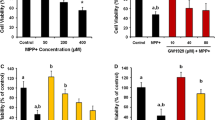
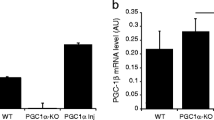
Abstract
Parkinson’s disease (PD) is a progressive neurodegenerative disease affecting over five million individuals worldwide. The exact molecular events underlying PD pathogenesis are still not clearly known. Glia maturation factor (GMF), a neuroinflammatory protein in the brain plays an important role in the pathogenesis of PD. Mitochondrial dysfunctions and oxidative stress trigger apoptosis leading to dopaminergic neuronal degeneration in PD. Peroxisome proliferator-activated receptor-gamma coactivator-1 alpha (PGC-1α or PPARGC-α) acts as a transcriptional co-regulator of mitochondrial biogenesis and energy metabolism by controlling oxidative phosphorylation, antioxidant activity, and autophagy. In this study, we found that incubation of immortalized rat dopaminergic (N27) neurons with GMF influences the expression of peroxisome PGC-1α and increases oxidative stress, mitochondrial dysfunction, and apoptotic cell death. We show that incubation with GMF reduces the expression of PGC-1α with concomitant decreases in the mitochondrial complexes. Besides, there is increased oxidative stress and depolarization of mitochondrial membrane potential (MMP) in these cells. Further, GMF reduces tyrosine hydroxylase (TH) expression and shifts Bax/Bcl-2 expression resulting in release of cytochrome-c and increased activations of effector caspase expressions. Transmission electron microscopy analyses revealed alteration in the mitochondrial architecture. Our results show that GMF acts as an important upstream regulator of PGC-1α in promoting dopaminergic neuronal death through its effect on oxidative stress-mediated apoptosis. Our current data suggest that GMF is a critical risk factor for PD and suggest that it could be explored as a potential therapeutic target to inhibit PD progression.














Similar content being viewed by others
References
Blum D, Torch S, Lambeng N, Nissou M, Benabid AL, Sadoul R, Verna JM (2001) Molecular pathways involved in the neurotoxicity of 6-OHDA, dopamine and MPTP: contribution to the apoptotic theory in Parkinson’s disease. Prog Neurobiol 65(2):135–172
Frank C, Pari G, Rossiter JP (2006) Approach to diagnosis of Parkinson disease. Can Fam Physician 52:862–868
Cory-Slechta DA, Thiruchelvam M, Di Monte DA (2008) Letter regarding: Paraquat: the red herring of Parkinson’s disease research. Toxicol Sci 103(1):215–216. https://doi.org/10.1093/toxsci/kfm309
Eriksen JL, Wszolek Z, Petrucelli L (2005) Molecular pathogenesis of Parkinson disease. Arch Neurol 62(3):353–357. https://doi.org/10.1001/archneur.62.3.353
Kieburtz K, Wunderle KB (2013) Parkinson’s disease: evidence for environmental risk factors. Mov Disord 28(1):8–13. https://doi.org/10.1002/mds.25150
Ryan SD, Dolatabadi N, Chan SF, Zhang X, Akhtar MW, Parker J, Soldner F, Sunico CR, Nagar S, Talantova M, Lee B, Lopez K, Nutter A, Shan B, Molokanova E, Zhang Y, Han X, Nakamura T, Masliah E, Yates JR, 3rd, Nakanishi N, Andreyev AY, Okamoto S, Jaenisch R, Ambasudhan R, Lipton SA (2013) Isogenic human iPSC Parkinson’s model shows nitrosative stress-induced dysfunction in MEF2-PGC1alpha transcription. Cell 155 (6):1351–1364. doi:https://doi.org/10.1016/j.cell.2013.11.009
Nicholls DG, Budd SL (2000) Mitochondria and neuronal survival. Physiol Rev 80(1):315–360. https://doi.org/10.1152/physrev.2000.80.1.315
