Abstract
Reactive astrogliosis is a cellular manifestation of neuroinflammation and occurs in response to all forms and severities of the central nervous system (CNS)’s injury and disease. Both astroglial proliferation and antiapoptotic processes are aspects of astrogliosis. However, the underlying mechanism of this response remains poorly understood. In addition, little is known about why activated astrocytes are more resistant to stress and inflammation. CCAAT/enhancer binding protein delta (CEBPD) is a transcription factor found in activated astrocytes that surround β-amyloid plaques. In this study, we found that astrocytes activation was attenuated in the cortex and hippocampus of APPswe/PS1 E9 (AppTg)/Cebpd −/−mice. Furthermore, an increase in apoptotic astrocytes was observed in AppTg/Cebpd −/−mice, suggesting that CEBPD plays a functional role in enhancing the antiapoptotic ability of astrocytes. We found that Zinc Finger Protein 179 (ZNF179) was a CEBPD-regulated gene that played an antiapoptotic, but not proliferative, role in astrocytes. The transcriptions of the proapoptotic genes, insulin-like growth factor binding protein 3 (IGFBP3) and BCL2-interacting killer (BIK), were suppressed by ZNF179 via its interaction with the promyelocytic leukemia zinc finger (PLZF) protein in astrocytes. This study provides the first evidence that ZNF179, PLZF, IGFBP3, and BIK contributed to the novel CEBPD-induced antiapoptotic feature of astrocytes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aging is accompanied by a low-grade chronic neuroinflammation and aged rat neurons show signs of programmed cell death [1, 2]. Astrocyte is the most abundant cell type of glial cells and suggests being involved in the induction of neuroinflammation. Previous studies reported no change in the number of astrocytes in the hippocampus of male aged mice [2] and activated astrocytes are more resistant to death signals in highly inflammatory environments [3, 4]. Interestingly, the number of astrocytes increases by approximately 20 % in aging brains [5], suggesting that reactive gliosis responds to injured or damaged neurons during aging. The above discoveries indicated that the antiapoptosis of aged astrocytes contributes to the death of neurons. However, the underlying mechanisms have not been investigated. Alzheimer’s disease (AD) is pathologically characterized by the age-dependent deposition of β-amyloid (Aβ) in senile plaques and is associated with neuroinflammation. Aβ can activate astrocytes, thus promoting inflammatory cytokines production, which is believed to modulate the development and/or progression of AD. Astrogliosis (also known as astrocytosis or reactive astrocytes) is characterized by the proliferation and hypertrophy of astrocytes, and is usually observed in neurodegenerative disorders and central nervous system (CNS) injuries [6].
CCAAT/enhancer binding protein delta (CEBPD) is a member of the CCAAT/enhancer binding protein (C/EBP) family and has been shown to be activated in many inflammatory diseases, including AD [7, 8] and rheumatoid arthritis [9]. CEBPD is responsive to tumor necrosis factor alpha (TNFα), interleukin-1 beta (IL-1β), interleukin 6 (IL-6), lipopolysaccharide, and interferon gamma [8, 10]. In astrocytes, CEBPD activation attenuates macrophage-mediated phagocytosis of damaged neurons and promotes chemoattraction and migration of microglia/macrophages through PTX3 and MCP-1, respectively [8, 11]. However, the function of CEBPD in astrocytes and neuroinflammation-related diseases, especially its potential role in astrogliosis, remains largely uninvestigated.
Zinc finger protein 179 (ZNF179 or RNF112) belongs to the RING finger protein family, which is characterized by a zinc binding domain that serves as a potent protein binding interface. Mouse ZNF179 (Znf179) is specifically expressed in the mouse brain [12, 13]. Znf179 is expressed exclusively in the mouse brain and the expression is high in MAP2-positive cells and moderate in glial fibrillary acidic protein (GFAP)-positive cells in normal subjects [14]. However, the ZNF179 biology in brain remains largely unclear. A recent study showed that Znf179 regulates cell cycle exit, which is critical for neuronal differentiation and plays a potent role in cell survival [12].
Herein, we found that the loss of GFAP signal associated with the increase of caspase 3 signal was observed in AppTg/Cebpd −/− mice. Focusing on the survival (or resistance to stress-induced apoptosis) of astrocytes in inflammatory environment, we first showed that CEBPD played a functional role in inducing the resistance of cell death in an inflammatory environment. ZNF179 is attenuated following the IL-1β stimulation in neuronal cells. We further found that ZNF179 is responsive to CEBPD induction in astrocytes and contributes the CEBPD-induced antiapoptosis. Two pro-apoptotic genes insulin-like growth factor binding protein 3 (IGFBP3) and BCL2-interacting killer (BIK) were identified by a system approach and were negatively regulated by the complex of ZNF179 and promyelocytic leukemia zinc finger (PLZF). These discoveries provide a new insight into the resistance of astrocytes to inflammation and the control of astrogliosis.
Materials and Methods
Materials
The CEBPD and green fluorescent protein (GFP) antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The GFAP antibody was purchased from Invitrogen (Carlsbad, CA, USA). The caspase 3 antibody was purchased from Cell Signaling Technology (Danvers, MA, USA). The ZNF179 antibody used for immunofluorescence was purchased from GeneTex (Irvine, CA, USA), and the ZNF179 antibody used in the Western blot and immunoprecipitation assays was obtained from Dr. Yi-Chao Lee. The TRIzol RNA extraction reagent, Dulbecco’s modified Eagle’s medium (DMEM), and SuperScript™ III were purchased from Invitrogen (Carlsbad, CA, USA). All oligonucleotides were synthesized by MDBio Inc. (Taipei, Taiwan). Fetal bovine serum (FBS) was purchased from HyClone Laboratories (Logan, UT, USA).
