Abstract
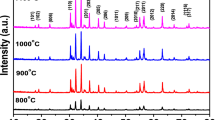
In the present investigation, the distribution of iron ions at octahedral and tetrahedral sites in BaFe12O19 prepared by employing four different synthesis techniques, namely, solid-state reaction, oxalate precursor route, sol–gel and wet chemical methods, have been examined using Mossbauer studies and compared with magnetization data. It was observed that the iron ions distribute in different preferential order at various sites for hexaferrites prepared using different synthesis methods, which is confirmed by Mossbauer spectroscopy. Prepared samples were characterized by X-ray diffraction, Fourier transform infrared spectroscopy, and Field emission scanning electron microscopy. Rietveld refinement of all samples revealed an M-type hexagonal structure confirming P63/mmc space group along with a minor peak belonging to the α-Fe2O3 phase, except for the sample synthesized by sol–gel route. A uniform spherical shape with a small grain size was observed in sol–gel prepared samples and the Williamson–Hall method was adopted to estimate the average crystallite size, which varies between 72 and 129 nm. The room temperature magnetization studies reveal that the sample synthesized via sol–gel route shows high coercivity and saturation magnetization values due to their smaller grain sizes. Mossbauer spectra of all BaFe12O19 samples were fitted with five sexets assigned to the hexagonal crystal structure of 4f2, 4f1, 2a, 12k and 2b sites, where the variation in their relative areas confirms the redistribution of iron ions at these sites.
Graphical abstract









Similar content being viewed by others
References
Kaplan T A and Menyuk N 2007 Philos. Mag. 87 3711
Perez-Juache T J, Guerrero A L, Cabal-Velarde J G, Mirabal-Garcia M, Palomares-Sanchez S A, Matutes-Aquino J A et al 2016 Phys. B 503 183
Kaur T, Kumar S, Hamid B and Basharat B 2015 Appl. Phys. A 119 1531
Lin H, Watanabe Y, Kimura M, Hanabusa K and Shirai H 2003 J. Appl. Polym. Sci. 87 1239
Mohsen Q 2010 J. Alloys Compd. 500 125
Mali A and Ataie A 2004 Ceram. Int. 30 1979
Lee W J and Fang T T 1995 J. Mater. Sci. 30 4349
Kaur P, Chawla S K, Meena S S, Yusuf S M and Narang S B 2016 Ceram. Int. 42 14475
Prathap S, Madhuri W and Meena S S 2021 Mater. Charact. 177 111168
Trukhanov A V, Kostishyn V G, Panina L V, Jabarov S H, Korovushkin V V, Truknanov S V et al 2017 Ceram. Int. 43 12822
Baykal A, Gungunes H, Sozeri H, Amir Md, Auwal I, Asiri S et al 2017 Ceram. Int. 43 15486
Pashkova E V, Solovyova E D, Kolodiazhnyi T V, Ivanitskii V P and Belous A G 2014 J. Magn. Magn. Mater. 368 1
Galvão S B, Lima A C, De Medeiros S N, Soares J M and Paskocimas C A 2014 Mater. Lett. 115 38
Qiu J, Wang Y and Gu M 2006 Mater. Lett. 60 2728
Chanda S, Bharadwaj S, Pavan Kumar N, Siva Kumar K V and Kalyana Lakshmi Y 2020 Appl. Phys. A 126 894
Chanda S, Bharadwaj S, Srinivas A, Siva Kumar K V and Kalyana Lakshmi Y 2021 J. Phys. Chem. Solids 155 110120
Rodríguez-Carvajal J 2001 CEA/Saclay (France) https://cdifx.univ-rennes1.fr/fps/fp_rennes.pdf
Maswadeh Yazan, Mahamood Sami H, Awadallah Ahmad and Aloqail Aynour N 2015 IOP Conf. Ser. Mater. Sci. Eng. 92 012019
Radwan M, Rashad M M and Hessien M M 2007 J. Mater. Process. Technol. 181 106
Jotania R B, Khomane R B, Chauhan C C, Menon S K and Kulkarni B D 2008 J. Magn. Magn. Mater. 320 1095
Kaur N and Kaur M 2014 Process. Appl. Ceram. 8 137
Rane M V, Bahadur D, Nigam A K and Srivastava C M 1999 J. Magn. Magn. Mater. 192 288
Singhal S, Namgyal T, Singh J, Chandra K and Bansal S 2011 Ceram. Int. 37 1833
Mallick K K 2007 J. Magn. Magn. Mater. 312 418
Mosleh Z, Kameli P, Ranjbar M and Salamati H J C I 2014 Ceram. Int. 40 7279
Xie T, Xu L and Liu C 2012 Powder Technol. 232 87
Wang Z, Xie Y, Wang P, Ma Y, Jin S and Liu X 2011 J. Magn. Magn. Mater. 323 3121
Liu Y, Li Y, Zhang H, Chen D and Wen Q 2010 J. Appl. Phys. 107 09A507
Batlle X, Obradors X, Rodriguez-Carvajal J, Pernet M, Cabanas M V and Vallet M 1991 J. Appl. Phys. 70 1614
Behera P and Ravi S 2019 J. Electron. Mater. 48 5062
Hessien M M, Rashad M M and El Barawy K 2008 J. Magn. Magn. Mater. 320 336
Evans B J, Grandjean F, Lilot A P, Vogel R H and Gerard A 1987 J. Magn. Magn. Mater. 67 123
Johnson C E, Ridout M S and Cranshaw T E 1963 Proc. Phys. Soc. 81 1079
Kim Y J and Kim S S 2002 IEEE Trans. Magn. 383 108
Mohammed J, Sharma J, Yerima K U, Chouank T, Carol T, Basandrai D, Kumar A et al 2020 Phys. B Condens. Matter. 585 412115
Auwal I A, Güngüneş H, Güner S, Shirsath S E, Sertkol M and Baykal A 2016 Mater. Res. Bull. 80 263
Li Z W, Ong C K, Yang Z, Wei F L, Zhou X Z, Zhao J H et al 2000 Phys. Rev. B 62 6530
Dushaq G H, Mahmood S H, Bsoul I, Juwhari H K, Lahlouh B and AlDamen M A 2013 Acta Metall. Sin. 26 509
Nedkov I, Koutzarova T, Ghelev C, Lukanov P, Lisjak D, Makovec D et al 2006 J. Mater. Res. 21 2606
Acknowledgements
YKL and SC acknowledge Dr. K. V. Siva Kumar (Rtd. Professor), Department of Physics, Sri Krishnadevaraya University, Anantapur, Andhra Pradesh, India, for his continuous support and encouragement in this work. We sincerely thank Prof. M Vithal, Department of Chemistry, for the discussion. We are also thankful to UGC-NRC, School of Physics, University of Hyderabad, Telangana, for providing the XRD and FESEM facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chanda, S., Bharadwaj, S., Reddy, V.R. et al. An analysis of iron ion occupation in barium hexaferrites prepared employing different synthesis techniques from magnetic and Mossbauer studies. Bull Mater Sci 47, 81 (2024). https://doi.org/10.1007/s12034-023-03139-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-023-03139-3