Lee SB, Bae IH, Bae YS, Um HD (2006) Link between mitochondria and NADPH oxidase 1 isozyme for the sustained production of reactive oxygen species and cell death. J Biol Chem 281(47):36228–36235. https://doi.org/10.1074/jbc.M606702200
Mattson MP (2000) Apoptosis in neurodegenerative disorders. Nat Rev Mol Cell Biol 1(2):120–129. https://doi.org/10.1038/35040009
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909. https://doi.org/10.1016/S0896-6273(03)00568-3
Hu XM, Zeng FD (2004) Protective effects of sodium beta-aescin on ischemia-reperfusion injury in rat brain. Yao Xue Xue Bao 39(6):419–423
Selvakumar GP, Manivasagam T, Rekha KR, Jayaraj RL, Elangovan N (2015) Escin, a novel triterpene, mitigates chronic MPTP/p-induced dopaminergic toxicity by attenuating mitochondrial dysfunction, oxidative stress, and apoptosis. J Mol Neurosci 55(1):184–197. https://doi.org/10.1007/s12031-014-0303-x
Islam MT (2017) Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol Res 39(1):73–82. https://doi.org/10.1080/01616412.2016.1251711
Urrutia PJ, Mena NP, Nunez MT (2014) The interplay between iron accumulation, mitochondrial dysfunction, and inflammation during the execution step of neurodegenerative disorders. Front Pharmacol 5:38. https://doi.org/10.3389/fphar.2014.00038
Cheng A, Hou Y, Mattson MP (2010) Mitochondria and neuroplasticity. ASN Neuro 2 (5):e00045. doi:https://doi.org/10.1042/AN20100019, AN20100019
Lin J, Handschin C, Spiegelman BM (2005) Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab 1 (6):361–370. doi:https://doi.org/10.1016/j.cmet.2005.05.004
Scarpulla RC (2002) Nuclear activators and coactivators in mammalian mitochondrial biogenesis. Biochim Biophys Acta 1576(1–2):1–14. https://doi.org/10.1016/S0167-4781(02)00343-3
Scarpulla RC (2011) Metabolic control of mitochondrial biogenesis through the PGC-1 family regulatory network. Biochim Biophys Acta 1813(7):1269–1278. https://doi.org/10.1016/j.bbamcr.2010.09.019
St-Pierre J, Drori S, Uldry M, Silvaggi JM, Rhee J, Jager S, Handschin C, Zheng K et al (2006) Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell 127(2):397–408. https://doi.org/10.1016/j.cell.2006.09.024
Austin S, St-Pierre J (2012) PGC1alpha and mitochondrial metabolism—emerging concepts and relevance in ageing and neurodegenerative disorders. J Cell Sci 125(Pt 21):4963–4971. https://doi.org/10.1242/jcs.113662
Kaplan R, Zaheer A, Jaye M, Lim R (1991) Molecular cloning and expression of biologically active human glia maturation factor-beta. J Neurochem 57(2):483–490. https://doi.org/10.1111/j.1471-4159.1991.tb03777.x
Lim R, Zaheer A, Lane WS (1990) Complete amino acid sequence of bovine glia maturation factor beta. Proc Natl Acad Sci U S A 87 (14):5233–5237
Zaheer A, Fink BD, Lim R (1993) Expression of glia maturation factor β mRNA and protein in rat organs and cells. J Neurochem 60(3):914–920. https://doi.org/10.1111/j.1471-4159.1993.tb03237.x
Lim R, Zaheer A (1995) Phorbol ester stimulates rapid intracellular phosphorylation of glia maturation factor. Biochem Biophys Res Commun 211(3):928–934. https://doi.org/10.1006/bbrc.1995.1901
Lim R, Zaheer A (1996) In vitro enhancement of p38 mitogen-activated protein kinase activity by phosphorylated glia maturation factor. J Biol Chem 271(38):22953–22956. https://doi.org/10.1074/jbc.271.38.22953
Kempuraj D, Thangavel R, Natteru PA, Selvakumar GP, Saeed D, Zahoor H, Zaheer S, Iyer SS et al (2016) Neuroinflammation Induces Neurodegeneration. J Neurol Neurosurg Spine 1(1)