Animals
The APPswe/PS1/E9 bigenic (AppTg) mice were obtained from the Jackson Laboratory (stock no. 004462, Bar Harbor, ME, USA). The AppTg mice were crossed with Cebpd-deficient mice (Cebpd −/−), a kind gift from Dr. E. Sterneck [15], on the C57BL/6 genetic background. Female mice heterozygous for AppTg mice was intercrossed with Cebpd −/− homozygous mice; the offspring (AppTg+/−/Cebpd +/−) were then bred to each other to produce the AppTg/Cebpd −/− mice in this study.
Cell Culture and Isolation of Primary Mouse Astrocytes
Human U373MG cells, an established cell line derived from human astrocytoma and HeLa cells (human cervical epithelioid carcinoma cell line), were cultured in DMEM. SH-SY5Y cells (human neuroblastoma cell line) were maintained in DMEM/F12. All media contained 10 % FBS, 100 μg/mL streptomycin, and 100 units/ml penicillin. U373MG cell lines stably expressing hemagglutinin (HA), HA-ZNF179, or HA-CEBPD were selected and maintained by regular media containing G418. The primary mouse brain astrocytes were isolated from Cebpd +/+ or Cebpd −/− mice using mechanical dissociation of the brain cortex from newborn pups. The isolated cells were then filtered through a 70-μm nylon strainer and cultured in the previously described medium [16] with the addition of poly-l-lysine (Invitrogen, Carlsbad, CA, USA).
Cell Survival, Proliferation, and Fluorescence-Activated Cell Sorting Analysis
For the cell survival assay, cells were plated and cultured in the aforementioned regular medium for 16 h. The experimental cells were then treated with or without 100 μg/mL methanesulfonate (MMS; Sigma, St. Louis, MO, USA) or 5 ng/mL IL-1β (Invitrogen, Carlsbad, CA, USA). Next, the media was removed and replaced with diluted 3-(4,5-cimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) reagent for 4 h. The samples were then measured spectrophotometrically at 595 nm using an ELISA plate reader. For cell proliferation assay, daily cell culture samples were counted in a Neubauer chamber after trypsinization. Viability was assessed by Trypan blue exclusion. Assays were performed in triplicates. For the apoptosis assay, cells were plated and cultured in the aforementioned regular medium for 16 h. The experimental cells were then treated with or without MMS or IL-1β for 24 h. As indicated times or experimental conditions, the cells were fixed in cold 70 % ethanol at −20 °C overnight and then suspended in phosphate-buffered saline (PBS) containing 0.2 % Triton X-100, 0.1 mg/ml RNase and 40 μg/mL propidium iodide at room temperature for 1 h. The apoptosis of experimental cells was analyzed using flow cytometry (FACSCalibur; BD Biosciences, Mountain View, CA).
Reverse Transcription-PCR and Quantitative PCR
Total RNA was extracted using the TRIsure RNA extraction reagent. The synthesis of complementary DNA (cDNA) was completed with an reverse transcription (RT) reaction using SuperScript III. Quantitative PCR (Q-PCR) was conducted using KAPA SYBR FAST qPCR Master Mix (Life Technologies Corporation and Kapa Biosystems Inc.). PCR was conducted using a CFX connect real-time PCR system (Bio-Rad) with the following pairs of specific primers: primer sequences human CEBPD (S): 5-′GCCATGTACGACGACGAGAG-3′ and CEBPD (AS): 5-′TGTGATTGCTGTTGAAGAGGTC-3′; mouse Cebpd (S): 5-′CTCCCGCACACAACATACTG -3′ and Cebpd (AS): 5-′AGTCATGCTTTCCCGTGTTC-3′, ZNF179 (S): 5-′GAGCAGGGAAACAAGGATCA-3′ and ZNF179 (AS): 5-′GGTGGGATGAGTCACGATCA-3′, RYBP (S): 5-′TGACATTGCAGTGGTGGTTT-3′ and RYBP (AS): 5-′CCATGTCAGGACTGGATGTG-3′, BIK (S): 5-′CCTGGACCCTATGGAGGACT-3′ and BIK (AS): 5-′GGTGAAACCGTCCATGAAAC-3′, GADD45B (S): 5-′ACCTGCATTGTCTCCTGGTC-3′ and GADD45B (AS): 5-′TTTGTTTGTGGCAGCAACTC-3′, IGFBP3 (S): 5-′AGGGCACTCTGGGAACCTAT-3′ and IGFBP3 (AS): 5-′TGCAGTCATCCGAAGAATTG′.
Western Blot Analysis
Cells were harvested and lysed with modified radioimmunoprecipitation assay (RIPA) buffer [50 mM Tris–HCl (pH 7.4), 150 mM sodium chloride, 1 mM ethylenediamine tetraacetic acid, 1 % NP40, 0.25 % sodium deoxycholate, 1 mM dithiothreitol, 10 mM NaF, 1 mM PMSF, 1 μg/mL aprotinin, and 1 μg/mL leupeptin]. Lysates were resolved on a sodium dodecyl sulfate-containing 10 % polyacrylamide gel, then transferred to a polyvinylidene difluoride nylon membrane and probed with primary antibodies for target proteins at 4 °C overnight. The specific proteins were detected by peroxidase-conjugated secondary antibody incubated at room temperature for 1 h. The signals were revealed by an enhanced chemiluminescence Western blot system from Pierce (Rockford, IL, USA).
Luciferase Reporter Assay
The 5′ flanking regions of ZNF179, IGFBP3 and BIK genes were obtained by PCR with U373MG genomic DNA and then individually cloned into a pGL3 basic vector. The primers for the PCR of the genomic DNA were: ZNF179 (S): 5′-KpnI-GGGGTACCCCGCGCCAAGCCTATCACATATCC-3′, ZNF179 (AS): 5′-HindIII-CCCAAGCTTGGGCTGCGGTAGGTAGAAGGTGAGG-3′; IGFBP3(S): 5′-MluI-CGACGCGTCGAGTGCTACACTAACCAGTGGTC-3′, IGFBP3 (AS): 5′-HindIII-CCCAAGCTTGGGAATCCAGGCAGGAAGCGGCTGATC-3′, BIK(S):5′-NheI-CTAGCTAGCTAGCTCTTCCTCCTTTTGATCAGC-3′, and BIK (AS):5′-BglII-GAAGATCTTCTAAAACCTGGGCACGGCTC-3′. For the reporter assay, cells were transfected with the reporters and expression vectors as indicted using polyJet (SignaGen, Ijamsville, MD). The lysates of transfected cells were harvested following the manufacturer’s instructions for the luciferase assay.
