Abstract
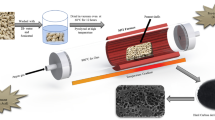
Higher proton conducting polymer blend electrolytes (HPCPBEs) of cornstarch:poly vinyl pyrrolidone (PVP):ammonium thiocyanate (NH4SCN) have been prepared by solution casting technique. Enhancement of amorphous nature of the polymer blend electrolytes have confirmed by X-ray diffraction analysis. The presence of functional groups in Fourier transform infrared spectrum shows the complexation between salt and host polymer matrix. At ambient temperature, 70 wt% NH4SCN mixed composition has obtained the maximum conductivity (1.36 × 10–4 S cm–1). The dielectric properties of the prepared samples have been analysed. The dielectric spectra and modulus spectra reveal the non-Debye behaviour of the polymer electrolytes. Low activation energy (0.14 eV) with regression value of 0.98 has been revealed from temperature dependence of ionic conductivity studies. Wagner’s polarization technique is used to confirm the conductivity due to the ions. Electrochemical behaviour of HPCPBE has been analysed by cyclic voltammetry. An electrochemical cell has fabricated using the HPCPBE and the observed open circuit voltage is 1.15 V.












Similar content being viewed by others
References
Hodge R M, Edward G H and Simon G P 1996 Polymer 37 1371
Rolland J, Poggi E, Vlad A and Gohy J F 2015 Polymer 68 344
Long L, Wang S, Xiao M and Meng Y 2016 J. Mater. Chem. A 4 10038
Kumar P and Yashonath S 2006 J. Chem. Sci. 118 135
Rudhziah S, Rani M S A, Ahmad A and Mohamed N S 2014 Polymers 6 2371
Samsudin A S and Isa M I N 2012 J. Appl. Sci. 12 174
Wu I D and Chang F C 2007 Polymer 48 989
Rao M, Geng X, Liao Y, Hu S and Li W 2012 J. Membrane Sci. 39 937
Premalatha M, Vijaya N, Selvasekarapandian S and Selvalakshmi S 2016 Ionics 22 1299
Chai M N and Isa M I N 2016 Sci. Rep. 6 27328
Hofmann A, Schulz M and Hanemann T 2013 Electrochim. Acta 89 823
Bhargav P B, Mohan V M, Sharma A and Rao V N 2009 Curr. Appl. Phys. 9 165
Laura C M, Hunter O F and Jennifer L S 2019 ACS Appl. Energy Mater. 2 6355
Sreekanth T, Jaipal Reddy M, Subramanyam S and Subba Rao U 1999 Mater. Sci. Eng. B 64 107
Ramya C S, Savitha T, Selvasekarapandian S and Hirankumar G 2005 Ionics 11 436
Xu Y and Hanna M A 2005 J. Polym. Environ. 13 221
Kadir M F Z, Majid S R and Arof A K 2010 Electrochim. Acta 55 1475
Mohamad S A, Yahya R, Ibrahim Z A and Arof A K 2007 Sol. Energy Mater. Sol. Cells 91 1194
Woo H J, Majid S R and Arof A K 2013 Solid State Ionics 252 102
Aziz N A, Majid S R and Arof A K 2012 J. Non-Cryst. Solids 358 1581
Ananda Jothi M, Vanitha D and Asath Bahadur S 2019 Int. J. Recent Technol. Eng. 8 2277
Shukur M F, Ithnin R and Kadir M F Z 2014 Electrochim. Acta 136 204
Venkata Subba Rao C, Ravi M, Raja V, Balaji Bhargav P, Sharma A K and Narasimha Rao V V R 2012 Iran. Polymer J. 21 531
Selva Kumar K and Ramesh Prabhu M 2014 Int. J. Chem. Tech. Res. 6 5740
Devi D A, Smitha B, Sridhar S and Aminabhavi T M 2006 J. Membrane Sci. 280 45
Teoh K H, Lim C-S and Ramesh S 2014 Measurement 48 87
Ramesh S and Arof A K 2001 Mater. Sci. Eng. B 85 11
Mendes J F, Paschoalin R T, Carmona V B, Neto A R S, Marques A C P, Marconcini J M et al 2016 Carbohydr. Polym. 137 452
Jinisha B, Anilkumar K M, Manoj M, Pradeep V S and Jayalekshmi S 2017 Electrochim. Acta 235 210
De Santana H, Pelisson L, Janiaski D R, Zaia C T B V and Zaia D A M 2010 J. Serbian Chem. Soc. 75 1381
Arof A K, Amirudin S, Yusof S Z and Noor I M 2014 Phys. Chem. Chem. Phys. 16 1856
Hema M, Selvasekerapandian S, Hirankumar G, Sakunthala A, Arunkumar D and Nithya H 2009 J. Phys. Chem. Solids 70 1098
Lizen F, Zhimin D, Ce-Wen N and Ming L 2002 Electrochim. Acta 48 205
Muthulakshmi S, Iyyapushpam S and Pathinettam Padiyan D 2014 Bull. Mater. Sci. 371 575
Rizwana R, Radha Krishna T, James A R and Sarah P 2007 Cryst. Res. Technol. 42 699
Mariappan C R and Govindaraj G 2002 Mater. Sci. Eng. B 94 82
Muralidharan P, Venkateswarlu M and Satyanarayana N 2004 Mater. Chem. Phys. 88 138
Aziz S B 2016 Appl. Phys. A 122 1
Woo H J, Majid S R and Arof A K 2012 Mater. Chem. Phys. 134 755
Winie T and Arof A K 2006 Ionics 12 149
Samsudin A S and Isa M I N 2014 J. Korean Phys. Soc. 65 1441
Shukur M F, Ibrahim F M, Majid N A, Ithnin R and Kadir M F Z 2013 Phys. Scr. 88 025601
Ben Taher Y, Oueslati A, Maaloul N K, Khirouni K and Gargouri M 2015 Appl. Phys. A 120 1537
Sundaramahalingam K, Nallamuthu N, Manikandan A, Vanitha D and Muthuvinayagam M 2018 Physica B: Condens. Matter 547 55
Vanitha D, Asath Bahadur S, Nallamuthu N and Athimoolam S 2018 Ionics 24 139
Dieterich W and Maass P 2002 Chem. Phys. 284 439
Rajeswari N, Selvasekarapandian S, Prabu M, Karthikeyan S and Sanjeeviraja C 2013 Bull. Mater. Sci. 36 333
Hashmi S A, Kumar A and Tripathi S K 2007 J. Phys. D: Appl. Phys. 40 6527
Kim Y S, Balland V, Limoges B and Costentin C 2017 Phys. Chem. Chem. Phys. 19 17944
Yesappa L, Niranjana M, Ashokkumar S P, Vijeth H, Basappa M, Jishnu D et al 2018 RSC Adv. 8 15297
Subba Reddy C V, Sharma A K and Narasimha Rao V V R 2003 J. Power Sources 114 338
Acknowledgement
We gratefully acknowledge the International Research Centre (IRC), Kalasalingam Academy of Research and Education, for providing facilities and equipment to carry out the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jothi, M.A., Vanitha, D., Bahadur, S.A. et al. Promising biodegradable polymer blend electrolytes based on cornstarch:PVP for electrochemical cell applications. Bull Mater Sci 44, 65 (2021). https://doi.org/10.1007/s12034-021-02350-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-021-02350-4