Abstract
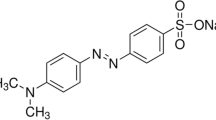
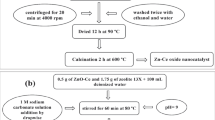
In this research, the dye photo-catalytic removal was investigated using zirconia \((\hbox {ZrO}_{2})\) nanocatalysts, zeolite (Ze), \(\hbox {ZrO}_{{2}}\)–Ze with different percentages and optimized \(\hbox {ZrO}_{{2}}\)–Ze doped via urea, copper oxide and cerium oxide. In order to determine the optimal conditions, the effects of different catalysts and parameters such as dye concentration, UV lamp power, amount of the loaded catalyst and pH were investigated. The response surface methodology was used to obtain optimal experimental conditions. Physical and chemical properties of materials were investigated by X-ray diffraction, Fourier transform infrared, scanning electron microscopy and Brunauer–Emmett–Teller. Completely methyl orange (MO)-dye removal (100%) was achieved at optimal conditions under UV light during 80 min. The optimal operational condition for MO photo-catalytic decomposition using an optimal N-doped 10 wt% \(\hbox {ZrO}_{{2}}\)–zeolite nanocatalyst was obtained at UV lamp power, pH, catalyst loading and dye concentration of 15 W, 3, 0.4 g \(\hbox {l}^{-1}\) and 5 mg \(\hbox {l}^{-1}\), respectively. Photo-catalytic degradation kinetics of MO described well using pseudo-first order which is in accordance with the Langmuir–Hinshelwood model (\(k_{\mathrm{app}} = 0.031~\hbox {min}^{-1})\).






Similar content being viewed by others
References
Mansouri M, Tanzifi M, Lotfi H and Nademi M 2017 St. Cerc. St. CICBIA 18 153
Alalm M G, Ookawara S, Fukushi D, Sato A and Tawfi A 2016 J. Hazard. Mater. 302 225
Ahmad T, Shahazad M and Phul R 2017 Mater. Sci. Eng. Int. J. 1 100
Mahadwad O K, Parikh P A, Jasra R V and Patil C 2011 Bull. Mater. Sci. 34 551
Khatamian M, Hashemian S and Sabaee S 2010 Mater. Sci. Semicond. Process. 13 156
Ökte A N and Yilmaz Ö 2008 Appl. Catal. B 85 92
Bhattacharjee S, Sarkar S H, Raybarman U and Panja J 2016 Int. J. Innov. Res. Sci. Technol. 2 284
Panpa W, Sujaridworakun P and Jinawath S 2008 Appl. Catal. B 80 271
Zhao J, Wang X, Zhang L, Hou X, Li Y and Tang C 2011 J. Hazard. Mater. 188 231
Ismail S, Ahmad Z A, Berenov A and Lockman Z 2011 Corros. Sci. 53 1156
Basahel S N, Ali T T, Mokhtar M and Narasimharao K 2015 Nanoscale Res. Lett. 10 73
Yoa S, Jia X, Jiao L, Zhu C and Shi Z 2012 Indian J. Chem. 51A 1049
Mzoughi M, Anku W W, Oppong S O B, Shukla S K, Agorku E S and Govender P P 2016 Res. Artic. Adv. Mater. Lett. 7 946
Amaladhas P T and Thavamani S S 2013 Adv. Mater. Lett. 4 688
Ivanova T, Harizanova A, Koutzarova T and Vertruyen B 2010 Cryst. Res. Technol. 45 1154
Hao Y, Li J, Yang X, Wang X and Lu L 2004 Mater. Sci. Eng. A 367 243
Manivannan M and Rajendran S 2011 Int. J. Eng. Sci. Technol. 3 8048
Devi L G and Rajashekhar K E 2011 J. Mol. Catal. A: Chem. 334 65
Xin W, Zhu D, Liu G, Hua Y and Zhou W 2012 Int. J. Photoenergy 2012 1
Barakat M 2011 J. Hydro. Environ. Res. 5 137
Zhao S, Ramakrishnan G, Shen P, Su D and Orlov A 2013 Chem. Eng. J. 217 266
Chakrabarti S and Dutta B K 2004 J. Hazard. Mater. 112 269
Mansouri M, Atashi H, Khalilipour M M, Setareshenas N and Shahraki F 2013 J. Korean Chem. Soc. 57 769
Huang C R and Shu H Y 1995 J. Hazard. Mater. 41 47
Espino-Estévez M R, Fernández-Rodríguez C, González-Díaz O M, Araña J, Espinós J P, Ortega-Méndez J A et al 2016 Chem. Eng. J. 298 82
Muruganandham M, Shobana N and Swaminathan M 2006 J. Mol. Catal. A: Chem. 246 154
Sakthivel S, Neppolian B, Murugesan L and Venkatakri S H 2003 Sol. Energy Mater. Sol. Cells. 77 65
Qamar M, Saquib M and Muneer M 2005 Dyes Pigm. 65 1
Zhu H, Jiang R, Fu Y, Guan Y, Xiaob L, Yao J et al 2012 Desalination 286 41
Nodehi A, Atashi H and Mansouri M 2019 J. Dispersion Sci. Technol. 40 766
Arabi A, Fazli M and Ehsani M H 2018 Bull. Mater. Sci. 41 77
Ahmed T, Phul R, Alam P, Lone I H, Shahazad M, Ahmed J et al 2017 RSC Adv. 7 27549
Acknowledgements
MM thanks the University of Ilam, for the award research fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mansouri, M., Mozafari, N., Bayati, B. et al. Photo-catalytic dye degradation of methyl orange using zirconia–zeolite nanoparticles. Bull Mater Sci 42, 230 (2019). https://doi.org/10.1007/s12034-019-1933-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-019-1933-y