Co-Immunoprecipitation Assay
The lysates of U373MG cells were prepared using an immunoprecipitation lysis buffer [50 mM NaCl, 0.5 % NP-40, 10 mM Tris–HCl, (pH 8.0)]. The supernatant was collected and incubated with anti-GFP antibody at 4 °C for at least 4 h. Protein-A/G agarose beads were added to the lysates and the mixtures were incubated and rotated at 4 °C for 1 h. The beads were collected using centrifugation and washed three times with modified RIPA buffer. The proteins bound to the beads were eluted by adding 2X electrophoresis sample buffer and then subjected to Western blot analysis.
Lentiviral Short Hairpin RNA Knockdown
The virus was produced from Phoenix cells using a co-transfection of the various short hairpin RNA (shRNA) expression vectors in combination with pMD2.G and psPAX2 expression vectors. The expression vectors were obtained from the National RNAi Core Facility located at the Genomic Research Center of Institute of Molecular Biology, Academia Sinica, Taiwan. After determining the viral infection efficiency, the lentivirus containing shβ-galactosidase (shLacZ), shCEBPD, or shZNF179 were used to infect U373MG cells for 48 h. The shRNA sequences in the lentiviral expression vectors were shLacZ, 5′-CCGGTGTTCGCATTATCCGAACCATCTCGAGATGGTTCGGATAATGCGAACATTTTTG-3′, shCEBPD, 5′-CCGGGCCGACCTCTTCAACAGCAATCTCGAGATTGCTGTTGAAGAGGTCGGCTTTTT-3′ and shZNF179, 5′-CCGGCTTCATGGACTCCTACACGATCTCGAGATCGTGTAGGAGTCCATGAAGTTTTTG-3′.
Chromatin Immunoprecipitation Assay
Briefly, U373MG cells were treated with 1 % formaldehyde for 15 min. The cross-linked chromatin was then prepared and sonicated to an average size of 500 bp. The DNA fragments were immunoprecipitated with specific antibodies recognizing CEBPD, ZNF179, PLZF, or control rabbit immunoglobulin G (IgG) at 4 °C for 12–16 h. After reversal of the cross-linking between proteins and genomic DNA, the precipitated DNA was amplified by PCR with primers related to the specific regions on the genomic loci of target genes. The primers included ZNF179-I forward, 5′-GGGCTCTGTACATAGTAGGTACTC-3′ and ZNF179-I reverse, 5′-GCCTCTACTGGGCCAGCTGAGGTC-3′ or ZNF179-II forward, 5′-CTTGGTGCACCCATCTTTGCATC-3′ and ZNF179-II reverse, 5′-AGGCATTGCTATGATCTGGGTGAG-3′. IGFBP3 forward, 5′-CACGTGAGAGTCTTCTTGCGTTGAG-3′ and IGFBP3 reverse, 5′-ATACAGCGCTCCGCATTCGTGTG-3′, or BIK forward, 5′-CCCCTGGCTTCGGGTATGGATCAC-3′ and BIK reverse, 5′-TATCGGGGGGATCGATCCATTGAC-3′.
Immunofluorescence Analysis
The frozen male mouse brain sections (~15–16 months of age) were treated with protein blocker/antibody diluents (Bio SB, Santa Barbara, CA, USA) for 1 h. In the same buffer solution, the sections were incubated overnight with primary antibodies at 4 °C. These primary antibodies included cleaved caspase 3, GFAP, Aβ, Znf179, and CEBPD. Statistical analysis of immunofluorescent staining intensity in cortex or hippocampus of brain slices from AppTg or AppTg/Cebpd−/− mice was done using TissueQuest 4.0 image software. For the staining of cell cultures, U373MG cells were post-fixed in 4 % paraformaldehyde in PBS for 20 min, followed by 70 % methanol in PBS at −20 °C for 10 min. The fixed U373MG cells were further incubated with primary antibodies against target proteins in 3 % BSA at 4 °C. Pretreated slides of the tissue sections or U373MG cells were washed with 0.2 % Triton X-100 in PBS and then incubated with Alexa 488- or 555-conjugated secondary antibodies for 1 h at room temperature then washed again with 0.2 % Triton X-100 in PBS. Next, the glass slides were counter-stained and mounted with ProLong Gold antifade reagent with 4′,6-diamidino-2-phenylindole for immunofluorescence microscopy.
Statistical Analysis
All experiments were repeated at least three times, and data were analyzed for statistical significance by two-tailed unpaired Student’s t test using Prism 5 software. The data were expressed as means ± SEM. Differences were considered statistically significant when indicated by asterisks.
Results
Astrocyte Activation is Attenuated in the Area Surrounding β-Amyloid Plaques in AppTg/Cebpd −/− Mice
Activated astrocytes are largely resistant to cell death in a neuroinflammatory environment. In AD patients and AppTg mice, the expression of CEBPD and mouse CEBPD (Cebpd), respectively, is elevated and localized within astrocytes [7, 8] (Fig. S1A). The immunoreactivity for GFAP, a specific astrocyte marker, was attenuated in both the cortex and hippocampus of AppTg/Cebpd −/− mice when compared with AppTg mice (Fig. 1a, b). To assess whether apoptosis contributes to the reduction of astrocytes in AppTg/Cebpd −/− mice, activated caspase 3, an apoptosis marker, was used to measure the death of astrocytes in brain sections from AppTg and AppTg/Cebpd −/− mice. As shown in Fig. 1c, d, activated caspase 3 was upregulated and colocalized with the GFAP-positive astrocytes in AppTg/Cebpd −/− mice. This suggests that CEBPD contributes to the survival of astrocytes in AppTg mice.
