and YC perform the experiments. ZW contributed to the data acquisition. CX and SQ contributed to the analysis and interpretation of data. TL drafted the manuscript. KQ contributed to revising of article. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
This study was approved by the Animal Care and Use Committee, and the experimental protocol was by the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Supplementary file3
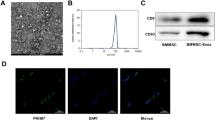
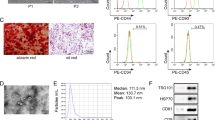
Supplementary figure 1. Isolation of BMSC and its exosome. BMSC marker expressions (A), BMSC morphology (B), size distribution of BMSC exosome (C), exosome marker expressions (D). (PNG 2873 kb)

Supplementary file4
Supplementary figure 2. Inflammasome-related pyroptosis after H2O2 treatment. Protein expression of cleaved caspase 1, GSDMD, NLRP3, IL-1β, and IL-18 (A) between control and H2O2 groups. mRNA expression of GSDMD, NLRP3, IL-1β, and IL-18 (B) between control and H2O2 groups. (PNG 1281 kb)

Supplementary file5
Supplementary figure 3. Circ_003564 expression after transfection. Circ_003564 expression in BMSC after transfection (A), circ_003564 expression in BMSC exosome after transfection (B). (PNG 337 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Y., Chen, Y., Wang, Z. et al. Bone Marrow Mesenchymal Stem Cell Exosome Attenuates Inflammasome-Related Pyroptosis via Delivering circ_003564 to Improve the Recovery of Spinal Cord Injury. Mol Neurobiol 59, 6771–6789 (2022). https://doi.org/10.1007/s12035-022-03006-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-03006-y