Kempuraj D, Khan MM, Thangavel R, Xiong Z, Yang E, Zaheer A (2013) Glia maturation factor induces interleukin-33 release from astrocytes: Implications for neurodegenerative diseases. J NeuroImmune Pharmacol 8(3):643–650. https://doi.org/10.1007/s11481-013-9439-7
Zaheer S, Wu Y, Bassett J, Yang B, Zaheer A (2007) Glia maturation factor regulation of STAT expression: a novel mechanism in experimental autoimmune encephalomyelitis. Neurochem Res 32(12):2123–2131. https://doi.org/10.1007/s11064-007-9383-0
Zaheer S, Wu Y, Sahu SK, Zaheer A (2011) Suppression of neuro inflammation in experimental autoimmune encephalomyelitis by glia maturation factor antibody. Brain Res 1373:230–239. https://doi.org/10.1016/j.brainres.2010.12.003
Ito J, Kato T, Yamakawa Y, Kato H, Sakazaki Y, Lim R, Tanaka R (1982) Interaction of glia maturation factor with the glial cell membrane. Brain Res 243(2):309–314
Choudhury A, Marks DL, Proctor KM, Gould GW, Pagano RE (2006) Regulation of caveolar endocytosis by syntaxin 6-dependent delivery of membrane components to the cell surface. Nat Cell Biol 8(4):317–328. https://doi.org/10.1038/ncb1380
Aerbajinai W, Liu L, Zhu J, Kumkhaek C, Chin K, Rodgers GP (2016) Glia maturation factor-gamma regulates monocyte migration through modulation of beta1-integrin. J Biol Chem 291(16):8549–8564. https://doi.org/10.1074/jbc.M115.674200
Afeseh Ngwa H, Kanthasamy A, Anantharam V, Song C, Witte T, Houk R, Kanthasamy AG (2009) Vanadium induces dopaminergic neurotoxicity via protein kinase Cdelta dependent oxidative signaling mechanisms: relevance to etiopathogenesis of Parkinson’s disease. Toxicol Appl Pharmacol 240(2):273–285. https://doi.org/10.1016/j.taap.2009.07.025
Afeseh Ngwa H, Kanthasamy A, Gu Y, Fang N, Anantharam V, Kanthasamy AG (2011) Manganese nanoparticle activates mitochondrial dependent apoptotic signaling and autophagy in dopaminergic neuronal cells. Toxicol Appl Pharmacol 256(3):227–240. https://doi.org/10.1016/j.taap.2011.07.018
Zawada WM, Banninger GP, Thornton J, Marriott B, Cantu D, Rachubinski AL, Das M, Griffin WS et al (2011) Generation of reactive oxygen species in 1-methyl-4-phenylpyridinium (MPP+) treated dopaminergic neurons occurs as an NADPH oxidase-dependent two-wave cascade. J Neuroinflammation 8(1):129. https://doi.org/10.1186/1742-2094-8-129
Hansen MB, Nielsen SE, Berg K (1989) Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J Immunol Methods 119(2):203–210. https://doi.org/10.1016/0022-1759(89)90397-9
Zaheer A, Knight S, Zaheer A, Ahrens M, Sahu SK, Yang B (2008) Glia maturation factor overexpression in neuroblastoma cells activates glycogen synthase kinase-3beta and caspase-3. Brain Res 1190:206–214. https://doi.org/10.1016/j.brainres.2007.11.011
Khan MM, Kempuraj D, Zaheer S, Zaheer A (2014) Glia maturation factor deficiency suppresses 1-methyl-4-phenylpyridinium-induced oxidative stress in astrocytes. J Mol Neurosci 53(4):590–599. https://doi.org/10.1007/s12031-013-0225-z
Zaheer A, Zaheer S, Sahu SK, Knight S, Khosravi H, Mathur SN, Lim R (2007) A novel role of glia maturation factor: induction of granulocyte-macrophage colony-stimulating factor and pro-inflammatory cytokines. J Neurochem 101(2):364–376. https://doi.org/10.1111/j.1471-4159.2006.04385.x
Lunov O, Zablotskii V, Churpita O, Chanova E, Sykova E, Dejneka A, Kubinova S (2014) Cell death induced by ozone and various non-thermal plasmas: therapeutic perspectives and limitations. Sci Rep 4(1):7129. https://doi.org/10.1038/srep07129