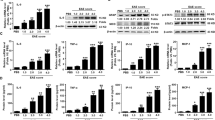
Astrocyte activation was decreased in AppTg/Cebpd −/− mice. a Astrocyte numbers were decreased in the area surrounding β-amyloid plaques in AppTg/Cebpd −/− mice. The brain tissue was subjected to immunofluorescence with anti-GFAP and anti-β-amyloid. b Quantitative analysis of GFAP staining in cortex and hippocampus brain sections of AppTg and AppTg/Cebpd −/− mice using TissueQuest software. c Loss of CEBPD promotes astrocyte death in AppTg/Cebpd −/− mice. The brain tissue was subjected to immunofluorescence with anti-GFAP and anti-cleaved caspase-3 (cCap3). d Quantitative analysis of GFAP and cCap3 immunofluorescent signal in whole brain sections of AppTg and AppTg/Cebpd −/− mice using TissueQuest software. (**p < 0.01, Student’s t test). cCap3 cleaved caspase-3; GFAP glial fibrillary acidic protein
CEBPD Plays an Antiapoptotic Role in Astrocytes
Several reports have suggested that astrocytes are more resistant to death in an inflammatory environment when compared to neurons [3, 17, 18]. CEBPD is known to be activated in astrocytes in response to IL-1β and TNFα [8, 11]. We first tested whether the activated CEBPD contributes to the antiapoptosis of astrocytes. After 24 h of IL-1β treatment, CEBPD indeed responded to IL-1β stimulation and the level of apoptosis in U373MG and primary mouse astrocytes was not significantly different from that before treatment (Fig. 2a, b). In contrast to astrocytes, we found decreased CEBPD in and increased death of neuronal SH-SY5Y cells after 24 h of IL-1β treatment (Fig. S1B). To assess if CEBPD plays an antiapoptotic role in astrocytes, we generated U373MG cells that stably expressed CEBPD. As shown in Fig. 2c, the overexpression of CEBPD in U373MG cells increased survival and reduced apoptosis after exposure to MMS, a strong apoptosis inducer. Importantly, in Cebpd +/+ and Cebpd −/− primary astrocytes, the cells lacking CEBPD showed increased cell death after IL-1β treatment (Fig. 2d). These data suggest that increased CEBPD in astrocytes has a functional role in antiapoptosis
CEBPD promotes astrocyte survival and activation after exposure to inflammatory cytokines. a Primary mouse astrocyte and b U373MG cells are resistant to IL-1β-induced cell death. The dead cells were stained with PI and were localized to the sub-G1 population using flow cytometry. RT-PCR and Western blot analyses were conducted with specific primers and the indicated antibodies using total RNA and protein lysates harvested from IL-1β-treated cells. c CEBPD protects U373MG cells against MMS-induced death. The MTT assay and PI staining were conducted as indicated. d Loss of CEBPD in primary astrocyte cultures increased cell death after IL-1β treatment. The PI staining was conducted as indicated. (*p < 0.05, **p < 0.01, Student’s t test). CD CEBPD; n.s. not significant
ZNF179 Gene is a Direct Target of CEBPD
We found that the expression of ZNF179 was responsive to IL-1β treatment in astrocytes (Fig. 3a). Furthermore, the expression of ZNF179 paralleled that of CEBPD in astrocytes (Fig. S1C). We next sought to determine the relationship between ZNF179 and CEBPD. A reporter assay showed that Cebpd could activate a Znf179 promoter-driven reporter, but the expressed Znf179 had no effect on a Cebpd promoter-driven reporter (Fig. S2). These data suggest that CEBPD is an upstream regulator of the ZNF179 gene in astrocytes. Moreover, the effect of IL-1β-induced Znf179 transcription was attenuated in primary astrocyte cultures derived from Cebpd −/− mice (Fig. 3b). A similar result was found for CEBPD-related ZNF179 transcription and expression in U373MG cells (Fig. 3c). Using a serial deletion reporter assay, we identified a potent CEBPD responsive region in the ZNF179 promoter at −282/+72 bp (Fig. 3d). In addition, an in vivo DNA binding assay showed that the binding of CEBPD on the ZNF179 promoter was responsive to IL-1β in U373MG cells (Fig. 3e). These data suggest that CEBPD regulates ZNF179 transcription by directly binding to the ZNF179 promoter region.
CEBPD directly regulates ZNF179 transcription in U373MG cells. a Primary mouse astrocytes (left panel) and U373MG cells (right panel) were responsive to IL-1β treatment. RT-PCR and Western blot analyses were conducted with specific primers and the indicated antibodies using total RNA and protein lysates harvested from IL-1β-treated cells. b The lack of Cebpd did not induce Znf179 expression in primary astrocyte cultures. A Q-PCR assay was performed using total RNA harvested from Cebpd +/+ and Cebpd −/− primary astrocytes. c CEBPD participates in IL-1β-induced ZNF179 transcription. RT-PCR and Western blots were performed with total RNA and protein lysates harvested from stable U373MG cells with pMT-CEBPD expression vector (right panel) or IL-1β-treated U373MG cells with or without attenuation of CEBPD (left panel). d The identification of CEBPD responsive motifs on the ZNF179 promoter region. The representation of reporter constructs (upper panel). A reporter assay was conducted using the luciferase activity of the ZNF179 reporter/CEBPD expression vector co-transfected cell lysates. e CEBPD directly binds to the ZNF179 promoter in vivo. A chromatin immunoprecipitation assay was performed with the immunoprecipitation products at the indicated Abs from U373MG cells treated with IL-1β. (*p < 0.05, **p < 0.01, ***p < 0.001, Student’s t test). shCD short hairpin CEBPD; HA/CD HA-tagged CEBPD; n.s. not significant; IP immunoprecipitation; Abs antibodies
ZNF179 Expression in Astrocytes Contributes to Antiapoptosis
Although a previous study suggested that ZNF179 might contribute to the survival of neurons [12], the effect of ZNF179 in astrocytes is unknown. ZNF179 has been suggested to be a brain-specific gene [12, 19]. An immunofluorescence assay showed that ZNF179 was detectable in the GFAP-positive astrocytes of AppTg mice and was attenuated in the astrocytes of AppTg/Cebpd −/− mice (Fig. 4a). To determine if ZNF179 plays an antiapoptotic role, we established the stable expression of ZNF179 in human cervical HeLa cells. After MMS or TNFα exposure, HeLa cells with ZNF179 showed a resistance to MMS- or TNFα-induced apoptosis (Fig. S3A and S3B). A similar result was observed in U373MG cells stably expressing ZNF179 (Fig. 4b, c). A gain-of-function approach was used to assess if the lack of ZNF179 or CEBPD impacts the IL-1β-induced apoptosis of astrocytes. U373MG cells with reduced ZNF179 showed a loss of CEBPD and increased apoptosis after IL-1β exposure (Fig. 4d). Furthermore, the attenuation of ZNF179 decreased the CEBPD-related antiapoptotic effect after MMS exposure (Fig. 4e). These results suggest that ZNF179 is expressed in the astrocytes of AppTg mice, and it contributes to astrocytic CEBPD-dependent antiapoptotic effects.
