Abstract
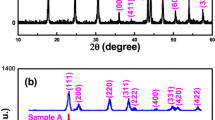
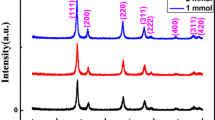
Cerium oxide (CeO2) nanopebbles have been synthesized using a facile hydrothermal method. X-ray diffraction pattern (XRD) and transmission electron microscopy analyses confirm the presence of CeO2 nanopebbles. XRD shows the formation of cubic fluorite CeO 2 and the average particle size estimated from the Scherrer formula was found to be 6.69 nm. X-ray absorption spectrum of CeO2 nanopebbles exhibits two main sharp white lines at 880 and 898 eV due to the spin orbital splitting of M 4 and M 5. Optical absorption for the synthesized CeO2 nanopebbles exhibited a blue shift (E g = 3.35 eV) with respect to the bulk CeO2 (E g = 3.19 eV), indicating the existence of quantum confinement effects.
Similar content being viewed by others
References
Campbell C T and Peden C H F 2005 Science 309 713
Tsunekawa S, Ishikawa K, Li Z Q, Kawazoe Y and Kasuya Y 2000 Phys. Rev. Lett. 85 3440
Liu G F, Wang Q Y, Zhao B, Shen M Q and Zhou R X 2011 J. Hazard. Mater. 186 911
Imanaka N, Masui T, Hirai H and Adachi G 2003 Chem. Mater. 15 2289
Atkinson A, Barnett S, Gorte R J, Irvine J T S, McEvoy A J, Mogensen M B, Singhal S and Vohs J M 2004 Nat. Mater. 3 17
Chen J, Patil, Seal S and McGinnis J G 2006 Nat. Nanotechnol. 1 142
Chen F J, Cao Y L and Jia D Z 2011 Appl. Surf. Sci. 257 9226
Masui T, Yamamoto M, Sakata T, Mori H and Adachi G Y 2000 J. Mater. Chem. 10 353
Rovira L G, Amaya J M, Haro M L, Rio E D, Hungria A B, Midgley P, Calvino J J, Bernal S and Botana F J 2009 Nano Lett. 9 1395
Yuan Q, Duan H H, Li L L, Li Z X, Duan W T, Zhang L S, Song W G and Yan C H 2010 Adv. Mater. 22 1475
Zhang D, Fu H, Shi L, Fang J and Li Q 2007 J. Solid State Chem. 180 654
Zhou K, Wang X, Sun X, Peng Q and Li Y 2005 J. Catal. 229 206
Wu L, Wiesmann H J, Moodenbaugh A R, Klie R F, Zhu Y, Welch D O and Suenaga M 2004 Phys. Rev. B69 125415
Yang S and Gao L 2006, J. Am. Chem. Soc. 128 9330
Lehnen T, Schlafer J, Mathur S and Anorg Z 2014 Anorg. Allg. Chem. 640 819
Deori K, Gupta D, Saha B and Deka S 2014 ACS Catal. 4 3169
Yang Z, Yang Y, Liang H and Liu L 2009 Mater. Lett. 63 1774
Yang Z, Zhou K, Liu X, Tian Q, Lu D and Yang S 2007 Nanotechnology 18 185606
Nguyena T D, Dinha C T, Mrabet D, Tran-Thi M N and Doa T O 2013 J. Colloid Interface Sci. 394 100
Zhang Y C, Lei M, Huang K, Liang C, Wang Y J, Ding S S, Zhang R, Fan D Y, Yang H J and Wang Y G 2014 Mater. Lett. 116 46
Mai H X, Sun L D, Zhang Y W, Si R, Feng W, Zhang H P, Liu H C and Yan C H 2005 J. Phys. Chem. B 109 24380
Zhang F, Chan S W, Spanier J E, Apak E, Jin Q, Robinson R D and Herman I P 2002 Appl. Phys. Lett. 80 127
Liyanage A D, Perera S D, Tan K, Chabal Y and Balkus K J 2014 ACS Catal 4 577
Taniguchi T, Katsumata K, Omata S, Okada K and Matsushita N 2011 Cryst. Growth Des. 11 3754
Niewa R, Hu Z, Grazioli C, Robler U, Golden M S, Knupfer M, Fink J, Giefers H, Wortmann G, de Groot F M F and DiSalvo F J 2002 J. Alloys Compd. 346 129
Wu G S, Xie T, Yuan X Y, Cheng B C and Zhang L D 2004 Mater. Res. Bull. 39 1023
Zhang F, Jin Q and Chan S W 2004 J. Appl. Phys. 95 4319
Ho C, Yu J C, Kwong T, Mak A C and Lai S 2005 Chem. Mater. 17 4514
Tsunekawa S, Fukuda T and Kasuya A 2000 J. Appl. Phys. 87 1318
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
ARUL, N.S., MANGALARAJ, D. & HAN, J.I. Facile hydrothermal synthesis of CeO2 nanopebbles. Bull Mater Sci 38, 1135–1139 (2015). https://doi.org/10.1007/s12034-015-0992-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-015-0992-y