Kasibhatla S, Amarante-Mendes GP, Finucane D, Brunner T, Bossy-Wetzel E, Green DR (2006) Acridine orange/ethidium bromide (AO/EB) staining to detect apoptosis. CSH Protoc 2006(3). https://doi.org/10.1101/pdb.prot4493
Rajendra Prasad N, Karthikeyan A, Karthikeyan S, Reddy BV (2011) Inhibitory effect of caffeic acid on cancer cell proliferation by oxidative mechanism in human HT-1080 fibrosarcoma cell line. Mol Cell Biochem 349(1–2):11–19. https://doi.org/10.1007/s11010-010-0655-7
Tamilselvam K, Braidy N, Manivasagam T, Essa MM, Prasad NR, Karthikeyan S, Thenmozhi AJ, Selvaraju S, Guillemin GJ (2013) Neuroprotective effects of hesperidin, a plant flavanone, on rotenone-induced oxidative stress and apoptosis in a cellular model for Parkinson’s disease. Oxidative Med Cell Longev :102741. doi:https://doi.org/10.1155/2013/102741, 2013, 1, 11
Sastre E, Caracuel L, Prieto I, Llevenes P, Aller MA, Arias J, Balfagon G, Blanco-Rivero J (2016) Decompensated liver cirrhosis and neural regulation of mesenteric vascular tone in rats: role of sympathetic, nitrergic and sensory innervations. Sci Rep 6(1):31076. https://doi.org/10.1038/srep31076
Xu S, Zhou Z, Zhang L, Yu Z, Zhang W, Wang Y, Wang X, Li M et al (2010) Exposure to 1800 MHz radiofrequency radiation induces oxidative damage to mitochondrial DNA in primary cultured neurons. Brain Res 1311:189–196. https://doi.org/10.1016/j.brainres.2009.10.062
Dickson DW (2012) Parkinson's disease and parkinsonism: neuropathology. Cold Spring Harb Perspect Med 2(8). https://doi.org/10.1101/cshperspect.a009258
Drechsel DA, Liang LP, Patel M (2007) 1-methyl-4-phenylpyridinium-induced alterations of glutathione status in immortalized rat dopaminergic neurons. Toxicol Appl Pharmacol 220(3):341–348. https://doi.org/10.1016/j.taap.2007.02.002
Anantharam A, Diverse-Pierluissi MA (2002) Biochemical approaches to study interaction of calcium channels with RGS12 in primary neuronal cultures. Methods Enzymol 345:60–70. https://doi.org/10.1016/S0076-6879(02)45007-0
Kaul S, Kanthasamy A, Kitazawa M, Anantharam V, Kanthasamy AG (2003) Caspase-3 dependent proteolytic activation of protein kinase C delta mediates and regulates 1-methyl-4-phenylpyridinium (MPP+)-induced apoptotic cell death in dopaminergic cells: Relevance to oxidative stress in dopaminergic degeneration. Eur J Neurosci 18(6):1387–1401
Hancock DB, Martin ER, Mayhew GM, Stajich JM, Jewett R, Stacy MA, Scott BL, Vance JM et al (2008) Pesticide exposure and risk of Parkinson’s disease: a family-based case-control study. BMC Neurol 8(1):6. https://doi.org/10.1186/1471-2377-8-6
Collier TJ, Kanaan NM, Kordower JH (2011) Ageing as a primary risk factor for Parkinson’s disease: evidence from studies of non-human primates. Nat Rev Neurosci 12(6):359–366. https://doi.org/10.1038/nrn3039
Yong-Kee CJ, Sidorova E, Hanif A, Perera G, Nash JE (2012) Mitochondrial dysfunction precedes other sub-cellular abnormalities in an in vitro model linked with cell death in Parkinson's disease. Neurotox Res 21(2):185–194. https://doi.org/10.1007/s12640-011-9259-6
Munch G, Gasic-Milenkovic J, Dukic-Stefanovic S, Kuhla B, Heinrich K, Riederer P, Huttunen HJ, Founds H et al (2003) Microglial activation induces cell death, inhibits neurite outgrowth and causes neurite retraction of differentiated neuroblastoma cells. Exp Brain Res 150(1):1–8. https://doi.org/10.1007/s00221-003-1389-5