ZNF179 attenuates the IL-1β/MMS-induced death of U373MG cells. a Znf179 is highly expressed in the astrocytes of AppTg mice when compared with AppTg/Cebpd −/− mice. Sagittal sections of brain cortex were prepared from AppTg and AppTg/Cebpd −/− mice and then subjected to immunofluorescence with anti-GFAP, and anti-Znf179 antibodies. b and c ZNF179 protects U373MG cells against MMS-induced death. The MTT assay and PI staining were conducted as indicated. d Loss of CEBPD or ZNF179 increased cell death after IL-1β treatment. The PI staining was conducted as indicated. e The knockdown of ZNF179 in CEBPD overexpressing cells increased cell death after MMS treatment. The PI staining was conducted as indicated (*p < 0.05, **p < 0.01, ***p < 0.001, Student’s t test). HZ HA-tagged ZNF179; shCD short hairpin CEBPD; shZN short hairpin ZNF179; GFAP glial fibrillary acidic protein; HA/CD HA-tagged CEBPD; shLacZ, shβ-galactosidase
The Identification of ZNF179-Regulated Genes in U373MG Cells
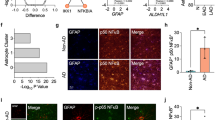
We demonstrated that ZNF179 plays a functional role in antiapoptosis. However, the ZNF179-responsive genes related to the antiapoptotic processes in astrocytes remain unknown. Genome-wide profiling and comparisons were conducted using U373MG cells with and without the stable expression of HA/ZNF179. As shown in Fig. 5a, we found that a total of 98 genes were significantly induced by ZNF179 and 339 genes were inhibited by ZNF179 in U373MG cells. Among these ZNF179-responsive genes, four proapoptotic genes, including RYBP [20], BIK [21], GADD45B [22] and IGFBP3 [23, 24], were downregulated by ZNF179. RT-PCR and Q-PCR assays confirmed the findings from the microarray profile (Fig. 5b).
The identification of ZNF179-regulated genes in astrocytes. a A microarray analysis was performed using total RNA harvested from stable U373MG cells with a pcDNA-HA-ZNF179 expression vector. b The verification of CEBPD responsive genes in antiapoptosis. RT-PCR and Q-PCR assays were conducted using total RNA harvested from HA and HZ U373MG cells. (*p < 0.05, **p < 0.01, Student’s t test). HZ HA-tagged ZNF179
ZNF179 Inhibits IGFBP3 and BIK Expression by Interacting with PLZF in Astrocytes
We next sought to determine if these ZNF179-regulated genes were also responsive to IL-1β. By Q-PCR analysis, we found that only the IGFBP3 and BIK transcripts out of the four ZNF179-responsive proapoptotic genes were downregulated in IL-1β-treated astrocytes (Fig. 6a). PLZF protein belongs to the family of Krüppel-like zinc finger proteins. Our recent study showed that ZNF179 can interact with PLZF in neurons [25]. Here, a Co-immunoprecipitation assay showed that ZNF179 also interacted with PLZF in astrocytes (Fig. 6b). Focusing on IGFBP3 and BIK, a serial deletion of IGFBP3 and BIK promoters were cloned into a pGL3 basic vector. We identified two putative PLZF binding motifs at −535/−543 and −452/−460 of the IGFBP3 promoter region and three motifs at −498/−488, −425/−416, and −376/−368 of the BIK promoter region (Fig. 6c, upper panel). Using a reporter assay, we found that overexpression of PLZF repressed the activities of IGFBP3 and BIK reporters containing potent PLZF binding sites. The co-transfection of PLZF and ZNF179 expression vectors showed an extensive effect on the repression of IGFBP3 and BIK reporter activity (Fig. 6c, bottom panel). Furthermore, the loss of ZNF179 reduced the PLZF-mediated repression of IGFBP3 and BIK reporter activity after IL-1β exposure (Fig. 6d). To verify if the IGFBP3 and BIK genes were directly regulated via the binding of PLZF and ZNF179, lysates of IL-1β-treated U373MG cells were subjected to a chromatin immunoprecipitation assay. Our results show that the binding of PLZF and ZNF179 to the IGFBP3 and BIK promoter was responsive to IL-1β in U373MG cells (Fig. 6e). These data suggest that PLZF is an important component of IL-1β-induced ZNF179-mediated suppression of IGFBP3 and BIK transcription.
ZNF179 downregulates IGFBP3 and BIK expression through interaction with PLZF. a IGFBP3 and BIK expression was downregulated after IL-1β treatment. The Q-PCR assay was performed using total RNA harvested from U373MG cells. b The interaction of ZNF179 and PLZF was revealed using immunoprecipitation analysis of an EGFP-PLZF plasmid transiently transfected together with HA or HA-ZNF179 plasmids. c The interaction of ZNF179 with PLZF can downregulate IGFBP3 and BIK promoter activity. A representation of reporter constructs are shown (upper panel). Using cell lysates, the reporter assay was conducted by examining the luciferase activity of a co-transfected IGFBP3 reporter or BIK reporter/ZNF179 expression vector/PLZF expression vector. d ZNF179 reduces IGFBP3 reporter or BIK reporter activities after IL-1β treatment in U373MG cells. Lentiviral vectors were used to express shLacZ and shZNF179. After a 2-day infection, these constructs were co-transfected with expression and promoter vectors. A reporter assay was conducted using the luciferase activity within cell lysates. e The interaction of ZNF179 with PLZF can directly binds to the IGFBP3 and BIK promoter in vivo. A chromatin immunoprecipitation assay was performed with the immunoprecipitation products at the indicated Abs from U373MG cells treated with IL-1β. (*p < 0.05, **p < 0.01, ***p < 0.001, Student’s t test). shZN short hairpin ZNF179; FL full length; PI promoter I; PII promoter II; shLacZ shβ-galactosidase
Discussion
In addition to AD, neuroinflammation has been suggested to play a critical role in the pathogenesis of several neurodegenerative disorders, including Parkinson’s disease and Huntington’s disease [26]. Activation of astrocytes and microglia has been associated with the pathogenesis of AD. However, the precise mechanisms involved in this process remain largely unclear. CEBPD is critical for the activation of astrocytes, but not for the microglia and neurons that surround Aβ plaques in AD (Fig. S1A) [7, 8, 11]. Under oxidative stress or inflammation, activated astrocytes were more resistant to apoptosis when compared with neurons (Fig. S1A and 2A) [3]. Our previous study showed that a significant reduction in astrocytes was observed in AppTg/Cebpd −/− mice [11]. In this study, we demonstrated that the loss of Cebpd promoted the death of GFAP-positive cells in the cortex and hippocampus of AppTg mice partly through the induction of apoptosis (Fig. 1c). Moreover, we demonstrated that the increase of CEBPD in astrocytes reduced MMS- or IL-1β-induced apoptosis (Fig. 2). These data suggest that CEBPD plays an antiapoptotic role in astrocytes, especially in a stress environment.
Elevated CEBPD expression has been mainly found in the astrocytes of AD patients and AppTg mice [7, 8, 11]. We determined that the activation of CEBPD in astrocytes results in attenuation of macrophage-mediated phagocytosis of damaged neurons, and also contributes to microglial activation and migration [8, 11]. In response to damaged or inflammatory environments, neurons are more sensitive than astrocytes because they have lower antioxidant capabilities and are therefore prone to excitotoxicity [27]. Our novel findings suggest that astrocytic CEBPD plays an antiapoptotic role, which contributes to the resistance of inflammation-induced apoptosis in the inflamed brain. Meanwhile, we also showed the first evidence to support ZNF179’s antiapoptotic role, particularly in astrocytes within a damaged or inflammatory environment.
CEBPD is induced in age-associated disorders, such as AD, atherosclerosis [28], type 2 diabetes [29], and rheumatoid arthritis [9, 30]. These discoveries imply that CEBPD may play a central role in inflammation-related diseases. Our previous [8, 11] and current studies indicate that the inactivation of CEBPD in astrocytes could improve the survival of neuron in inflammatory diseases. Small chemical drugs (<400 Da) may cross the blood–brain barrier via lipid-mediate-free diffusion [31]. Carnosic acid can penetrate the blood–brain barrier and could be a useful drug for Aβ-induced neurodegeneration as AD [32]. We previously demonstrated that rosmanol (molecular weight 346.41 Da), a natural carnosic acid, derived from the herb rosemary and inotilone (molecular weight 218.21 Da), an unusual 5-methyl-3(2H)-furanone derivative, can inhibit CEBPD activation in macrophages [9]. Therefore, rosmanol and inotilone can be tested toward the prevention or treatment of AD in the future.
Znf179 is specifically expressed in the brain and can contribute to neuronal differentiation and survival [12]. Unlike neurons, the increase of ZNF179 in astrocytes was responsive to inflammatory cytokines (Figs. 2, 3, and S1C). Here, we found that the transcription of ZNF179 gene is responsive to IL-1β through the action of enhanced binding and transactivation activity of CEBPD on its promoter region in astrocytes. Importantly, the antiapoptotic role of ZNF179 in astrocytes may explain why active astrocytes are resistant to cell death in an inflammatory environment. Interestingly, the GSK3β inhibitor, LiCl, enhanced neuron survival but caused growth retardation in astrocytes [33]. A study showed that CEBPD is a GSK3β substrate, and CEBPD phosphorylation contributes to the transcriptional regulation of CEBPD-mediated downstream target genes. Therefore, the relevance of GSK3β-mediated phosphorylation of CEBPD in ZNF179 transcriptional activation in astrocytes requires further investigation.