Attwell D, Laughlin SB (2001) An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab 21(10):1133–1145. https://doi.org/10.1097/00004647-200110000-00001
Dias V, Junn E, Mouradian MM (2013) The role of oxidative stress in Parkinson’s disease. J Parkinsons Dis 3(4):461–491. https://doi.org/10.3233/JPD-130230
Hwang O (2013) Role of oxidative stress in Parkinson’s disease. Exp Neurobiol 22(1):11–17. https://doi.org/10.5607/en.2013.22.1.11
Rahman K (2007) Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging 2(2):219–236
Floyd RA (1999) Antioxidants, oxidative stress, and degenerative neurological disorders. Proc Soc Exp Biol Med 222(3):236–245. https://doi.org/10.1046/j.1525-1373.1999.d01-140.x
Marshall KA, Reist M, Jenner P, Halliwell B (1999) The neuronal toxicity of sulfite plus peroxynitrite is enhanced by glutathione depletion: implications for Parkinson's disease. Free Radic Biol Med 27(5–6):515–520. https://doi.org/10.1016/S0891-5849(99)00094-5
Vila M, Ramonet D, Perier C (2008) Mitochondrial alterations in Parkinson’s disease: new clues. J Neurochem 107(2):317–328. https://doi.org/10.1111/j.1471-4159.2008.05604.x
Balaban RS, Nemoto S, Finkel T (2005) Mitochondria, oxidants, and aging. Cell 120(4):483–495. https://doi.org/10.1016/j.cell.2005.02.001
Navarro A, Boveris A (2007) The mitochondrial energy transduction system and the aging process. Am J Physiol Cell Physiol 292(2):C670–C686. https://doi.org/10.1152/ajpcell.00213.2006
Rohas LM, St-Pierre J, Uldry M, Jager S, Handschin C, Spiegelman BM (2007) A fundamental system of cellular energy homeostasis regulated by PGC-1alpha. Proc Natl Acad Sci U S A 104(19):7933–7938. https://doi.org/10.1073/pnas.0702683104
Kempuraj D, Selvakumar GP, Zaheer S, Thangavel R, Ahmed ME, Raikwar S, Govindarajan R, Iyer S et al (2017) Cross-talk between glia, neurons and mast cells in neuroinflammation associated with Parkinson’s disease. J NeuroImmune Pharmacol. https://doi.org/10.1007/s11481-017-9766-1
Kempuraj D, Thangavel R, Yang E, Pattani S, Zaheer S, Santillan DA, Santillan MK, Zaheer A (2015) Dopaminergic toxin 1-methyl-4-phenylpyridinium, proteins alpha-synuclein and glia maturation factor activate mast cells and release inflammatory mediators. PLoS One 10(8):e0135776. https://doi.org/10.1371/journal.pone.0135776
Kanthasamy A, Jin H, Mehrotra S, Mishra R, Kanthasamy A, Rana A (2010) Novel cell death signaling pathways in neurotoxicity models of dopaminergic degeneration: relevance to oxidative stress and neuroinflammation in Parkinson’s disease. Neurotoxicology 31(5):555–561. https://doi.org/10.1016/j.neuro.2009.12.003
Zawada WM, Banninger GP, Thornton J, Marriott B, Cantu D, Rachubinski AL, Das M, Griffin WST et al (2011) Generation of reactive oxygen species in 1-methyl-4-phenylpyridinium (MPP+) treated dopaminergic neurons occurs as an NADPH oxidase-dependent two-wave cascade. J Neuroinflammation 8(1):1–13. https://doi.org/10.1186/1742-2094-8-129
Khan MM, Zaheer S, Thangavel R, Patel M, Kempuraj D, Zaheer A (2015) Absence of glia maturation factor protects dopaminergic neurons and improves motor behavior in mouse model of parkinsonism. Neurochem Res 40(5):980–990. https://doi.org/10.1007/s11064-015-1553-x
Zaheer A, Yang B, Cao X, Lim R (2004) Decreased copper-zinc superoxide dismutase activity and increased resistance to oxidative stress in glia maturation factor-null astrocytes. Neurochem Res 29(8):1473–1480. https://doi.org/10.1023/B:NERE.0000029558.82943.00