PLZF has been suggested to be a transcriptional repressor and antiapoptotic for neurons in stroke [34]. PLZF interacts with SMRT/N-CoR-mSin3A-HDAC-repressing complexes to suppress gene transcription [35, 36]. Several lines of evidence suggest that PLZF has antiapoptotic effects. For example, PLZF can inhibit caspase 3 and caspase 7 activity in neurons [34]; however, the specific mechanism remains unknown. In this study, we further found that ZNF179 expression was activated and interacted with PLZF upon IL-1β treatment to promote antiapoptosis in astrocytes. IGFBP3 is a common pro-apoptosis gene, and it has been shown to enhance and attenuate the actions of IGF-I [37]. IGF-I is mediated by the IGF-I receptor and interacts with IGFBP3 to inhibit cell growth and induce apoptosis [24]. Previous studies showed that IGFBP3 activates the apoptotic pathway through Bcl2 family [38]. Increased PLZF attenuated IGFBP3 transcription. A constitutive binding of PLZF and increased binding of ZNF179 on the IGFBP3 promoter were observed after IL-1β treatment. However, the nuclear-cytoplasmic shuttle of ZNF179 did not change in response to IL-1β stimulation (Fig. S4). This observation implies that cytosolic and nuclear ZNF179 may play distinct roles in IL-1β-treated astrocytes. These data also suggest that IL-1β can induce nuclear ZNF179 binding to the preexisting PLZF on IGFBP3 and BIK promoter to suppress its transcription. Although the transcription of the proapoptotic GADD45B and RYBP genes is repressed by the overexpression of ZNF179, our preliminary results show that exposure to IL-1β has no effect on GADD45B and RYBP transcription. These data suggest that ZNF179 can interact with other IL-1β-independent transcription repressors or complexes to repress gene transcription; however, this hypothesis needs to be further investigated
References
Franceschi C (2007) Inflammaging as a major characteristic of old people: can it be prevented or cured? Nutr Rev 65(12):S173–S176
Cerbai F, Lana D, Nosi D, Petkova-Kirova P, Zecchi S, Brothers HM, Wenk GL, Giovannini MG (2012) The neuron-astrocyte-microglia triad in normal brain ageing and in a model of neuroinflammation in the rat hippocampus. PLoS One 7(9):e45250
Garwood CJ, Pooler AM, Atherton J, Hanger DP, Noble W (2011) Astrocytes are important mediators of Abeta-induced neurotoxicity and tau phosphorylation in primary culture. Cell Death Dis 2:e167
Song JH, Bellail A, Tse MC, Yong VW, Hao C (2006) Human astrocytes are resistant to Fas ligand and tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis. J Neurosci 26(12):3299–3308
Salminen A, Ojala J, Kaarniranta K, Haapasalo A, Hiltunen M, Soininen H (2011) Astrocytes in the aging brain express characteristics of senescence-associated secretory phenotype. Eur J Neurosci 34(1):3–11
Herrmann JE, Imura T, Song BB, Qi JW, Ao Y, Nguyen TK, Korsak RA, Takeda K, Akira S, Sofroniew MV (2008) STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci 28(28):7231–7243
Li R, Strohmeyer R, Liang Z, Lue LF, Rogers J (2004) CCAAT/enhancer binding protein delta (C/EBPdelta) expression and elevation in Alzheimer’s disease. Neurobiol Aging 25(8):991–999
Ko CY, Chang LH, Lee YC, Sterneck E, Cheng CP, Chen SH, Huang AM, Tseng JT, Wang JM (2012) CCAAT/enhancer binding protein delta (CEBPD) elevating PTX3 expression inhibits macrophage-mediated phagocytosis of dying neuron cells. Neurobiol Aging 33(2):422 e411–425
Chang LH, Huang HS, Wu PT, Jou IM, Pan MH, Chang WC, Wang DD, Wang JM (2012) Role of macrophage CCAAT/enhancer binding protein delta in the pathogenesis of rheumatoid arthritis in collagen-induced arthritic mice. PLoS One 7(9):e45378
Cardinaux JR, Allaman I, Magistretti PJ (2000) Pro-inflammatory cytokines induce the transcription factors C/EBPbeta and C/EBPdelta in astrocytes. Glia 29(1):91–97
Ko CY, Wang WL, Wang SM, Chu YY, Chang WC, Wang JM (2014) Glycogen synthase kinase-3beta-mediated CCAAT/enhancer-binding protein delta phosphorylation in astrocytes promot es migration and activation of microglia/macrophages. Neurobiol Aging 35(1):24–34
Pao PC, Huang NK, Liu YW, Yeh SH, Lin ST, Hsieh CP, Huang AM, Huang HS, Tseng JT, Chang WC, Lee YC (2011) A novel RING finger protein, Znf179, modulates cell cycle exit and neuronal differentiation of P19 embryonal carcinoma cells. Cell Death Differ 18(11):1791–1804
Seki N, Hattori A, Muramatsu M, Saito T (1999) cDNA cloning of a human brain finger protein, BFP/ZNF179, a member of the RING finger protein family. DNA Res Int J Rapid Publ Rep Genes Genomes 6(5):353–356
Orimo A, Inoue S, Ikeda K, Sato M, Kato A, Tominaga N, Suzuki M, Noda T, Watanabe M, Muramatsu M (1998) Molecular cloning, localization, and developmental expression of mouse brain finger protein (Bfp)/ZNF179: distribution of bfp mRNA partially coincides with the affected areas of Smith-Magenis syndrome. Genomics 54(1):59–69
Sterneck E, Paylor R, Jackson-Lewis V, Libbey M, Przedborski S, Tessarollo L, Crawley JN, Johnson PF (1998) Selectively enhanced contextual fear conditioning in mice lacking the transcriptional regulator CCAAT/enhancer binding protein delta. Proc Natl Acad Sci U S A 95(18):10908–10913
Allan SM, Rothwell NJ (2003) Inflammation in central nervous system injury. Philos Trans R Soc Lond Ser B Biol Sci 358(1438):1669–1677
Saijo K, Winner B, Carson CT, Collier JG, Boyer L, Rosenfeld MG, Gage FH, Glass CK (2009) A Nurr1/CoREST pathway in microglia and astrocytes protects dopaminergic neurons from inflammation-induced death. Cell 137(1):47–59
Franke H, Sauer C, Rudolph C, Krugel U, Hengstler JG, Illes P (2009) P2 receptor-mediated stimulation of the PI3-K/Akt-pathway in vivo. Glia 57(10):1031–1045