Hastings TG (2009) The role of dopamine oxidation in mitochondrial dysfunction: implications for Parkinson’s disease. J Bioenerg Biomembr 41(6):469–472. https://doi.org/10.1007/s10863-009-9257-z
Zhu L, Wang Q, Zhang L, Fang Z, Zhao F, Lv Z, Gu Z, Zhang J, Wang J, Zen K, Xiang Y, Wang D, Zhang C-Y (2010) Hypoxia induces PGC-1[alpha] expression and mitochondrial biogenesis in the myocardium of TOF patientsCell Res 20 (6):676-687. doi:http://www.nature.com/cr/journal/v20/n6/suppinfo/cr201046s1.html
Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, Troy A, Cinti S et al (1999) Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 98(1):115–124. https://doi.org/10.1016/S0092-8674(00)80611-X
Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM (1998) A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92(6):829–839. https://doi.org/10.1016/S0092-8674(00)81410-5
Zheng B, Liao Z, Locascio JJ, Lesniak KA, Roderick SS, Watt ML, Eklund AC, Zhang-James Y, Kim PD, Hauser MA, Grunblatt E, Moran LB, Mandel SA, Riederer P, Miller RM, Federoff HJ, Wullner U, Papapetropoulos S, Youdim MB, Cantuti-Castelvetri I, Young AB, Vance JM, Davis RL, Hedreen JC, Adler CH, Beach TG, Graeber MB, Middleton FA, Rochet JC, Scherzer CR, Global PDGEC (2010) PGC-1alpha, a potential therapeutic target for early intervention in Parkinson’s disease Sci Transl Med 2 (52):52ra73. doi:https://doi.org/10.1126/scitranslmed.3001059
Ma D, Li S, Lucas EK, Cowell RM, Lin JD (2010) Neuronal inactivation of peroxisome proliferator-activated receptor gamma coactivator 1alpha (PGC-1alpha) protects mice from diet-induced obesity and leads to degenerative lesions. J Biol Chem 285(50):39087–39095. https://doi.org/10.1074/jbc.M110.151688
Cui L, Jeong H, Borovecki F, Parkhurst CN, Tanese N, Krainc D (2006) Transcriptional repression of PGC-1alpha by mutant huntingtin leads to mitochondrial dysfunction and neurodegeneration. Cell 127(1):59–69. https://doi.org/10.1016/j.cell.2006.09.015
Tsunemi T, La Spada AR (2012) PGC-1alpha at the intersection of bioenergetics regulation and neuron function: from Huntington’s disease to Parkinson’s disease and beyond. Prog Neurobiol 97(2):142–151. https://doi.org/10.1016/j.pneurobio.2011.10.004
Jiang H, Kang S-U, Zhang S, Karuppagounder S, Xu J, Lee Y-K, Kang B-G, Lee Y, Zhang J, Pletnikova O, Troncoso JC, Pirooznia S, Andrabi SA, Dawson VL, Dawson TM (2016) Adult conditional knockout of PGC-1α leads to loss of dopamine neurons. eneuro 3 (4). doi:https://doi.org/10.1523/eneuro.0183-16.2016
Nijland PG, Witte ME, van het Hof B, van der Pol S, Bauer J, Lassmann H, van der Valk P, de Vries HE et al (2014) Astroglial PGC-1alpha increases mitochondrial antioxidant capacity and suppresses inflammation: implications for multiple sclerosis. Acta Neuropathol Commun 2(1):170. https://doi.org/10.1186/s40478-014-0170-2
Shin JH, Ko HS, Kang H, Lee Y, Lee YI, Pletinkova O, Troconso JC, Dawson VL et al (2011) PARIS (ZNF746) repression of PGC-1alpha contributes to neurodegeneration in Parkinson’s disease. Cell 144(5):689–702. https://doi.org/10.1016/j.cell.2011.02.010
Stevens DA, Lee Y, Kang HC, Lee BD, Lee YI, Bower A, Jiang H, Kang SU et al (2015) Parkin loss leads to PARIS-dependent declines in mitochondrial mass and respiration. Proc Natl Acad Sci U S A 112(37):11696–11701. https://doi.org/10.1073/pnas.1500624112
Lopert P, Day BJ, Patel M (2012) Thioredoxin reductase deficiency potentiates oxidative stress, mitochondrial dysfunction and cell death in dopaminergic cells. PLoS One 7(11):e50683. https://doi.org/10.1371/journal.pone.0050683