Kimura T, Arakawa Y, Inoue S, Fukushima Y, Kondo I, Koyama K, Hosoi T, Orimo A, Muramatsu M, Nakamura Y, Abe T, Inazawa J (1997) The brain finger protein gene (ZNF179), a member of the RING finger family, maps within the Smith-Magenis syndrome region at 17p11.2. Am J Med Genet 69(3):320–324
Stanton SE, Blanck JK, Locker J, Schreiber-Agus N (2007) Rybp interacts with Hippi and enhances Hippi-mediated apoptosis. Apoptosis Int J Program Cell Death 12(12):2197–2206
Chinnadurai G, Vijayalingam S, Rashmi R (2008) BIK, the founding member of the BH3-only family proteins: mechanisms of cell death and role in cancer and pathogenic processes. Oncogene 27(Suppl 1):S20–S29
Ou DL, Shen YC, Yu SL, Chen KF, Yeh PY, Fan HH, Feng WC, Wang CT, Lin LI, Hsu C, Cheng AL (2010) Induction of DNA damage-inducible gene GADD45beta contributes to sorafenib-induced apoptosis in hepatocellular carcinoma cells. Cancer Res 70(22):9309–9318
Williams AC, Smartt H, AM HZ, Macfarlane M, Paraskeva C, Collard TJ (2007) Insulin-like growth factor binding protein 3 (IGFBP-3) potentiates TRAIL-induced apoptosis of human colorectal carcinoma cells through inhibition of NF-kappaB. Cell Death Differ 14(1):137–145
Kielczewski JL, Hu P, Shaw LC, Li Calzi S, Mames RN, Gardiner TA, McFarland E, Chan-Ling T, Grant MB (2011) Novel protective properties of IGFBP-3 result in enhanced pericyte ensheathment, reduced microglial activation, increased microglial apoptosis, and neuronal protection after ischemic retinal injury. Am J Pathol 178(4):1517–1528
Lin DY, Huang CC, Hsieh YT, Lin HC, Pao PC, Tsou JH, Lai CY, Hung LY, Wang JM, Chang WC, Lee YC (2013) Analysis of the interaction between zinc finger protein 179 (Znf179) and promyelocytic leukemia zinc finger (Plzf). J Biomed Sci 20:98–107
Hsiao HY, Chen YC, Chen HM, Tu PH, Chern Y (2013) A critical role of astrocyte-mediated nuclear factor-kappaB-dependent inflammation in Huntington’s disease. Hum Mol Genet 22(9):1826–1842
Swanson RA, Ying W, Kauppinen TM (2004) Astrocyte influences on ischemic neuronal death. Curr Mol Med 4(2):193–205
Takata Y, Kitami Y, Yang ZH, Nakamura M, Okura T, Hiwada K (2002) Vascular inflammation is negatively autoregulated by interaction between CCAAT/enhancer-binding protein-delta and peroxisome proliferator-activated receptor-gamma. Circ Res 91(5):427–433
Gao H, Bryzgalova G, Hedman E, Khan A, Efendic S, Gustafsson JA, Dahlman-Wright K (2006) Long-term administration of estradiol decreases expression of hepatic lipogenic genes and improves insulin sensitivity in ob/ob mice: a possible mechanism is through direct regulation of signal transducer and activator of transcription 3. Mol Endocrinol 20(6):1287–1299
Nishioka K, Ohshima S, Umeshita-Sasai M, Yamaguchi N, Mima T, Nomura S, Murata N, Shimizu M, Miyake T, Yoshizaki K, Suemura M, Kishimoto T, Saeki Y (2000) Enhanced expression and DNA binding activity of two CCAAT/enhancer-binding protein isoforms, C/EBPbeta and C/EBPdelta, in rheumatoid synovium. Arthritis Rheum 43(7):1591–1596
Pardridge WM (2012) Drug transport across the blood–brain barrier. J Cerebr Blood F Met 32(11):1959–1972
Azad N, Rasoolijazi H, Joghataie MT, Soleimani S (2011) Neuroprotective effects of carnosic acid in an experimental model of Alzheimer’s disease in rats. Cell J 13(1):39–44
Gilad GM, Gilad VH (2007) Astroglia growth retardation and increased microglia proliferation by lithium and ornithine decarboxylase inhibitor in rat cerebellar cultures: cytotoxicity by combined lithium and polyamine inhibition. J Neurosci Res 85(3):594–601
Seidel K, Kirsch S, Lucht K, Zaade D, Reinemund J, Schmitz J, Klare S, Li Y, Schefe JH, Schmerbach K, Goldin-Lang P, Zollmann FS, Thone-Reineke C, Unger T, Funke-Kaiser H (2011) The promyelocytic leukemia zinc finger (PLZF) protein exerts neuroprotective effects in neuronal cells and is dysregulated in experimental stroke. Brain Pathol 21(1):31–43
Parrado A, Robledo M, Moya-Quiles MR, Marin LA, Chomienne C, Padua RA, Alvarez-Lopez MR (2004) The promyelocytic leukemia zinc finger protein down-regulates apoptosis and expression of the proapoptotic BID protein in lymphocytes. Proc Natl Acad Sci U S A 101(7):1898–1903
Girard N, Tremblay M, Humbert M, Grondin B, Haman A, Labrecque J, Chen B, Chen Z, Chen SJ, Hoang T (2013) RARalpha-PLZF oncogene inhibits C/EBP alpha function in myeloid cells. Proc Natl Acad Sci U S A 110(33):13522–13527
Brosseau C, Pirianov G, Colston KW (2013) Role of insulin-like growth factor binding protein-3 in 1,25-dihydroxyvitamin-d3-induced breast cancer cell apoptosis. Int J Cell Biol 2013 ArticleID:960378
Firth SM, Baxter RC (2002) Cellular actions of the insulin-like growth factor binding proteins. Endocr Rev 23(6):824–854
Acknowledgments
This work was supported by grant NHRI-EX102-10143NI from the National Health Research Institutes, grant NSC100-2320-B-006-020 from the National Science Council, Ministry of Health and Welfare (MOHW103-TD-B-111-05), and the Headquarters of University Advancement at the National Cheng Kung University from the Ministry of Education.
Conflict of interest
None of the authors has a conflict of interest to declare in relation to the present research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yi-Chao Lee and Chiung-Yuan Ko contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PPTX 21772 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Wang, SM., Lee, YC., Ko, CY. et al. Increase of Zinc Finger Protein 179 in Response to CCAAT/Enhancer Binding Protein Delta Conferring an Antiapoptotic Effect in Astrocytes of Alzheimer’s Disease. Mol Neurobiol 51, 370–382 (2015). https://doi.org/10.1007/s12035-014-8714-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-8714-9