Lopert P, Patel M (2014) Nicotinamide nucleotide transhydrogenase (Nnt) links the substrate requirement in brain mitochondria for hydrogen peroxide removal to the thioredoxin/peroxiredoxin (Trx/Prx) system. J Biol Chem 289(22):15611–15620. https://doi.org/10.1074/jbc.M113.533653
Mosley RL, Benner EJ, Kadiu I, Thomas M, Boska MD, Hasan K, Laurie C, Gendelman HE (2006) Neuroinflammation, oxidative stress and the pathogenesis of Parkinson’s disease. Clin Neurosci Res 6(5):261–281. https://doi.org/10.1016/j.cnr.2006.09.006
Viswanath V, Wu Y, Boonplueang R, Chen S, Stevenson FF, Yantiri F, Yang L, Beal MF et al (2001) Caspase-9 activation results in downstream caspase-8 activation and bid cleavage in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson's disease. J Neurosci 21(24):9519–9528
Barzilai A, Daily D, Zilkha-Falb R, Ziv I, Offen D, Melamed E, Shirvan A (2003) The molecular mechanisms of dopamine toxicity. Adv Neurol 91:73–82
Luo X, Budihardjo I, Zou H, Slaughter C, Wang X (1998) Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94(4):481–490
Kaimori JY, Takenaka M, Nakajima H, Hamano T, Horio M, Sugaya T, Ito T, Hori M et al (2003) Induction of glia maturation factor-beta in proximal tubular cells leads to vulnerability to oxidative injury through the p38 pathway and changes in antioxidant enzyme activities. J Biol Chem 278(35):33519–33527. https://doi.org/10.1074/jbc.M301552200
Offen D, Ziv I, Barzilai A, Gorodin S, Glater E, Hochman A, Melamed E (1997) Dopamine-melanin induces apoptosis in PC12 cells; possible implications for the etiology of Parkinson's disease. Neurochem Int 31(2):207–216
Lim R, Zaheer A, Kraakevik JA, Darby CJ, Oberley LW (1998) Overexpression of glia maturation factor in C6 cells promotes differentiation and activates superoxide dismutase. Neurochem Res 23(11):1445–1451. https://doi.org/10.1023/A:1020715126326
Martinou JC, Green DR (2001) Breaking the mitochondrial barrier. Nat Rev Mol Cell Biol 2(1):63–67. https://doi.org/10.1038/35048069
Antonsson B, Martinou JC (2000) The Bcl-2 protein family. Exp Cell Res 256(1):50–57. https://doi.org/10.1006/excr.2000.4839
Ullah I, Ullah N, Naseer MI, Lee HY, Kim MO (2012) Neuroprotection with metformin and thymoquinone against ethanol-induced apoptotic neurodegeneration in prenatal rat cortical neurons. BMC Neurosci 13(1):11. https://doi.org/10.1186/1471-2202-13-11
Gao L, Zhou W, Symmes B, Freed CR (2016) Re-cloning the N27 dopamine cell line to improve a cell culture model of Parkinson’s disease. PLoS One 11(8):e0160847. https://doi.org/10.1371/journal.pone.0160847
Ziegler U, Groscurth P (2004) Morphological features of cell death. News Physiol Sci 19:124–128
Acknowledgements
This work was supported by Veterans Affairs Merit Award I01BX002477 and National Institutes of Health Grants NS073670 and AG048205 to AZ. We would like to thank Dr. Alexander Jurkevich, Associate Director of Molecular Cytology core, University of Missouri, Columbia, MO, for his help in preparation and validation of confocal images.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Selvakumar, G.P., Iyer, S.S., Kempuraj, D. et al. Glia Maturation Factor Dependent Inhibition of Mitochondrial PGC-1α Triggers Oxidative Stress-Mediated Apoptosis in N27 Rat Dopaminergic Neuronal Cells. Mol Neurobiol 55, 7132–7152 (2018). https://doi.org/10.1007/s12035-018-0882-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-0882-6